Emerging applications of postbiotics to sustainable livestock production systems
John R Otto 1 Felista W Mwangi 2 Shedrach B Pewan 3 Benjamin WB Holman 4 Aduli EO Malau-Aduli 1
- School of Environmental and Life Sciences, College of Engineering, Science and Environment, The University of Newcastle, Callaghan, NSW 2308, Australia
- School of Medicine and Public Health, College of Health, Medicine and Wellbeing, The University of Newcastle, Callaghan, NSW 2308 Australia
- National Veterinary Research Institute, Private Mail Bag 01 Vom, Vom, Plateau State, Nigeria
- Wagga Wagga Agricultural Institute, New South Wales Department of Primary Industries & Regional Development, Wagga Wagga, NSW 2650, Australia
Article Information
- Date Received: 27/05/2025
- Date Revised: 21/06/2025
- Date Accepted: 05/07/2025
- Date Published Online: 07/07/2025
Copyright: © 2025 The Authors. Published by MARCIAS AUSTRALIA, 32 Champion Drive, Rosslea, Queensland 4812, Australia. This is an open access publication under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Citation: Otto JR, Mwangi FW, Pewan SB, Holman BWB, Malau-Aduli AEO (2025). Emerging applications of postbiotics to sustainable livestock pro-duction systems. Aust J Agric Vet Anim Sci, 1(1), 100002 https://doi.org/
Abstract
The growing challenge of drug-resistant microbes emerging from increased antibiotic use in livestock production has prompted bans and intensified research into natural feed additives for sustainable and improved animal production. While probiotics (live bacteria) have been commonly used to enhance animal health and growth, heightened concerns about probiotic stability and propensity to transfer the antibiotic resistance gene, limit their efficiency and viability. Currently, postbiotics are emerging as the more stable and natural alternative gut health promoters, in the light of their potential to increase nutrient intake, absorption, growth, fertility, and carcass quality. Postbiotics are beneficial left-over wastes (by-products and compounds) from the metabolism and digestion of probiotics and prebiotics (substrates for live bacteria) in the gut. Postbiotics include antimicrobial peptides that slow down the growth of harmful bacteria, short-chain fatty acids that help healthy bacteria flourish, amino acids, microbial cell fragments, extracellular polysaccharides, and vitamins B and K. Emerging evidence suggests that postbiotics may offer environmental benefits in mitigating microplastic contamination and reducing methane emissions in livestock production. This review examines existing gaps in the light of current knowledge on postbiotics and their impact on animal health, milk production, carcass quality, and reproductive outcomes in livestock, and proposes future research direction to foster a better understanding of the role of postbiotics in enhancing sustainable monogastric and ruminant livestock production.
Keywords
Postbiotics; antibiotic resistance; methane; microplastic; health; carcass quality; milk
Highlights
- Postbiotics offer greater stability than probiotics, hence better alternatives to antibiotics in livestock production
- Postbiotic supplements enhance livestock health, dairy and meat production through improved gut health
- Postbiotics enhance environmental sustainability via reduced microplastic contamination and methane emissions
- Further research on optimal dosage in single and combined diets with feed additives and mechanisms of action in diverse livestock is needed
1.0 Introduction
The global population is estimated to exceed 9 billion by 2050 (Gu et al., 2021). This necessitates innovation from the livestock industry so as to meet growing demands for ruminant products, especially in developing countries, where median incomes have increased and fostered dietary shifts (Jia et al., 2023). The livestock industry is a major contributor to global food security and rural economies, meaning that sustainable management practices must be adopted to remain relevant (Yitbarek, 2019; Henchion et al., 2021). Specifically, the livestock sector must improve animal productivity and ensure that product quality aligns with consumer preference for healthy, antibiotic-free, and sustainably produced foods (Ponnampalam & Holman, 2023). For ruminants, sustainability is driven by the duo of nutrient conversion efficiency and health-beneficial rumen microbiome. These two key drivers can be optimised by livestock producers through innovative feeding technologies and interventions with postbiotics.
Postbiotics are non-living bioactive compounds, that are effective natural alternatives to probiotics and antibiotics in livestock feeding systems (Şanli, 2024). Postbiotics are preferred for improving milk production and ruminal efficiency (Thomas et al., 2023), animal health and performance (Sharma et al., 2020), while antibiotics are regulated and discouraged in many markets and production regions. Unlike probiotics which pose risks of antibiotic-resistant genes and are sensitive to environmental conditions, postbiotics are stable, non-toxic, and offer consistent health benefits by promoting beneficial bacteria and inhibiting harmful pathogens within the gastrointestinal microbiome (Liu et al., 2023). Postbiotics have been reported to have anti-inflammatory, antioxidant, and immunomodulatory properties (Liu et al., 2023; Şanlı, 2024). Studies in poultry have shown that supplementation with postbiotics Lactobacillus plantarum strains, RS5, RI11 and UL4 can improve egg quality, production performance, faecal parameters and plasma cholesterol in laying hens (Loh et al., 2014), growth rates, immune responsiveness, caecal microbiota and volatile fatty acids in broilers (Kareem et al., 2015; Kareem et al., 2017), mitigate the adverse impacts of heat stress, improve antioxidant enzyme activity, lipid profile and meat quality in broilers (Humam et al., 2020). These positive outcomes were purportedly propelled by postbiotic antibacterial metabolites such as organic acids and bacteriocins, and their inhibitory effects on pathogenic bacteria proliferation (Nakamura et al., 2012; Nguyen Tien Thanh et al., 2010). Supplementation with postbiotic metabolites of Lactobacillus plantarum strains was associated with lower cooking loss and enhanced nutritional value with a positive impact on egg production (Choe et al., 2012) and broiler meat quality (Humam et al., 2020). Furthermore, piglets supplemented with postbiotic strains of Lactobacillus plantarum L-137 exhibited stronger immune responses and improved growth, particularly during the finishing stages (Tartrakoon et al., 2023).
There are comparatively fewer investigations of postbiotic effects on ruminant health, performance, and product quality. Studies in dairy cattle have reported that supplementation with postbiotic fermentation products of the Saccharomyces cerevisiae strain increased growth rates, gut health, fermentation, and milk production (Alugongo et al., 2017; Coleman et al., 2023). Published reports on the effect of postbiotics in ruminant livestock are diverse and inconsistent in the literature. For instance, supplementation of pre- and postweaning calves with Saccharomyces cerevisiae fermentation products was reported to have no effect on liveweights or blood urea nitrogen, fatty acids, insulin-like growth factor-1, glucose, and total protein, and it resulted in lower faecal scores in supplemented calves than the control (Alugongo et al., 2017). In another ruminant study, the supplementation of veal calves with a combination of Saccharomyces cerevisiae and Lactobacillus acidophilus fermentation improved growth rates and immune system responsiveness (Thorsteinsson and Vestergaard, 2020). Supplementation of lactating goats with yeast fermentation postbiotic products improved fibre digestibility and milk production, particularly protein and lactose content, without affecting fat concentration or methane emissions (Fernández et al., 2023). In weaning lambs, supplementation with Lactobacillus plantarum probiotics led to gains in liveweight, nutrient digestibility, and fibre-degrading bacteria without altering ruminal pH and blood lipid profiles (Izuddin et al., 2020). The variability in results from the aforementioned studies may be attributed to diversity in postbiotic varieties, supplementation levels, and animal species. Understanding the contributions of these variables to postbiotic effectiveness is necessary for adoption by the livestock industry.
In general, supplementation with postbiotics promotes livestock productivity and health. The mechanisms behind these outcomes seem to be tied to enhanced nutrient digestibility (Izuddin et al., 2019b) and optimisation of the microbial community with beneficial microbes within the rumen and/or gastrointestinal tract (Sharma et al., 2020). These contributions are driven mainly by the production of volatile fatty acids (VFAs), especially propionic acid, a key energy source that supports growth and fat metabolism in animals via its association with in vivo blood glucose (Liu et al., 2023). However, critical areas such as the impact of postbiotic-driven VFAs on microbiome population balance, growth, development, meat and milk qualities, the expressions of genes involved in production, reproduction and fertility, microplastics and methane emissions, remain to be fully understood. There is an imperative, therefore, to review the current literature and critique investigations of postbiotic supplementation and its effects on livestock health, growth, reproduction, nutrient intake, and carcass quality. The aim of this review was to identify key research priorities necessary to optimise postbiotic use in the livestock industry and the best practice to achieve sustainable and productive livestock systems. Only studies dealing with livestock responses to postbiotic supplementation were included in this review.
2.0 What are postbiotics?
When bacteria ferment plant polysaccharides, they produce postbiotics, including short-chain fatty acids (SCFAs), i.e., acetate, butyrate, and propionate. The effects of these SCFAs vary based on factors such as the gut environment, the host, dietary intake, and the types and amounts of SCFAs present. SCFAs, especially acetate and propionate have been reported to have potential role to play in livestock production (Mirza et al., 2020). Thus, postbiotics include short-chain fatty acids, exopolysaccharides, cell wall fragments, enzymes (Liu et al., 2023) as well as the exopolysaccharides produced by probiotic bacteria (Liu et al., 2023; Sharma et al., 2020). The specific benefits of these varieties of postbiotics can vary based on the microorganism, their molecular makeup, and the microbiome of the host animal. Ongoing research is essential to fully understand the role of exopolysaccharides in animal gut health, nutrition, and the complex dynamics of the gut ecosystem. Rumen microorganisms produce a variety of microbial enzymes that play a key role in the digestion, metabolism, and nutrient utilization in animals. These enzymes, particularly those from Lactobacillus species, i.e., E-3, E-18, Cellulases and fermentum, are abundant and have antioxidative properties (Kullisaar et al., 2002).
Postbiotics may be sourced from different microorganisms, the source of which may have implications on their activities. For example, postbiotics from various Lactobacillus species, including L. acidophilus, L. casei, L. fermentum, L. rhamnosus, L. paracasei, and L. delbrueckii, have proven effective for animal use (Islam et al., 2021; Liu et al., 2023; Sevin et al., 2021). Indeed, research involving sheep, dairy, and poultry have investigated postbiotics produced by Lactobacillus plantarum (Sharma et al., 2020), with most known postbiotics are derived from this particular strain (Sharma et al., 2020). Postbiotics from other bacteria like Bifidobacterium species, Faecalibacterium prausnitzii, and Bacillus coagulans have also been reported as viable supplements for animal use (Liu et al., 2023; Şanlı, 2024). Moreover, postbiotics for animal use have also been produced using yeast, i.e., Saccharomyces cerevisiae (Coleman et al., 2023; Thomas et al., 2023), and fungi, i.e., Aspergillus oryzae (Kaufman et al., 2021; Ríus et al., 2022).
Bacteria and yeast generate liquid metabolites known as cell-free supernatants, which contain bacteriocins and organic acids with antibacterial properties (Liu et al., 2023). These postbiotics play a vital role in promoting gut health by facilitating the growth of beneficial bacteria and preventing the colonization of harmful pathogens within the gastrointestinal tract of host animals (Liu et al., 2023). These postbiotics have been reported in livestock production (Spaggiari et al., 2022). Despite their potential, the specific functions of cell-free supernatants remain largely unknown in ruminants, as they are affected by factors such as microbial composition, growth conditions, and experimental design.
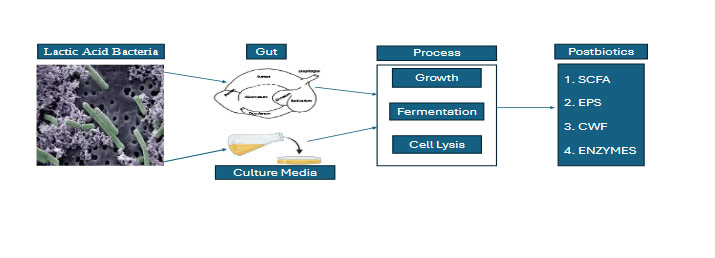
Figure 1. A schematic diagram of the process of postbiotics synthesis from lactic acid bacteria in microbiological culture media and gastrointestinal tract. SCFA, Short-chain Fatty Acids; EPS, Exopolysaccharides and CWF, Cell Wall Fragments.
3.0 Mechanisms of action of postbiotics
The microbiome of the gastrointestinal tract is a complex system that works in tandem with the gut mucosal immune system to maintain gut health. Postbiotics play a crucial role in this process by modulating the host microbiota, reducing inflammation, aiding digestion, regulating intestinal permeability, and supporting the immune system (Aghebati-Maleki et al., 2021). They support the proliferation of beneficial bacteria while inhibiting pathogenic microbes in the gastrointestinal tract (Şanlı, 2024). Unlike probiotics, postbiotics are more stable under various environmental conditions, such as changes in temperature, humidity, light, and pH (Izuddin et al., 2019b).
Postbiotics inhibit the formation of biofilms by pathogenic bacteria by affecting their quorum sensing communication network (Hossain et al., 2021; Liu et al., 2023; Şanlı, 2024; Yun et al., 2014). For instance, lipoteichoic acid and peptidoglycan postbiotics from Lactobacillus plantarum have been shown to prevent biofilm formation by Staphylococcus aureus (Tsai et al., 2021). Postbiotics activate the mucosal immune system, helping to prevent and eliminate harmful bacteria, such as Enterobacteriaceae (Izuddin et al., 2019a). They also create an acidic environment in the gut by lowering the rumen pH, making it inhabitable for harmful bacteria (Sevin et al., 2021; Wei et al., 2022).
Postbiotics antibacterial effects arise from their completion for the same binding receptors as pathogenic microorganisms in the intestinal barrier as well as from their inclusion of antibacterial compounds, i.e., bacteriocins, that deter the increment of pathogenic bacteria (Humam et al., 2021; Sevin et al., 2021; Wei et al., 2022). For example, postbiotics from Lactococcus and Lactobacillus species have demonstrated antibacterial properties against pathogens causing mastitis in dairy cattle (Bouchard et al., 2013; Malvisi et al., 2016; Pellegrino et al., 2019). Furthermore, an invitro study of anti-listeria properties of probiotics of Lactobacillus spp. demonstrated the biofilm removal activity of L. monocytogenes, indicating their antibacterial ability that can be applied in livestock production (Moradi et al., 2019).
Exopolysaccharides, a component of postbiotics produced by the lactic bacteria Lactobacillus paracasei, support the production of beneficial VFA, including butyric and propionic acids, when consumed by another gut microbiota (Dias et al., 2018; Izuddin et al., 2019a, 2019b; Martyniak et al., 2021). The production and absorption of propionic acid enhance blood glucose levels, as propionic acid is a precursor to glucose. The rise in propionic acid and glucose levels in ruminants, particularly dairy cows, helps regulate negative energy balance during the transition period (Coleman et al., 2023; Şanlı, 2024). Additionally, propionic acid reduces hydrogen production in the rumen, which is crucial for methanogens and methane production (Izuddin et al., 2019b; Jeyanathan et al., 2014). Long-term use of postbiotics can lead to increased levels of blood total protein, because of the increased production of blood urea nitrogen (Şanlı, 2024). However, postbiotics have not been reported to affect blood triglycerides, since they do not digest or absorb crude fat (Izuddin et al., 2019b). Postbiotics also contribute to the development and enlargement of rumen papillae (Izuddin et al., 2019a). These enlarged papillae increase the surface area available for nutrient absorption and facilitate the adherence of beneficial microorganisms to the rumen wall.
Lipoteichoic acid is a postbiotic with potent anti-inflammatory properties that can benefit ruminants by modulating their immune responses. It acts to reduce excessive inflammation by regulating the activity of macrophages, neutrophils, T and B lymphocytes, and monocytes (Liu et al., 2023; Şanlı, 2024). This postbiotic achieves its effects by controlling cytokine production in intestinal mucosal cells and interleukin levels, which aids in reducing inflammation (Lee et al., 2022; Liu et al., 2023; Şanlı, 2024).
Ruminants naturally produce reactive oxygen species (ROS) during metabolic processes (Lin et al., 2018; Thibessard et al., 2004). When the balance tips towards higher oxidant activity and reduced antioxidant defences, ruminants can experience oxidative stress (Lombardi et al., 2017). This imbalance can result in tissue damage, potentially lead to organ failure if not properly managed, as well as result in reduced meat and milk shelf-life and quality (Ponnampalam et al., 2022). Postbiotic supplementation offers a promising means to ameliorate the effects of ROS, by enhancing the activity of antioxidant enzymes such as catalase, superoxide dismutase (SOD), and glutathione peroxidase, which help mitigate oxidative damage to DNA, tissues, and organs (Şanlı, 2024). For instance, a study by Izuddin et al. (Izuddin et al., 2020) demonstrated that postbiotic of Lactobacillus plantarum supplementation increased serum and ruminal glutathione peroxidase (GPX) enzymes in lambs. Meanwhile, Coleman et al. (Coleman et al., 2023) observed a reduction in ferric reducing antioxidant power (FRAP) levels in plasma of Holstein cows fed with Saccharomyces cerevisiae fermentation product postbiotic to 139 μmol/L compared to control at 149 μmol/L. Postbiotic supplementation has additional effects on ruminants. For example, an in vitro fermentation study has shown that postbiotic from Lactobacillus plantarum RG14 increases the population of cellulolytic bacteria such as F. succinogenes and R. flavefaciens, in rumen fluid of goats, which are essential for digestion of dry matter (DM) and neutral detergent fibre (NDF) (Izuddin et al., 2018). Similar research from the same group, found that long-term supplementation of postbiotics from Lactobacillus plantarum RG14 in lambs leads to lower leukocyte and platelet counts, indicating an enhanced anti-inflammatory response to infections (Izuddin et al., 2019a). However, short-term supplementation of postbiotics of Saccharomyces cerevisiae fermentation product was observed not to have the same anti-inflammatory effect on Holstein cows (Coleman et al., 2023). This highlights the potential need for sustained supplementation to achieve the desired immune-modulating effects in ruminants. Research to confirm this supplementation duration effect is necessary to interpret postbiotic effects on livestock.
4.0 Benefits of Postbiotics on Livestock Production
4.1. Animal health
Animal health is a critical concern for the livestock industry in Australia, where health-related issues can lead to substantial economic losses each year (Bell et al., 2018; Lane et al., 2015). Previously, antibiotic drugs were widely used to combat these issues. However, the overuse of these drugs has contributed to antimicrobial resistance, prompting restrictions to their use for promoting growth and health in animals (Şanlı, 2024; Sharma et al., 2020). This has prompted research towards finding natural alternatives for animal feed additives that are both safe for animals and humans without compromising animal welfare (Sharma et al., 2020). The microbiome of the gastrointestinal tract plays a vital role in animal health by supporting immune function, enhancing digestion and nutrient absorption, and preventing the growth of harmful bacteria (Cristofori et al., 2021). When this balance is disrupted, it can impair gut barrier function, leading to health complications and disease susceptibility in animals (Wu et al., 2022). In response, probiotics and prebiotics have been investigated for use as natural growth promoters and preventive treatments for pathogens in livestock (Lambo et al., 2021). Probiotics, which are live microorganisms, can improve livestock health by regulating the gut microbiota, enhancing nutrient absorption, supporting immune responses, preventing inflammation, preventing liver damage, and bone health, and enhancing brain function (Carpi et al., 2022; de Sire et al., 2022; Lambo et al., 2021; Trzeciak and Herbet, 2021). However, environmental factors like temperature and pH can compromise their effectiveness, limiting their large-scale use. Instead, postbiotics are more stable, resilient to environmental changes (pH and temperature), and can be mass-produced without losing their beneficial properties (Hossain et al., 2021; Thorakkattu et al., 2022). Studies have shown that postbiotics have immunomodulatory, anti-inflammatory, antibacterial and antioxidant effects, helping to strengthen the intestinal barrier, promote nutrient intake, and maintain a healthy gut microbiota (Fathima et al., 2022; Ríus et al., 2022; Scott et al., 2022; Štofilová et al., 2022; Vinderola et al., 2022). They modulate the immune response to enhance well-being and downgrade foodborne pathogens in poultry (Kareem et al., 2017). These actions show postbiotics to be a promising natural alternative to antibiotic drugs, with comparative advantages to probiotics (Sharma et al., 2020).
Research has demonstrated the effectiveness of postbiotics in improving livestock health. For instance, supplementing post-weaning lambs with postbiotics Lactobacillus plantarum RG14 led to increased rumen papillae height and width, and reduction of pathogenic Enterobacteriaceae population in the jejunum without affecting beneficial lactic acid bacteria, suggesting that postbiotics could assist in regulating the gut microbiome to support animal health by eliminating harmful pathogens (Izuddin et al., 2019a). Similarly, a study of Lohman Brown layers found that the supplementation of postbiotics metabolites of L. plantarum (TL1, RS5, RG14, RG11 and RI11) lowered faecal pH, increased lactic acid bacteria population in the gut, and reduced the prevalence of pathogenic Enterobacteriaceae (Loh et al., 2014). When supplemented to broiler chicks, postbiotics Lactobacillus plantarum RG14 combined with inulin was reported to increase beneficial bacterial populations of Bifidobacteria and reducing Enterobacteria and E. coli pathogens populations in the gastrointestinal tract (Kareem et al., 2017). This combination also modulated the immune response by influencing cytokine production (Kareem et al., 2017). Research of piglets reported that postbiotic metabolites of Lactobacillus plantarum supplementation reduced diarrheal incidents (Loh et al., 2013) and prevented the colonization of the gut by harmful pathogenic bacteria (Humam et al., 2021; Price et al., 2010). Postbiotics derived from Lactococcus and Lactobacillus strains have also been shown to produce antibacterial compounds, i.e., bacteriocins, that restrict the proliferation of pathogens that cause diseases like mastitis in ruminants (Malvisi et al., 2016; Pellegrino et al., 2019; Sevin et al., 2021). These examples demonstrate the potential application of postbiotics to enhance livestock health. However, further research is needed to fully understand their mechanisms and optimize their use, especially in ruminants. Studies should focus on identifying the most effective strain and dosages for specific conditions while ensuring quality control for practical applications. Importantly research should assess the impact of postbiotics on gut microbiome dysbiosis and eubiosis, especially looking into the influence of beneficially bacteria population. By improving livestock health, postbiotics offer a real means to optimise livestock resilience and productivity in modern production system.
4.2. Growth performance
Postbiotics play a crucial role in enhancing livestock growth by optimizing feed efficiency, improving nutrient intake and digestion, and maximizing nutrient utilization (Fernández et al., 2023; Xu et al., 2023). They affect and regulate the expression of genes that are associated with growth (Humam et al., 2019; Kareem et al., 2016). These benefits translate into faster growth rates, ultimately improving overall animal performance. By harnessing the multifaceted roles of postbiotics, producers can adopt more efficient and productive livestock management. Growth performance of livestock affects feed costs, market access, production, management costs, reproductive performance and culling rates. Postbiotics enhance feed efficiency by modifying metabolism, augmenting digestion and lowering bacterial enzyme activity (Urban et al., 2024). This has real implications. For example, a study of Large White × Landrace pigs found that feed conversion ratio (FCR) was correlated with growth traits (Arthur et al., 2009). Furthermore, by improving the FCR for a steer by just 10%, a feedlot can significantly enhance its profitability. This improvement in FCR and residual feed intake (RFI) resulted in cost savings of $0.14 and $0.05 per kilogram of gain, respectively. As a result, each steer generated an additional profit of $34.65, demonstrating how growth performance and feed efficiency can lead to substantial financial rewards in livestock operations (Retallick et al., 2013).
A study on post-weaning lambs found that supplementation with 0.9% postbiotic derived from Lactobacillus plantarum RG14 resulted in improvements to growth performance, nutrient intake, and nutrient digestibility (Izuddin et al., 2019b). These benefits were linked to enhanced rumen fermentation characteristics, an increase in ruminal cellulolytic bacteria populations, and favourable blood metabolite profiles (Izuddin et al., 2019b). An invitro study also corroborated the findings that postbiotic Lactobacillus plantarum RG14 contributed to better digestion by promoting the regeneration of the mucosa and intestinal walls (Izuddin et al., 2018). Additionally, the upregulation of hepatic IGF-1 and ruminal MCT-1 mRNA facilitated greater IGF-1 production in the liver and more efficient uptake of VFA through the ruminal epithelium (Izuddin et al., 2019b). In this same study, post-weaning lamb dieting on Lactobacillus plantarum RG14 had higher rumen papillae length and width, lower concentration of IgA in jejunum mucosa and population of Enterobacteriaceae in the jejunum, downregulated the expression of proinflammatory cytokines, IL-1β and TNF and anti-inflammatory cytokines, IL-10 mRNA and upregulated the expression of tight junction protein (TJP-1), claudin-1(CLDN-1) and claudin-4 (CLDN-4) genes. In a similar study, lamb that were supplemented with postbiotics of L. plantarum extract presented higher concentrations of serum and ruminal glutathione peroxidase (GPX) enzymes (Izuddin et al., 2020). In addition, expressions of GPX1, GPX4, and Cu/Zn Superoxide dismutase (SOD) genes were observed in Dorper lambs. In the same study, mechanism of regulation of rumen barrier function was observed through the expression of tight junction protein (TJP), occludin (OCLD), claudin-1 (CLDN1) and claudin-4 (CLDN4). Based on these studies, it was observed that postbiotics as a feed additive can improve ruminal epithelium growth, improve serum and ruminal antioxidant activity, immune status, and upregulate the intestinal barrier function and hepatic antioxidant enzymes to promote healthy gut that will promote growth of postweaning lambs.
Livestock must be resilient to various stressors, ranging from metabolic and nutritional challenges to pathogen exposure, diseases, handling, transportation, and environmental pressures (Burdick et al., 2011; Kaufman et al., 2021; Martens, 2023; Ríus et al., 2022; Thomas et al., 2023). These stressors can negatively impact feed intake and weaken the immune system, ultimately affecting growth (Izuddin et al., 2019b). For instance, weaning period is an important stressor for young animals, heavily influenced by their nutrition and environment (Karakus, 2014). Stress during weaning leads to reduced feed intake, which hampers growth rates and increases susceptibility to disease, thereby diminishing post-weaning performance (Freitas-de-Melo et al., 2022; Izuddin et al., 2019b; Karakus, 2014). Similarly, when young steers first arrive at a feedlot, the stress of transition can make them more vulnerable to illness, adversely affecting growth rates and carcass quality (Burdick et al., 2011). The transition period from prepartum to postpartum in lactating animals is another stressful phase, often leading to negative energy balance (NEBAL) and a higher risk of metabolic diseases, which in turn affects growth performance of primiparous dairy ruminants (Martens, 2023). Heat stress further exacerbates challenges for both young and lactating animals, leading to decreased growth and performance (Kaufman et al., 2021; Ríus et al., 2022). In dairy cattle, post-birth stress, particularly from E. coli infections in calves, poses a significant challenge (Galvão et al., 2005). Effective stress management is, therefore, essential for enhancing animal production. This involves nutritional interventions aimed at boosting health and performance across livestock species.
Postbiotics are being explored as potential feed additives that could replace antibiotics and complement probiotics, with the goal of improving rumen fermentation, immune function, and nutrient intake, absorption and utilisation, thereby improving livestock production through faster growth and development (Liu et al., 2023; Şanlı, 2024). Although systematic studies on the use of postbiotics in livestock are still limited, initial research has shown promising results. For example, adding postbiotics to the diets of piglets, broilers, and layers has been associated with improved growth performance (Kareem et al., 2016; Loh et al., 2014; Loh et al., 2010, 2013; Thu et al., 2011). A study conducted by Rius et al. (Ríus et al., 2022) demonstrated that administering a postbiotic derived from Aspergillus oryzae to Holstein calves under heat stress conditions enhanced energy-use efficiency, water absorption, and intestinal permeability. In a different study conducted in heifers, feeding postbiotics of Saccharomyces cerevisiae fermentation product (SCFP) to heat and humidity stressed animals improved their feed efficiency leading to greater body condition score (Thomas et al., 2023). Feeding postbiotics from cell wall products of yeast to new arrival cattle at feedlot has been shown to enhance energy metabolism and nutrient utilization during immune challenges, without triggering lipolysis or muscle catabolism (Sanchez et al., 2014).
Stresses in the environment may arise as a result of inadequate nutrition, high temperature and humidity, metabolic and infectious diseases, transportation and handling, physical and social separation from the dam which have the potential to adversely affect growth performance, productivity, health and welfare of the animals. Previous studies have reported the potential of feeding postbiotics to improve growth performances (Davis et al., 2004; Tartrakoon et al., 2023) in livestock during stressful conditions (Kaufman et al., 2021; Ríus et al., 2022; Thomas et al., 2023). The use of postbiotics in livestock has shown meaningful promise in improving liveweight gain and growth by promoting cellulolytic bacteria, which improve nutrient digestion and feed conversion efficiency. While previous studies focussed on poultry, there is limited research on ruminants, especially beef cattle, goats, and sheep. To fully unlock the potential of postbiotics in ruminants and integrate them into livestock management practices, this research gap must be addressed.
4.3. Milk production
The Holstein-Friesian dairy cattle have high genetic merit producing on average 6,000 litres of milk per year (Dairy Australia, 2023). High milk production is critical to meeting the rising demand of burgeoning global population (FAO, 2023), but serious consequences of negative energy balance (NEBAL) affect high-producing dairy cows during peripartum and postpartum, which can deplete energy reserves and lead to hormonal imbalances, metabolic diseases and fertility decline (Ford et al., 2024; Martens, 2023; Tufarelli et al., 2024; Wang et al., 2024). NEBAL results from decreased dry matter intake during peak lactation, leading to remobilisation of stored fat and glycogen (Anwar et al., 2022; Martens, 2023). This raises non-esterified fatty acids (NEFA), contributing to metabolic issues like ketosis and fatty liver disease, compromising milk production, reproduction and immune health (Abuelo et al., 2019; Anwar et al., 2022; Bouvier-Muller et al., 2016; Churakov et al., 2021; Leduc et al., 2021; Štolcová et al., 2024). To address NEBAL, farmers often supplement cattle with fermentable carbohydrates that can lower rumen pH (Huo et al., 1970), but may also disrupt the microbiota balance of the rumen and cause dysbiosis (Chaucheyras-Durand et al., 2012; Huo et al., 1970). Postbiotics could have application in these circumstances, particular as they aid in the production short-chain fatty acids, particularly propionic acid (Liu et al., 2023; Şanlı, 2024). This improves blood glucose level which assists in enhancing the energy status of lactating animal (Liu et al., 2023; Şanlı, 2024; Sharma et al., 2020). By enhancing energy and nutrient efficiency, postbiotics can support milk production and prevent metabolic diseases in dairy animals during peripartum and postpartum periods, making them a valuable feed supplement for modern dairy farming.
Previous research has shown that adding postbiotics of yeast fermentation product to the diet of lactating dairy goats can enhance both milk yield and composition (Fernández et al., 2023). Specifically, in this same study, the inclusion of yeast fermentation products was linked to increased levels of fat, protein, lactose, and nitrogen in milk. There was an increase in propionate levels and increased digestibility of NDF and ADF. Nitrogen intake and excretion in faeces and urine remained unchanged, but nitrogen content in milk increased. No changes were detected in DMI and ammonia-N levels were lower for goats supplemented with postbiotics. These improvements were proposed to be achieved through the promotion of better fibre digestibility, increasing ruminal propionate production, and reducing energy losses typically caused by methane production. A study of lactating ewes revealed that supplementing their diet with yeast product postbiotics, which are high in mannan-oligosaccharides, beta-glucans, and organic selenium, led to enhanced energy balance, increased milk production, improved milk composition, and better oxidative status (Christodoulou et al., 2023). Additionally, supplemented yeast product reduced the expression of proinflammatory genes during the peripartum phase (Christodoulou et al., 2023).
In a specific study, supplementing Probisan-Ruminants® postbiotics at 8 g/day before calving and 15 g/day after calving to primiparous and multiparous resulted in increased DMI and improved apparent tract digestibility of organic matter and neutral detergent fibre (Vicente et al., 2024). In the same study, supplementation with both Probisan-Ruminants® and Probisan-Ruminants-C® led to higher colostrum immunoglobulin levels and increased milk yield, with enhanced fat and protein content. The supplementation of 38 g/day of Saccharomyces cerevisiae fermentation products (SCFP) postbiotics has been shown to mitigate the severe impacts of grain-based subacute ruminal acidosis (SARA) on the rumen microbiota, particularly affecting the fibrolytic and lactate-producing bacteria of the Ruminococcaceae and Lachnospiraceae families (Guo et al., 2024). SARA affects microbial robustness leading to accumulation of organic acid volatile fatty acid that reduces the rumen pH and digestive tract barrier function causing accumulation of toxic immunogenic compounds of bacterial lipopolysaccharides (LPS) and histidine (Plaizier et al., 2012), resulting in reduced milk production and efficiency of production (Guo et al., 2024).
A study found that the supplementation of postbiotics derived from Saccharomyces cerevisiae in combination with a starch to dairy cows improved milk production (Dias et al., 2018). The cows produced more milk, including energy-corrected milk (ECM), and had increases in both fat and protein content. The supplementation also raised propionate levels, enhanced microbial nitrogen synthesis and increased rumen pH. Importantly, dietary nitrogen was more efficiently incorporated into milk true protein without increasing dry matter intake. In addition, the treatment reduced rumen ammonia levels, plasma haptoglobin, and rumen lactate concentrations. The improvements in milk yield were attributed to better fibre digestion and greater microbial nitrogen synthesis (Dias et al., 2018). A study conducted by Poppy et al. (Poppy et al., 2012) found similar results, although a decrease in dry matter intake was observed with supplementation. During late gestation and lactation periods, sows that consumed yeast-derived postbiotics showed increased backfat deposition and higher levels of lactose in their milk. Additionally, they produced more immunoglobulins (A and G) and transforming growth factor-β, which are essential for improving immune function and health of their piglets. By enhancing digestion, immunomodulatory effect, reducing oxidative stress, and improving nutrient absorption, postbiotics contribute to higher efficiency in milk production (Coleman et al., 2023; Fernández et al., 2023; Malvisi et al., 2016). The aforementioned studies provide insight into postbiotics effects on lactating animals, although the variability in their results, design, and the limited scope of these investigations suggest that further research is needed. Although in vitro studies with mostly postbiotics from lactobacilli strains show promising results on lactating animals, lack of sufficient data on in vivo studies, especially in ruminants, on milk solids, blood metabolites, VFA, methane emission warrant elucidation. Importantly, there is a paucity of studies on the impact of postbiotics on the saturated, monounsaturated, and polyunsaturated fatty acid profiles of milk.
Table 1. Effect of Saccharomyces cerevisiae postbiotic fermentation product supplementation on Holstein cows and sows
| Species | Main Effect | Reference |
| Holstein |
|
(Hristov et al., 2010) |
| Sow |
|
(Tsai et al., 2016) |
| Holstein |
|
(Jiang et al., 2018) |
| Holstein |
|
(Acharya et al., 2017) |
| Holstein |
|
(Olagaray et al., 2019) |
Note: ADFI, average daily feed intake, BW, body weight, IgA, Immunoglobulin A, IgG, immunoglobulin G.
Mastitis is a costly and prevalent infection of the ammary gland that impacts dairy producers via associated reductions to milk yield, altered milk composition, reduced fertility, udder inflammation, and increased treatment expenses and culling rates. Accounting for 38% of direct production costs, mastitis is influenced by factors like diet and gut health, with animals in NEBAL particularly susceptible due to weakened immune defences (Das et al., 2018; Sharun et al., 2021; Bouvier-Muller et al., 2016). Postbiotics could, therefore, offer an alternative to conventional treatments for mastitis in dairy cows (Aghebati-Maleki et al., 2021). Postbiotics inhibit pathogens like Streptococcus by lowering intracellular pH, disrupting cell membranes, and fostering a gut environment favourable to beneficial bacteria to combat harmful microorganisms (Homayouni Rad et al., 2021; Aguilar-Toalá et al., 2018). Bacteriocins postbiotics break down bacterial cells, inhibiting growth and vital bacterial functions (Gálvez et al., 2007; O’Connor et al., 2020). Notably, short-chain fatty acids such as propionate and butyrate stimulate growth of beneficial bacteria like Lactobacillus while reducing growth of harmful species such as Enterobacter. This shift in gut microbiota strengthens the blood-milk barrier, reduces inflammation, and lowers the expression of inflammatory genes, thus supporting mastitis prevention (Wang et al., 2024; Ali et al., 2021). For example, Lactobacillus sakei postbiotics were reported to contribute to a > 70% reduction in biofilm formation by pathogens like Staphylococcus aureus and Streptococcus agalactiae (Sevin et al., 2021). While these findings are promising, research on postbiotics on mastitis is still developing. More in vivo studies are needed to clarify the mechanisms, safety, and potential applications of postbiotics in mastitis prevention. Expanding this research could unlock new ways to enhance dairy cow health through targeted microbiota support and immune resilience.
4.4. Reproductive performance
Reproductive performance and fertility are economically important aspects of the profitable livestock production (Bormann et al., 2006; Kumar et al., 2017). In livestock production, fertility refers to ability of female to initiate oestrous early, conceive and sustain a pregnancy when inseminated during ovulation with quality semen from the male counterpart (Sasaki et al., 2016). Parameters for fertility are days open, calving intervals, conception rate, calving to first service interval, services per conception, first-service conception rate, calving to conception interval, pregnancy rate, nonreturn rate, neonate size, weaning weight, and quality semen (Bormann et al., 2006; Darwash et al., 1997; Kumaresan and Srivastava, 2022; Sasaki et al., 2016). Pregnancy occurs when viable egg is ovulated and inseminated at the right moment, alongside a balanced hormone cycle of oestradiol and progesterone (Darwash et al., 1997). Follicle development is driven by a complex hormonal system involving gonadotrophin releasing hormone, follicle stimulating hormone, luteinising hormone, and oestrogens and progestins, along with ovarian inhibin (Otto et al., 2014). After conception, the embryo must produce enough interferon-tau to prevent luteolysis by blocking the activity of endometrial oxytocin receptors (Kowalczyk et al., 2021; Madureira et al., 2024; Shabunin et al., 2019). Ruminants experience significant embryo losses, for example, in dairy cows a combination of early to late embryo losses approximate to 40% after 32 and 74 days of positive insemination (Moreira et al., 2001; Thatcher et al., 2019).
Clinical mastitis is a popular costly disease in the dairy industry, impacting not only the mammary gland but also reproductive performance. It affects dairy cattle, beef cattle, goats, sheep, and swine (Gabli et al., 2019; Gerjets et al., 2011; Gerjets and Kemper, 2009; Kumar et al., 2017). In dairy animals, mastitis has been linked to poor reproductive performance due to disrupted hormonal balance, reduced oocyte quality, fertilization failure, and an unfavourable uterine environment (Kumar et al., 2017). Infected animals often develop fever, which raises cytokine levels, leading to impaired oocyte maturation, disrupted uterine function, and hindered embryo development (Al-Katanani et al., 2002; Hansen et al., 2004). Studies confirm that fever from bacterial infections, both positive and negative, directly affects oocyte and embryo development (Al-Katanani et al., 2002; Gendelman et al., 2010; Wenz et al., 2001; Wilson et al., 2008), while also indirectly harming reproduction by reducing feed intake and body condition (Maltz et al., 1997).
Postbiotics can effectively offer reproductive health benefits when supplemented to livestock (Díaz Cano et al., 2021; Oguey and Thayer, 2024; Zaleska et al., 2015). Lactic acids, antimicrobial compounds, such as bacteriocins (Liu et al., 2023; Şanlı, 2024) and antioxidant (Aghebati-Maleki et al., 2021; Humam et al., 2021; Izuddin et al., 2020) inhibit harmful bacteria, i.e., E. coli, Listeria monocytogenes, Salmonella typhimurium, Enterococci, Staphylococcus aureus and Trueperella pyogenes (Choe et al., 2012; Hashem et al., 2024; Thanh et al., 2010), which cause uterine and mastitis diseases in dairy animals (Deng et al., 2015; Pellegrino et al., 2019). When administered intravaginally, mixtures of Lactobacillus sakei and Pediococcus acidilactici have been reported to reduce endometrial inflammation in postpartum dairy cows (Ametaj et al., 2014; Deng et al., 2015).
Administering postbiotics derived from Pichia guilliermondii yeast to sows during gestation resulted in a higher number of live-born piglets, and heavier litters with better survival rate that were weaned early (Oguey and Thayer, 2024). In rabbits, postbiotics from lactic acid bacteria have been found to improve semen quality, with a higher percentage of spermatozoa exhibiting acrosome integrity (Díaz Cano et al., 2021). Moreover, Lactococcus lactis, a probiotic that produces antimicrobial and anti-inflammatory compounds, has been found to reduce uterine inflammation and enhance fertility in buffaloes by decreasing endometritis (Hashem et al., 2024). Yeast supplemented to sows throughout their reproductive cycle showed no significant effect on litter size (Kim et al., 2008). In cows, supplementation with Saccharomyces cerevisiae yeast, the size of the first ovulatory follicle and pre-ovulatory surges of oestradiol tended to be higher postpartum, however, no changes were observed in the days to first ovulation, the number of follicular waves from parturition until ovulation, and the size of the first dominant follicle (AlIbrahim et al., 2010). The variability in results presented in the literature indicates that further research is needed to understand the roles of postbiotics in livestock reproduction and fertility regulation. Research is needed to explore the varying effects of different postbiotic strains, dosages, and administration methods across livestock species. Understanding these factors will help optimize their use for improving reproductive performance and overall productivity in the industry.
4.5. Environment
The global demand for meat and dairy products is rapidly rising, driving the need for intensified livestock production to maintain animal health and enhance growth (Rojas-Downing et al., 2017). However, climate change, driven by methane, carbon dioxide, and nitrous gas emissions, poses significant risks to livestock production by affecting feed quality, animal performance, milk yield, water availability, reproduction, and disease prevalence (Jeyanathan et al., 2014; Króliczewska et al., 2023; Rojas-Downing et al., 2017). To address this, policy makers are introducing incentives to industry adaption, for example, the Australian government introduced Emissions Reduction Fund (ERF) to help livestock producers reduce greenhouse gas (GHG) emissions (Black et al., 2021). To remain competitive globally, producers must adopt sustainable practices that reduce methane emissions and maintain productivity for high-quality livestock products (meat and milk). Research to reduce rumen methanogenesis is currently extensive, however, the application of postbiotics to achieve this outcome remain to be confirmed.
4.5.1. Methane emission
Methane (CH4) has 28-fold more global warming potential of carbon dioxide and is a major contributor to emissions, particularly from ruminants (Króliczewska et al., 2023). Globally, enteric fermentation in ruminants is responsible for about 30-40% of methane emissions (Lascano et al., 2012), with methane and nitrous oxide comprising 71% of total agricultural emissions (Suybeng et al., 2019). Methane is usually produced during the process OF fermentation to produce VFA (Canto, 2024; Johnson and Johnson, 1995), leading researchers to explore ways to reduce methanogenesis in rumen (Black et al., 2021; Króliczewska et al., 2023; Lascano et al., 2012), including using postbiotics and probiotics (Izuddin et al., 2019b; Jeyanathan et al., 2019) to manipulate rumen biochemical pathways to prevent methane emission. The mechanism is, rumen indwelling methanogens use hydrogen (H2), a fermentation by-product, to reduce CO2 and produce CH4 (Jeyanathan et al., 2014). However, postbiotics promote increased propionate synthesis by Propionibacterium spp. that reduces molecular H2 produced in the rumen, thus lowering methane production and enhancing energy intake from the diet by the animal (Şanlı, 2024).
In lactating goats, postbiotics significantly reduced CH4 emissions by 46 g/kg of milk fat, 97 g/kg of milk protein, and 3 g/kg of milk, with no overall difference in daily emissions. This suggests postbiotic supplementation during late lactation improves energy efficiency by reducing CH4 emissions relative to edible product yield (Fernández et al., 2023). Lactating primiparous cows feeding on a high-starch, high-fibre diet showed a numerical increase in CH₄ emissions when supplemented with Propionibacterium, with a 27% rise in CH₄ intensity per kg of milk. In contrast, supplementation with Lactobacillus bulgaricus or Lactobacillus pentosus had no effect on daily methane output and emission intensity (Jeyanathan et al., 2019). Early lactating dairy cows supplemented with active dried yeast of Saccharomyces cerevisiae did not affect total CH4 emissions but tended to increase CH4 per unit of feed intake and energy output (Muñoz et al., 2017). In a study conducted by Chung et al. (Chung et al., 2011), non-lactating Holstein cows, feeding an active dried strain of Saccharomyces cerevisiae yeast probiotics reduced CH4 emission intensity by 10%. Postbiotic supplementation in lambs led to a lower population of ruminal methanogens and protozoa (Izuddin et al., 2019b). The lamb consuming postbiotics had increased synthesis of propionate which was suggested to reduce methane production and improve energy retention (Izuddin et al., 2019b). Since propionate production utilizes molecular hydrogen (H2), a key precursor for methane, the reduced availability of molecular H2 likely results in decreased methane formation, reflecting a decline in the ruminal methanogen population (Şanlı, 2024). These results indicate that postbiotics have the propensity to reduce CH4 over probiotics in livestock, especially in ruminants of lactating animals. However, previous results on the effect of postbiotics on CH4 are inconsistent and conflicting, likely due to variations in strain, dosage, animal species, age, physiological status, and production systems. A few previous studies have focused on dairy and young animals, with limited research on beef and meat-producing livestock. Future research should aim to standardize the effects of postbiotics in lactating animals and explore their impact on meat producing animals. This will help unlock the commercial potential of postbiotics in the livestock industry and better understand their innovative benefits.
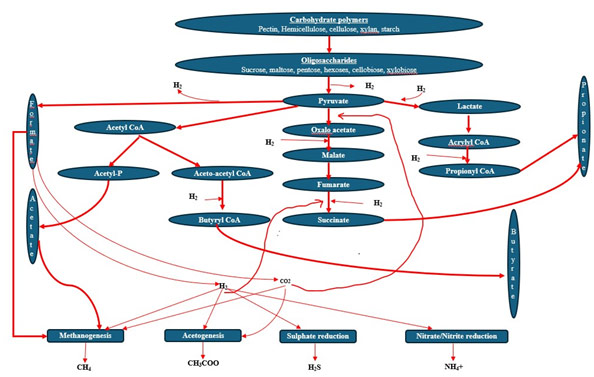
Figure 2: The potential of postbiotics to modulate rumen biochemical pathways to enhance propionate production that will decrease methanogenesis by utilizing available molecular hydrogen (H2). Adapted from Jeyanathan et al., (2014).
4.5.2. Microplastics and nanoplastics
Plastics typically enter animals through ingestion, traveling from the mouth and oesophagus to the stomach and intestines. Once absorbed into the bloodstream, these particles may contaminate milk, blood and muscle tissues. The first signs of toxicity manifested in the gastrointestinal tract are dysbiosis, damaged intestinal mucosa, and affected production of VFA and amino acids (Fackelmann and Sommer, 2019; Qiao et al., 2021; Xie et al., 2021). Previous research has largely focused on micro- and nanoplastic pollution in aquatic environments, overlooking their impact on livestock (Auta et al., 2017; Wang et al., 2016). Understanding how these plastics enter the food chain through livestock is critical for assessing risks to human health. This review explores the potential benefits of using postbiotics in the livestock industry to mitigate the risks posed by micro- and nanoplastics contamination.
The increasing demand for plastics, particularly polystyrene, is expected to drive global plastic production to 33 billion tons by 2030 (Bazeli et al., 2023; Yee et al., 2021; Yin et al., 2021). Polystyrene has a stable crystal structure and high molecular weight make it a preferred choice across various industries (Bazeli et al., 2023; Yin et al., 2021). However, this growth poses significant environmental and health concerns, particularly the rising accumulation of microplastics and nanoplastics, which have been detected in animal-derived products such as fish and meat (Azevedo-Santos et al., 2019; Geyer et al., 2017; Kedzierski et al., 2020). Microplastics (up to 5mm) and nanoplastics (as small as 0.1µm) (Alimi et al., 2018; Andrady, 2011; Lee et al., 2015; Yin et al., 2018) can enter the gastrointestinal systems of animals (Jani et al., 2011; Jin et al., 2019; Qiao et al., 2021; Walczak et al., 2015), leading to contamination of products intended for human consumption (Azevedo-Santos et al., 2019; Kedzierski et al., 2020). Once absorbed, these plastics disrupt the gut barrier, leading to microbial imbalances (dysbiosis) (Djouina et al., 2024; Fackelmann and Sommer, 2019; Hirt and Body-Malapel, 2020; Xie et al., 2021), which can result in serious health implications for both animals and humans (Bazeli et al., 2023; Chen et al., 2019; Hirt and Body-Malapel, 2020). In livestock, this microbial imbalance can impair essential functions like fibre digestion and nutrient absorption, affecting overall animal health, reproduction and production.
The potential contamination of livestock industry with microplastics could potentially have devastating health consequences (Lackner and Branka, 2024; Urli et al., 2023). Postbiotics, metabolites of inactive probiotics can help maintain gut barrier integrity and support immune function, potentially counteracting the harmful effects of microplastic-induced gut dysbiosis and inflammation (Liu et al., 2023; Xie et al., 2021).
Since microplastics are known to disrupt the gut microbiome (Fackelmann and Sommer, 2019; Qiao et al., 2021), postbiotics could be effective in restoring microbial balance (Liu et al., 2023). Their antioxidant properties may help mitigate the oxidative stress and cellular damage caused by these plastics (Humam et al., 2021; Izuddin et al., 2020). Given these benefits, postbiotics could reduce the likelihood of microplastic-induced gut barrier permeability, a critical concern in livestock exposed to these contaminants. However, more research is needed to explore the specific mechanisms by which postbiotics could mitigate the effects of micro- and nanoplastics in livestock, especially in ruminants. While previous studies have largely focused on the broader protective effects of postbiotics on gut health, targeted research in this area could help safeguard the livestock industry from plastic contamination.
4.6. Postbiotics and carcass characteristics
The primary objective of the meat industry is to deliver products that meet modern consumers’ expectations such as health benefits, flavour, and juiciness – among other meat eating characteristics. Some factors affecting meat eating include intramuscular fat levels, fat melting points, carcass pH decline rate, drip loss, meat colour, hot carcass weight, retail yield, tenderness, juiciness, and flavour (Doski and Yaseen, 2023; Mohammed and Kareem, 2022; Mwangi et al., 2019; Tripathi and Karim, 2011). Fat deposition and the fatty acid composition of meat significantly contribute to these eating characteristics (Malau-Aduli et al., 1998; Malau-Aduli et al., 1997). With the modern consumers increasingly now aware of the contribution of these meat quality attributes to health and nutrition, the market demand now heavily favours meat products that not only meet high sensory organoleptic standards but also offer health benefits. Therefore, the meat quality characteristics of livestock carcass are essential, as it impacts on market demand and consumer preferences.
In response to these evolving market demands, attempts by livestock producers in exploring natural alternative feed additives for improving carcass characteristics and meat eating quality attributes are continuing and gaining momentum (Wagner et al., 2016). The livestock industry is also investigating feed additives that can enhance both live animal performance and meat quality characteristics (Sanchez et al., 2014; Tripathi and Karim, 2011). While most previous research on ruminant carcass quality has focused on probiotics, postbiotics remain largely underexplored in this area. Current studies on postbiotics primarily centre on poultry, leaving a significant gap in knowledge regarding their potential benefits for ruminants.
Research in heat-stressed broilers supplemented with postbiotics from Lactobacillus plantarum found no significant impact on carcass yield or individual organ weights such as breast, leg, wing, gizzard, spleen, and heart (Humam et al., 2020). Studies using METALAC postbiotics and inulin + postbiotics observed no changes in carcass yield and quality (Jansseune et al., 2024; Kareem et al., 2015). Although carcass parameters like breast, thigh, and gizzard showed no significant differences across treatments, abdominal fat was notably reduced in broilers supplemented with postbiotics, and growth hormone receptor (GHR) and IGF-1 mRNA expression levels were higher (Danladi et al., 2022). Meat quality aspects such as drip loss, pH, and colour remained unchanged with combined postbiotic and phytobiotic feeding (Doski and Yaseen, 2023), though one study found that L. plantarum postbiotics reduced drip and cooking losses (Mohammed and Kareem, 2022). Additionally, dietary postbiotics significantly improved the relative weights of breast and leg muscles at specific supplementation levels, yet overall meat quality indicators like water holding capacity, surface and internal pH, and cooking loss showed no significant variation among treatment groups (Fang et al., 2024). While the findings are promising, the variability in the data highlights a need for further research to clarify postbiotics’ impact on carcass yield and quality. Establishing a consistent foundation will enable farmers to use these insights as reliable benchmarks to enhance meat production outcomes in poultry.
Previous studies in ruminants also show promise, but inconsistencies in results still remain. For example, a study conducted by Sanchez et al. (Sanchez et al., 2014) found that feeding yeast cell wall postbiotics to crossbred heifers enhanced their energy metabolism. This improvement helped to reduce both lipolysis and protein breakdown, leading to an increase in body weight of 8–11 kg. These results suggest that yeast cell wall postbiotic can play a key role in promoting efficient weight gain in livestock that can be translated to carcass yield. Increase body weight affects meat quality (Zomeño et al., 2023). Research by Tripathi and Karim (Tripathi and Karim, 2011) indicates that feed additives such as yeast products can improve live weight gain without compromising carcass quality, while also affecting the composition of various primal cuts. A previous study by Geng et al. (Geng et al., 2016) found that feeding active dry yeasts and yeast cultures to a finishing bull improved growth performance. Finishing bull dieting on these yeast cultures, increased their intramuscular fat content increased by 40.38% and 43.17%, respectively. These findings suggest that the addition of Saccharomyces cerevisiae yeasts to finishing bull could improve meat quality through intramuscular fat deposition. On the other hand, a study by Hinman et al. (Hinman et al., 1998) found that yeast culture did not influence any of the carcass characteristics measured of hot carcass weight and marbling but increased the propionate VFA. In another study, feeding Saccharomyces cerevisiae fermentation product reduced hot carcass weight but had no effect on the fat thickness at the 12th rib (Swyers et al., 2014). The most striking effect of supplementing with Saccharomyces cerevisiae yeasts (Dos Reis et al., 2024; Phesatcha et al., 2021), probiotics (Qadis et al., 2014; Rabelo et al., 2017), and postbiotics (Izuddin et al., 2019b) is the significant increase in the production of VFA, particularly propionate. Volatile fatty acids are key products of probiotic bacterial fermentation of polysaccharides (Liu et al., 2023). However, VFAs, including acetate, butyrate, and propionate, are essential components of postbiotics (Liu et al., 2023). Notably, propionate plays a vital role in hepatic gluconeogenesis, aiding in glucose production and thereby regulating carbohydrate metabolism and inhibiting cholesterol synthesis (Bush and Milligan, 1971; Hood et al., 1972). Increased ruminal production of propionate can enhance glucose production in the liver, which in turn supports fatty acid synthesis and intramuscular fat, which are essential factors in improving carcass quality (Bush and Milligan, 1971). As glucose serves as the primary carbon source for this synthesis, supplementing livestock diets with postbiotics can improve meat quality by enhancing intramuscular fat (Smith and Crouse, 1984), a crucial grading criterion in markets such as Australia, Japan, and the USA. Therefore, including postbiotics in animal feed may promote rumen fermentation that enhances VFA production, ultimately improving carcass characteristics and overall meat quality in livestock.
5. Challenges and Future Directions
5.1. Research Gaps and Future Research Suggestions:
- Postbiotics as a Natural Feed Additive: Postbiotics offer a promising alternative to antibiotics and probiotics, supporting animal health through immunomodulatory, anti-inflammatory, antibacterial, and antioxidant effects. However, studies on their effectiveness, particularly in small and large ruminants, show mixed results. Further research on their impact on rumen microbiomes, especially beneficial bacteria, is needed.
- Impact on Growth Performance: Growth performance is critical in livestock due to its effects on feed costs, carcass quality, reproductive success, and production. Research on postbiotics in enhancing growth has mainly focused on poultry, with limited insights into meat-producing ruminants like steers, sheep, and goats. More studies could clarify their benefits in these animals.
- Milk Yield and Composition: While postbiotics’ effects on milk yield, fat, protein, and related blood metabolites show promise, particularly in the U.S. production systems, research in Australia’s pasture-based systems is limited. Key areas needing investigation include mastitis mitigation, milk quality parameters, and milk fatty acid profiles, especially long-chain polyunsaturated fats.
- Reproductive Health and Fertility: Reproductive success is essential in livestock, yet postbiotics’ role in fertility factors such as sperm quality, ovulation, gestation intervals, and hormone regulation is largely unexplored. Their potential to reduce oxidative stress in sperm and oocytes, and support key hormones like progesterone, FSH, LH, prostaglandin and estrogen warrants further study.
- Carcass yield and Meat Quality: The consumer awareness of meat quality attributes to their health and nutrition is growing. The influence of postbiotics on ruminant meat’s intramuscular fat, fatty acid composition, and sensory organoleptic traits is an emerging interest that warrants further elucidation. Research has focused on poultry, leaving a gap in the understanding of ruminants.
- Environmental Impact and Methane Emission: While the role of methane emissions in adverse environmental outcomes is well known, research on how postbiotics might reduce these emissions in livestock production is limited. Additionally, postbiotics may mitigate the effects of microplastics and nanoplastics in livestock, though the mechanisms remain unclear and need further exploration for practical application.
Therefore, to fulfill the identified research gaps, the following objectives must be addressed:
- Evaluate how postbiotic feed additives influence the microbiome of both small and large ruminants and determine their effects on overall animal health.
- Investigate how feeding postbiotics to ruminants impacts growth traits, including live weight, body condition score, feed conversion efficiency, and nutrient intake, absorption and utilization.
- Study the influence of postbiotic supplementation on lactation parameters, blood metabolites, and fatty acid profiles in dairy animals. Additionally, explore the potential effects of postbiotics on reducing mastitis incidence.
- Analyse the impact of postbiotics on fertility metrics such as sperm quality, ovulation cycles, return to oestrus, service per conception and calving intervals. Assess changes in key reproductive hormones including progesterone, prostaglandin, luteinizing hormone, follicle-stimulating hormone, and estrogen.
- Explore how postbiotic supplements affect carcass characteristics and meat quality attributes, such as intramuscular fat, fat melting point, fatty acid profile, and sensory organoleptic characteristics.
- Assess the role of postbiotics in reducing methane emissions and microplastic accumulation in livestock products.
6.0 Conclusion
In summary, postbiotics represent a promising advancement for the livestock industry, with potential benefits spanning animal health, productivity, reproduction, environmental impact, and economic viability. As bioactive compounds generated by probiotics, postbiotics offer diverse advantages, from enhancing growth and fertility outcomes to improving milk production and carcass quality. This review highlights their potential to support gut health, immunity, and nutrient absorption, which translates into more resilient animals, improved feed efficiency, and reduced disease risk, ultimately lowering the need for antibiotics and associated costs. However, there remains a substantial knowledge gap regarding the effects of postbiotics on specific areas, including rumen microbiome, growth traits in ruminants, milk fatty acid profiles, meat quality traits, methane emission, reproductive hormones and fertility traits. Filling these gaps could pave the way for even more targeted applications, with profound, long-term implications for livestock health and performance. Emerging research also points to the potential for postbiotics to mitigate methane emissions and microplastic residues in meat and milk, addressing critical environmental and consumer health concerns. As modern consumers increasingly prioritise healthy, antibiotic-free products, postbiotics offer a great solution to this need. By addressing key challenges in livestock health, productivity, reproduction, environmental impact, and economic sustainability, postbiotics hold significant value for farmers, researchers, and industry stakeholders alike. Further research will be vital to unlocking their full potential in shaping a more sustainable and profitable future for the livestock industry.
Author Contributions: Conceptualization: John Roger Otto, Benjamin William Behrens Holman; Investigation and Methodology: John Roger Otto, Benjamin William Behrens Holman, Felista Waithira Mwangi, Shedrach Benjamin Pewan, Aduli Enoch Othniel Malau-Aduli; Project Administration: John Roger Otto, Supervision: Aduli Enoch Othniel Malau-Aduli; Validation: Aduli Enoch Othniel Malau-Aduli, Benjamin William Behrens Holman, Felista Waithira Mwangi, Shedrach Benjamin Pewan; Writing – Original draft: John Roger Otto, Benjamin William Behrens Holman; Writing – Review & Editing: Aduli Enoch Othniel Malau-Aduli, Shedrach Benjamin Pewan, Felista Waithira Mwangi, Benjamin William Behrens Holman. All authors have read and agreed to the published version of the manuscript.
Funding: Being a literature review, this research received no external funding.
Ethics Approval Statement: Although the study was conducted in accordance with the Declaration of Helsinki, a formal ethics approval by an Institutional Ethics Committee was not applicable since it was a literature review that did not involve the use of animals.
Informed Consent Statement: Not applicable since the literature review did not involve humans.
Data Availability Statement: Not applicable for literature reviews.
Acknowledgments: The authors would like to acknowledge the School of Environmental and Life Sciences, College of Engineering, Science, and Environment, The University of Newcastle, New South Wales, Australia, for providing computer resources, library and office space.
Conflicts of Interest: The authors declare no conflicts of interest.
Artificial Intelligence: AI was not used for this review.
References
Abuelo A, Hernández J, Benedito JL, Castillo C. 2019. Redox biology in transition periods of dairy cattle: Role in the health of periparturient and neonatal animals. Antioxidants, 8, 20. https://doi.org/10.3390/antiox8010020
Acharya S, Pretz JP, Yoon I, Scott MF, Casper DP. 2017. Effects of Saccharomyces cerevisiae fermentation products on the lactational performance of mid-lactation dairy cows. Translational Animal Science, 1, 221–228. https://doi.org/10.2527/tas2017.0028
Aghebati-Maleki L, Hasannezhad P, Abbasi A, Khani N. 2021. Antibacterial, antiviral, antioxidant, and anticancer activities of postbiotics: A review of mechanisms and therapeutic perspectives. Biointerface Research in Applied Chemistry, 12, 2629–2645. https://doi.org/10.33263/BRIAC122.26292645
Al-Ibrahim RM, Crowe MA, Duffy P, O‘Grady L, Beltman ME, Mulligan FJ. 2010. The effect of body condition at calving and supplementation with Saccharomyces cerevisiae on energy status and some reproductive parameters in early lactation dairy cows. Animal Reproduction Science, 121, 63–71. https://doi.org/10.1016/j.anireprosci.2010.04.183
Alimi OS, Farner Budarz J, Hernandez LM, Tufenkji N. 2018. Microplastics and nanoplastics in aquatic environments: Aggregation, deposition, and enhanced contaminant transport. Environmental Science & Technology, 52, 1704–1724. https://doi.org/10.1021/acs.est.7b05559
Al-Katanani YM, Paula-Lopes FF, Hansen PJ. 2002. Effect of season and exposure to heat stress on oocyte competence in Holstein cows. Journal of Dairy Science, 85, 390–396. https://doi.org/10.3168/jds.S0022-0302(02)74086-1
Alonso ME, González-Montaña JR, Lomillos JM. 2020. Consumers’ concerns and perceptions of farm animal welfare. Animals, 10, 385. https://doi.org/10.3390/ani10030385
Alugongo GM, Xiao JX, Chung YH, Dong SZ, Li SL, Yoon I, Wu ZH, Cao ZJ. 2017. Effects of Saccharomyces cerevisiae fermentation products on dairy calves: Performance and health. Journal of Dairy Science, 100, 1189–1199. https://doi.org/10.3168/jds.2016-11399
Ametaj BN, Iqbal S, Selami F, Odhiambo JF, Wang Y, Gänzle MG, Dunn SM, Zebeli Q. 2014. Intravaginal administration of lactic acid bacteria modulated the incidence of purulent vaginal discharges, plasma haptoglobin concentrations, and milk production in dairy cows. Research in Veterinary Science, 96, 365–370. https://doi.org/10.1016/j.rvsc.2014.02.007
Andrady AL. 2011. Microplastics in the marine environment. Marine Pollution Bulletin, 62, 1596–1605. https://doi.org/10.1016/j.marpolbul.2011.05.030
Anwar MM, El-Mokadem MY, Ramadan TA, Nour El-Din ANM, Taha TA, Sharaby MA, Salem MH, Rashad AMA. 2022. Implications of negative energy balance in lactating Barki and Rahmani ewes. Animal Production Science, 62, 1777–1788. https://doi.org/10.1071/AN22100
Arthur PF, Giles LR, Eamens GJ, Barchia IM, James KJ. 2009. Measures of growth and feed efficiency and their relationships with body composition and carcass traits of growing pigs. Animal Production Science, 49, 1105. https://doi.org/10.1071/AN09061
Auta HS, Emenike CU, Fauziah SH. 2017. Distribution and importance of microplastics in the marine environment: A review of the sources, fate, effects, and potential solutions. Environment International, 102, 165–176. https://doi.org/10.1016/j.envint.2017.02.013
Azevedo-Santos VM, Gonçalves GRL, Manoel PS, Andrade MC, Lima FP, Pelicice FM. 2019. Plastic ingestion by fish: A global assessment. Environmental Pollution, 255, 112994. https://doi.org/10.1016/j.envpol.2019.112994
Bazeli J, Banikazemi Z, Hamblin MR, Sharafati CR. 2023. Could probiotics protect against human toxicity caused by polystyrene nanoplastics and microplastics? Frontiers in Nutrition, 10. https://doi.org/10.3389/fnut.2023.1186724
Bell J, Gali J, Jakeman G. 2018. In: Acil Allen Consulting. Economic Contribution of Animal Health Products to Australia’s Livestock Industries, 2015-16, June 2018. A report submitted to Animal Medicines Australia, August 8, 2018. Accessible at: https://animalmedicinesaustralia.org.au/wp-content/uploads/2019/10/AMA-Economic-Contribution-Final-Report-9-August-2018.pdf
Black JL, Davison TM, Box I. 2021. Methane emissions from ruminants in Australia: Mitigation potential and applicability of mitigation strategies. Animals, 11, 951. https://doi.org/10.3390/ani11040951
Bormann JM, Totir LR, Kachman SD, Fernando RL, Wilson DE. 2006. Pregnancy rate and first-service conception rate in Angus heifers1. Journal of Animal Science, 84, 2022–2025. https://doi.org/10.2527/jas.2005-615
Bouchard DS, Rault L, Berkova N, Le Loir Y, Even S. 2013. Inhibition of Staphylococcus aureus invasion into bovine mammary epithelial cells by contact with live Lactobacillus casei. Applied and Environmental Microbiology, 79, 877–885. https://doi.org/10.1128/AEM.03323-12
Bouvier-Muller J, Allain C, Enjalbert F, Tabouret G, Portes D, Caubet C, Tasca C, Foucras G, Rupp R. 2016. Response to dietary-induced energy restriction in dairy sheep divergently selected for resistance or susceptibility to mastitis. Journal of Dairy Science, 99, 480–492. https://doi.org/10.3168/jds.2015-9785
Burdick NC, Randel RD, Carroll JA, Welsh TH. 2011. Interactions between temperament, stress, and immune function in cattle. International Journal of Zoology, 2011, 1–9. https://doi.org/10.1155/2011/373197
Bush RS, Milligan LP. 1971. Study of the mechanism of inhibition of ketogenesis by propionate in bovine liver. Canadian Journal of Animal Science, 51, 121–127. https://doi.org/10.4141/cjas71-016
Canto F. 2024. Enteric fermentation. CGIAR Research Program on Climate Change, Food Security and Agriculture (CCAFS), AgLEDx Resource Platform. https://agledx.ccafs.cgiar.org/emissions-led-options/sources-sinks/enteric-fermentation/.
Carpi RZ, Barbalho SM, Sloan KP, Laurindo LF, Gonzaga HF, Grippa PC, Zutin TLM, Girio RJS, Repetti CSF, Detregiachi CRP, Bueno PCS, Mazuqueli PESB, Goulart RA, Haber JFS. 2022. The effects of probiotics, prebiotics and synbiotics in Non-Alcoholic Fat Liver Disease (NAFLD) and Non-Alcoholic Steatohepatitis (NASH): A systematic review. International Journal of Molecular Sciences, 23, 8805. https://doi.org/10.3390/ijms23158805
Chaucheyras-Durand F, Chevaux E, Martin C, Forano E. 2012. Use of yeast probiotics in ruminants: Effects and mechanisms of action on rumen pH, fibre degradation, and microbiota according to the diet. In: Probiotic in Animals. Edited by Everlon Rigobelo. https://www.intechopen.com/books/2991. Published 03 October 2012, ISBN 978-953-51-0777-4, eBook (PDF) ISBN Doi 10.5772/3319 978-953-51-4274-4 https://doi.org/10.5772/50192
Chen Q, Zhang H, Allgeier A, Zhou Q, Ouellet JD, Crawford SE, Luo Y, Yang Y, Shi H, Hollert H. 2019. Marine microplastics bound dioxin-like chemicals: Model explanation and risk assessment. Journal of Hazardous Materials, 364, 82–90. https://doi.org/10.1016/j.jhazmat.2018.10.032
Choe DW, Loh TC, Foo HL, Hair-Bejo M, Awis QS. 2012. Egg production, faecal pH and microbial population, small intestine morphology, plasma and yolk cholesterol in laying hens given liquid metabolites produced by Lactobacillus plantarum strains. British Poultry Science, 53, 106–115. https://doi.org/10.1080/00071668.2012.659653
Christodoulou C, Skourtis A, Kyriakaki P, Satolias FF, Karabinas D, Briche M, Salah N, Zervas G, Mavrommatis A, Tsiplakou E. 2023. The effect of dietary supplementation with probiotic and postbiotic yeast products on ewe milk performance and immune oxidative status. Journal of Fungi, 9, 1139. https://doi.org/10.3390/jof9121139
Chung YH, Walker ND, McGinn SM, Beauchemin KA. 2011. Differing effects of 2 active dried yeast (Saccharomyces cerevisiae) strains on ruminal acidosis and methane production in non-lactating dairy cows. Journal of Dairy Science, 94, 2431–2439. https://doi.org/10.3168/jds.2010-3277
Churakov M, Karlsson J, Edvardsson RA, Holtenius K. 2021. Milk fatty acids as indicators of negative energy balance of dairy cows in early lactation. Animal, 15, 100253. https://doi.org/10.1016/j.animal.2021.100253
Coleman DN, Jiang Q, Lopes MG, Ritt L, Liang Y, Aboragah A, Trevisi E, Yoon I, Loor JJ. 2023. Feeding a Saccharomyces cerevisiae fermentation product before and during a feed restriction challenge on milk production, plasma biomarkers, and immune function in Holstein cows. Journal of Animal Science, 101. https://doi.org/10.1093/jas/skad019
Cristofori F, Dargenio VN, Dargenio C, Miniello VL, Barone M, Francavilla R. 2021. Anti-inflammatory and immunomodulatory effects of probiotics in gut inflammation: A door to the body. Frontiers in Immunology, 12. https://doi.org/10.3389/fimmu.2021.578386
Dairy Australia. 2023. Australian Dairy Industry in Focus. Southbank, Melbourne, Victoria, Australia. Accessible at: https://www.dairyaustralia.com.au/en/industry-reports/australian-dairy-industry-in-focus
Danladi Y, Loh TC, Foo HL, Akit H, Tamrin NA, Naeem Azizi M. 2022. Effects of postbiotics and paraprobiotics as replacements for antibiotics on growth performance, carcass characteristics, small intestine histomorphology, immune status and hepatic growth gene expression in broiler chickens. Animals, 12, 917. https://doi.org/10.3390/ani12070917
Darwash AO, Lamming GE, Woolliams JA. 1997. Estimation of genetic variation in the interval from calving to postpartum ovulation of dairy cows. Journal of Dairy Science, 80, 1227–1234. https://doi.org/10.3168/jds.S0022-0302(97)76051-X
Davis ME, Maxwell CV, Erf GF, Brown DC, Wistuba TJ. 2004. Dietary supplementation with phosphorylated mannans improves growth response and modulates immune function of weanling pigs. Journal of Animal Science, 82, 1882–1891. https://doi.org/10.2527/2004.8261882x
de Sire A, de Sire R, Curci C, Castiglione F, Wahli W. 2022. Role of dietary supplements and probiotics in modulating microbiota and bone health: The gut-bone axis. Cells, 11, 743. https://doi.org/10.3390/cells11040743
Deng Q, Odhiambo JF, Farooq U, Lam T, Dunn SM, Ametaj BN. 2015. Intravaginal lactic acid bacteria modulated local and systemic immune responses and lowered the incidence of uterine infections in periparturient dairy cows. PLoS One, 10, e0124167. https://doi.org/10.1371/journal.pone.0124167
Dias ALG, Freitas JA, Micai B, Azevedo RA, Greco LF, Santos JEP. 2018. Effect of supplemental yeast culture and dietary starch content on rumen fermentation and digestion in dairy cows. Journal of Dairy Science, 101, 201–221. https://doi.org/10.3168/jds.2017-13241
Díaz CJV, Argente MJ, García ML. 2021. Effect of postbiotic based on lactic acid bacteria on semen quality and health of male rabbits. Animals, 11, 1007. https://doi.org/10.3390/ani11041007
Djouina M, Loison S, Body-Malapel M. 2024. Recent progress in intestinal toxicity of microplastics and nanoplastics: Systematic review of preclinical evidence. Microplastics, 3, 217–233. https://doi.org/10.3390/microplastics3020013
Dos Reis BQQ, Sarturi JO, Silva K, Karges K, Johnson BJ, Rodrigues B, Goulais M. 2024. Effects of Saccharomyces cerevisiae and dietary type offered to beef cattle on intake, feeding behavior, nutrient digestibility, and ruminal fermentation. Journal of Animal Science, 102, 331–332. https://doi.org/10.1093/jas/skae234.378
Doski JMM, Yaseen KK. 2023. The effects of different levels of postbiotic and phytobiotic combination as feed additives on carcass, lipid profile, meat quality, and tibia bone in broiler chickens. Kirkuk University Journal for Agricultural Sciences, 14, 227–241. https://doi.org/10.58928/KU23.14324
Fackelmann G, Sommer S. 2019. Microplastics and the gut microbiome: How chronically exposed species may suffer from gut dysbiosis. Marine Pollution Bulletin, 143, 193–203. https://doi.org/10.1016/j.marpolbul.2019.04.030
Fang S, Fan X, Xu S, Gao S, Wang T, Chen Z, Li D. 2024. Effects of dietary supplementation of postbiotic derived from Bacillus subtilis ACCC 11025 on growth performance, meat yield, meat quality, excreta bacteria, and excreta ammonia emission of broiler chicks. Poultry Science, 103, 103444. https://doi.org/10.1016/j.psj.2024.103444
FAO. 2023. Food & Agriculture Organisation. Dairy Market Review – Emerging trends and outlook in 2023. Rome, Italy. https://openknowledge.fao.org/handle/20.500.14283/cc9105en
Fathima S, Shanmugasundaram R, Adams D, Selvaraj RK. 2022. Gastrointestinal microbiota and their manipulation for improved growth and performance in chickens. Foods, 11, 1401. https://doi.org/10.3390/foods11101401
Fernández C, Romero T, Badiola I, Díaz-Cano J, Sanzol G, Loor JJ. 2023. Postbiotic yeast fermentation product supplementation to lactating goats increases the efficiency of milk production by enhancing fiber digestibility and ruminal propionate, and reduces energy losses in methane. Journal of Animal Science, 101. https://doi.org/10.1093/jas/skac370
Ford HR, Mitchell TM, Scull T, Benitez OJ, Strieder-Barboza C. 2024. The effect of subclinical ketosis on the peripheral blood mononuclear cell inflammatory response and its crosstalk with depot-specific preadipocyte function in dairy cows. Animals, 14, 1995. https://doi.org/10.3390/ani14131995
Freitas-de-Melo A, Orihuela A, Hötzel MJ, Ungerfeld R. 2022. What do we know and need to know about weaning in sheep? An overview of weaning practises, stress and welfare. Frontiers in Animal Science, 3, 823188. https://doi.org/10.3389/fanim.2022.823188
Gabli Z, Djerrou Z, Gabli AE, Bensalem M. 2019. Prevalence of mastitis in dairy goat farms in Eastern Algeria. Veterinary World, 12, 1563–1572. https://doi.org/10.14202/vetworld.2019.1563-1572
Galvão KN, Santos JEP, Coscioni A, Villaseñor M, Sischo WM, Berge ACB. 2005. Effect of feeding live yeast products to calves with failure of passive transfer on performance and patterns of antibiotic resistance in fecal Escherichia coli. Reproduction Nutrition Development, 45, 427–440. https://doi.org/10.1051/rnd:2005040
Gendelman M, Aroyo A, Yavin S, Roth Z. 2010. Seasonal effects on gene expression, cleavage timing, and developmental competence of bovine preimplantation embryos. Reproduction, 140, 73–82. https://doi.org/10.1530/REP-10-0055
Geng C, Ren L, Zhou Z, Chang Y, Meng Q. 2016. Comparison of active dry yeast (Saccharomyces cerevisiae) and yeast culture for growth performance, carcass traits, meat quality and blood indexes in finishing bulls. Animal Science Journal, 87, 982–988. https://doi.org/10.1111/asj.12522
Gerjets I, Kemper N. 2009. Coliform mastitis in sows: A review. Journal of Swine Health and Production, 17, 97–105. https://doi.org/10.54846/jshap/583
Gerjets I, Traulsen I, Reiners K, Kemper N. 2011. Comparison of virulence gene profiles of Escherichia coli isolates from sows with coliform mastitis and healthy sows. Veterinary Microbiology, 152, 361–367. https://doi.org/10.1016/j.vetmic.2011.05.002
Geyer R, Jambeck JR, Law KL. 2017. Production, use, and fate of all plastics ever made. Science Advances, 3. https://doi.org/10.1126/sciadv.1700782
Gu D, Andreev K, Dupre M. 2021. Major trends in population growth around the world. China CDC Weekly, 3, 604–613. https://doi.org/10.46234/ccdcw2021.160
Guo J, Zhang Z, Guan LL, Yoon I, Plaizier JC, Khafipour E. 2024. Postbiotics from Saccharomyces cerevisiae fermentation stabilize microbiota in rumen liquid digesta during grain-based subacute ruminal acidosis (SARA) in lactating dairy cows. Journal of Animal Science and Biotechnology, 15, 101. https://doi.org/10.1186/s40104-024-01056-x
Hansen PJ, Soto P, Natzke RP. 2004. Mastitis and fertility in cattle – possible involvement of inflammation or immune activation in embryonic mortality. American Journal of Reproductive Immunology, 51, 294–301. https://doi.org/10.1111/j.1600-0897.2004.00160.x
Hashem NM, Essawi WM, El-Demerdash AS, El-Raghi AA. 2024. Biomolecule-producing probiotic bacterium Lactococcus lactis in free or nano-encapsulated form for endometritis treatment and fertility improvement in buffaloes. Journal of Functional Biomaterials, 15, 138. https://doi.org/10.3390/jfb15060138
Henchion M, Moloney AP, Hyland J, Zimmermann J, McCarthy S. 2021. Review: Trends for meat, milk and egg consumption for the next decades and the role played by livestock systems in the global production of proteins. Animal, 15, 100287. https://doi.org/10.1016/j.animal.2021.100287
Hinman DD, Sorensen SJ, Momont PA, Albin R, Cole NA. 1998. Effect of yeast culture on steer performance, apparent diet digestibility, and carcass measurements when used in a barley and potato finishing diet. The Professional Animal Scientist, 14, 173–177. https://doi.org/10.15232/S1080-7446(15)31819-2
Hirt N, Body-Malapel M. 2020. Immunotoxicity and intestinal effects of nano- and microplastics: A review of the literature. Particle and Fibre Toxicology, 17, 57. https://doi.org/10.1186/s12989-020-00387-7
Hood RL, Thompson EH, Allen CE. 1972. The role of acetate, propionate, and glucose as substrates for lipogenesis in bovine tissues. International Journal of Biochemistry, 3, 598–606. https://doi.org/10.1016/0020-711X(72)90018-3
Hossain MI, Mizan MFR, Roy PK, Nahar S, Toushik SH, Ashrafudoulla M, Jahid IK, Lee J, Ha SD. 2021. Listeria monocytogenes biofilm inhibition on food contact surfaces by application of postbiotics from Lactobacillus curvatus B.67 and Lactobacillus plantarum M.2. Food Research International, 148, 110595. https://doi.org/10.1016/j.foodres.2021.110595
Hristov AN, Varga G, Cassidy T, Long M, Heyler K, Karnati SKR, Corl B, Hovde CJ, Yoon I. 2010. Effect of Saccharomyces cerevisiae fermentation product on ruminal fermentation and nutrient utilization in dairy cows. Journal of Dairy Science, 93, 682–692. https://doi.org/10.3168/jds.2009-2379
Humam AM, Loh TC, Foo HL, Izuddin WI, Awad EA, Idrus Z, Samsudin AA, Mustapha NM. 2020. Dietary supplementation of postbiotics mitigates adverse impacts of heat stress on antioxidant enzyme activity, total antioxidant, lipid peroxidation, physiological stress indicators, lipid profile and meat quality in broilers. Animals, 10, 982. https://doi.org/10.3390/ani10060982
Humam AM, Loh TC, Foo HL, Izuddin WI, Zulkifli I, Samsudin AA, Mustapha NM. 2021. Supplementation of postbiotic RI11 improves antioxidant enzyme activity, upregulated gut barrier genes, and reduced cytokine, acute phase protein, and heat shock protein 70 gene expression levels in heat-stressed broilers. Poultry Science, 100, 100908. https://doi.org/10.1016/j.psj.2020.12.011
Huo W, Zhu W, Mao S. 1970. Effects of feeding increasing proportions of corn grain on concentration of lipopolysaccharide in the rumen fluid and the subsequent alterations in immune responses in goats. Asian-Australasian Journal of Animal Science, 26, 1437–1445. https://doi.org/10.5713/ajas.2013.13143
Islam MZ, Uddin ME, Rahman MT, Islam MA, Harun-ur-Rashid M. 2021. Isolation and characterization of dominant lactic acid bacteria from raw goat milk: Assessment of probiotic potential and technological properties. Small Ruminant Research, 205, 106532. https://doi.org/10.1016/j.smallrumres.2021.106532
Izuddin WI, Humam AM, Loh TC, Foo HL, Samsudin AA. 2020. Dietary postbiotic Lactobacillus plantarum improves serum and ruminal antioxidant activity and upregulates hepatic antioxidant enzymes and ruminal barrier function in post-weaning lambs. Antioxidants, 9, 250. https://doi.org/10.3390/antiox9030250
Izuddin WI, Loh TC, Foo HL, Samsudin AA, Humam AM. 2019a. Postbiotic L. plantarum RG14 improves ruminal epithelium growth, immune status and upregulates the intestinal barrier function in post-weaning lambs. Scientific Reports, 9, 9938. https://doi.org/10.1038/s41598-019-46076-0
Izuddin WI, Loh TC, Samsudin AA, Foo HL, Humam AM, Shazali N. 2019b. Effects of postbiotic supplementation on growth performance, ruminal fermentation and microbial profile, blood metabolite and GHR, IGF-1 and MCT-1 gene expression in post-weaning lambs. BMC Veterinary Research, 15, 315. https://doi.org/10.1186/s12917-019-2064-9
Izuddin WI, Loh TC, Samsudin AA, Foo HL. 2018. In vitro study of postbiotics from Lactobacillus plantarum RG14 on rumen fermentation and microbial population. Revista Brasileira de Zootecnia, 47. https://doi.org/10.1590/rbz4720170255
Jani P, Halbert GW, Langridge J, Florence AT. 2011. Nanoparticle uptake by the rat gastrointestinal mucosa: Quantitation and particle size dependency. Journal of Pharmacy and Pharmacology, 42, 821–826. https://doi.org/10.1111/j.2042-7158.1990.tb07033.x
Jansseune SCG, Lammers A, van Baal J, Blanc F, van der Laan MHP, Calenge F, Hendriks WH. 2024. Diet composition influences probiotic and postbiotic effects on broiler growth and physiology. Poultry Science, 103, 103650. https://doi.org/10.1016/j.psj.2024.103650
Jeyanathan J, Martin C, Eugène M, Ferlay A, Popova M, Morgavi DP. 2019. Bacterial direct-fed microbials fail to reduce methane emissions in primiparous lactating dairy cows. Journal of Animal Science and Biotechnology, 10, 41. https://doi.org/10.1186/s40104-019-0342-9
Jeyanathan J, Martin C, Morgavi DP. 2014. The use of direct-fed microbials for mitigation of ruminant methane emissions: A review. Animal, 8, 250–261. https://doi.org/10.1017/S1751731113002085
Jia J, Dawson TP, Wu F, Han Q, Cui X. 2023. Global meat demand projection: Quo vadimus? Journal of Cleaner Production, 429, 139460. https://doi.org/10.1016/j.jclepro.2023.139460
Jiang Y, Ogunade IM, Kim DH, Li X, Pech-Cervantes AA, Arriola KG, Oliveira AS, Driver JP, Ferraretto LF, Staples CR, Vyas D, Adesogan AT. 2018. Effect of adding clay with or without a Saccharomyces cerevisiae fermentation product on the health and performance of lactating dairy cows challenged with dietary aflatoxin. Journal of Dairy Science, 101, 3008–3020. https://doi.org/10.3168/jds.2017-13678
Jin Y, Lu L, Tu W, Luo T, Fu Z. 2019. Impacts of polystyrene microplastic on the gut barrier, microbiota and metabolism of mice. Science of The Total Environment, 649, 308–317. https://doi.org/10.1016/j.scitotenv.2018.08.353
Johnson KA, Johnson DE. 1995. Methane emissions from cattle. Journal of Animal Science, 73, 2483–2492. https://doi.org/10.2527/1995.7382483x
Karakus F. 2014. Weaning stress in lambs. Agriculture & Food, 2, 165–170. https://www.scientific-publications.net/en/issue/1000000/
Kareem KY, Loh TC, Foo HL, Akit H, Samsudin AA. 2016. Effects of dietary postbiotic and inulin on growth performance, IGF1 and GHR mRNA expression, caecal microbiota and volatile fatty acids in broilers. BMC Veterinary Research, 12, 163. https://doi.org/10.1186/s12917-016-0790-9
Kareem KY, Loh TC, Foo HL, Asmara SA, Akit H. 2017. Influence of postbiotic RG14 and inulin combination on cecal microbiota, organic acid concentration, and cytokine expression in broiler chickens. Poultry Science, 96, 966–975. https://doi.org/10.3382/ps/pew362
Kareem YK, Loh TC, Foo HL, Asmara SA, Akit H, Abdulla NR, FoongOoi M. 2015. Carcass, meat and bone quality of broiler chickens fed with postbiotic and prebiotic combinations. International Journal of Probiotics and Prebiotics, 10, 23–30. https://www.researchgate.net/publication/275893440_Carcass_Meat_and_Bone_Quality_of_Broiler_Chickens_Fed_with_Postbiotic_and_Prebiotic_Combinations
Kaufman JD, Seidler Y, Bailey HR, Whitacre L, Bargo F, Lüersen K, Rimbach G, Pighetti GM, Ipharraguerre IR, Ríus AG. 2021. A postbiotic from Aspergillus oryzae attenuates the impact of heat stress in ectothermic and endothermic organisms. Scientific Reports, 11, 6407. https://doi.org/10.1038/s41598-021-85707-3
Kedzierski M, Lechat B, Sire O, Le Maguer G, Le Tilly V, Bruzaud S. 2020. Microplastic contamination of packaged meat: Occurrence and associated risks. Food Packaging and Shelf Life, 24, 100489. https://doi.org/10.1016/j.fpsl.2020.100489
Kim SW, Brandherm M, Freeland M, Newton B, Cook D, Yoon I. 2008. Effects of yeast culture supplementation to gestation and lactation diets on growth of nursing piglets. Asian-Australasian Journal of Animal Science, 21, 1011–1014. https://doi.org/10.5713/ajas.2008.70438
Kowalczyk A, Czerniawska-Piątkowska E, Wrzecińska M. 2021. The importance of interferon-tau in the diagnosis of pregnancy. Biomedical Research International, 2021, 1–6. https://doi.org/10.1155/2021/9915814
Króliczewska B, Pecka-Kiełb E, Bujok J. 2023. Strategies used to reduce methane emissions from ruminants: Controversies and issues. Agriculture, 13, 602. https://doi.org/10.3390/agriculture13030602
Kullisaar T, Zilmer M, Mikelsaar M, Vihalemm T, Annuk H, Kairane C, Kilk A. 2002. Two antioxidative lactobacilli strains as promising probiotics. International Journal of Food Microbiology, 72, 215–224. https://doi.org/10.1016/S0168-1605(01)00674-2
Kumar N, Manimaran A, Kumaresan A, Jeyakumar S, Sreela L, Mooventhan P, Sivaram M. 2017. Mastitis effects on reproductive performance in dairy cattle: A review. Tropical Animal Health and Production, 49, 663–673. https://doi.org/10.1007/s11250-017-1253-4
Kumaresan A, Srivastava AK. 2022. Current concepts in bovine reproduction. Springer Nature, Singapore. https://doi.org/10.1007/978-981-19-0116-4
Lackner M, Branka M. 2024. Microplastics in farmed animals: A review. Microplastics, 3, 559–588. https://doi.org/10.3390/microplastics3040035
Lambo MT, Chang X, Liu D. 2021. The recent trend in the use of multi-strain probiotics in livestock production: An overview. Animals, 11, 2805. https://doi.org/10.3390/ani11102805
Lane J, Jubb T, Shephard R, Webb-Ware J, Fordyce G. 2015. Priority list of endemic diseases for the red meat industries. Project Report to Meat & Livestock Australia Limited, Sydney, Australia. https://era.dpi.qld.gov.au/id/eprint/5030/
Lascano CE, Carulla JE, Vargas JDJ. 2012. Strategies for reducing methane emissions from ruminants (Estratégias para a redução da emissão de metano por ruminantes). Revista Brasileira de Geografia Física, 4, 1315–1335. https://doi.org/10.26848/rbgf.v4i6.232794
Leduc A, Souchet S, Gelé M, Le Provost F, Boutinaud M. 2021. Effect of feed restriction on dairy cow milk production: A review. Journal of Animal Science, 99 (7), 1-12. https://doi.org/10.1093/jas/skab130
Lee J, Kim S, Kang CH. 2022. Immunostimulatory activity of lactic acid bacteria cell-free supernatants through the activation of NF-κB and MAPK signalling pathways in RAW 264.7 cells. Microorganisms, 10, 2247. https://doi.org/10.3390/microorganisms10112247
Lee J, Lee JS, Jang YC, Hong SY, Shim WJ, Song YK, Hong SH, Jang M, Han GM, Kang D, Hong S. 2015. Distribution and size relationships of plastic marine debris on beaches in South Korea. Archives of Environmental Contamination and Toxicology, 69, 288–298. https://doi.org/10.1007/s00244-015-0208-x
Lin X, Xia Y, Wang G, Yang Y, Xiong Z, Liv F, Zhou W, Ai L. 2018. Lactic acid bacteria with antioxidant activities alleviating oxidized oil induced hepatic injury in mice. Frontiers in Microbiology, 9. https://doi.org/10.3389/fmicb.2018.02684
Liu C, Ma N, Feng Y, Zhou M, Li H, Zhang X, Ma X. 2023. From probiotics to postbiotics: Concepts and applications. Animal Research and One Health 1, 92–114. https://doi.org/10.1002/aro2.7
Loh T, Choe D, Foo H, Sazili A, Bejo M. 2014. Effects of feeding different postbiotic metabolite combinations produced by Lactobacillus plantarum strains on egg quality and production performance, faecal parameters and plasma cholesterol in laying hens. BMC Veterinary Research, 10, 149. https://doi.org/10.1186/1746-6148-10-149
Loh TC, Thanh NT, Foo HL, Hair‐Bejo, M, Azhar BK. 2010. Feeding of different levels of metabolite combinations produced by Lactobacillus plantarum on growth performance, fecal microflora, volatile fatty acids and villi height in broilers. Animal Science Journal, 81, 205–214. https://doi.org/10.1111/j.1740-0929.2009.00701.x
Loh TC, Thu TV, Foo HL, Bejo MH. 2013. Effects of different levels of metabolite combination produced by Lactobacillus plantarum on growth performance, diarrhoea, gut environment and digestibility of postweaning piglets. Journal of Applied Animal Research, 41, 200–207. https://doi.org/10.1080/09712119.2012.741046
Lombardi P, Musco N, Cutrignelli M, Mollica M, Trinchese G, Calabrò S, Tudisco R, Grossi M, Mastellone V, Vassalotti G, Pero ME, Morittu VM, Infascelli F. 2017. The association of aloe and β-carotene supplementation improves oxidative stress and inflammatory state in pregnant buffalo cows. Buffalo Bulletin, 36, 497–503. http://kuojs.lib.ku.ac.th/index.php/BufBu/article/view/646/9
Madureira G, Mion B, van Winters B, Peñagaricano F, Li J, Ribeiro ES. 2024. Endometrial responsiveness to interferon-tau and its association with subsequent reproductive performance in dairy heifers. Journal of Dairy Science, 107, 7371–7391. https://doi.org/10.3168/jds.2023-24627
Malau-Aduli AEO, Siebert BD, Bottema CD, Pitchford WS. 1998. Breed comparison of the fatty acid composition of muscle phospholipids in Jersey and Limousin cattle. Journal of Animal Science, 76, 766. https://doi.org/10.2527/1998.763766x
Malau-Aduli AEO, Siebert BD, Bottema CDK, Pitchford WS. 1997. A comparison of the fatty acid composition of triacylglycerols in adipose tissue from Limousin and Jersey cattle. Australian Journal of Agricultural Research, 48, 715. https://doi.org/10.1071/A96083
Maltz E, Devir S, Metz JHM, Hogeveen H. 1997. The body weight of the dairy cow. Introductory study into body weight changes in dairy cows as a management aid. Livestock Production Science, 48, 175–186. https://doi.org/10.1016/S0301-6226(97)00024-9
Malvisi M, Stuknytė M, Magro G, Minozzi G, Giardini A, De Noni I, Piccinini R. 2016. Antibacterial activity and immunomodulatory effects on a bovine mammary epithelial cell line exerted by nisin A-producing Lactococcus lactis strains. Journal of Dairy Science, 99, 2288–2296. https://doi.org/10.3168/jds.2015-10161
Martens H. 2023. Invited Review – Increasing milk yield and negative energy balance: A Gordian Knot for dairy cows? Animals, 13, 3097. https://doi.org/10.3390/ani13193097
Martyniak A, Medyńska-Przęczek A, Wędrychowicz A, Skoczeń S, Tomasik PJ. 2021. Prebiotic, probiotic, synbiotic, paraprobiotic and postbiotic compounds in IBD. Biomolecules, 11, 1903. https://doi.org/10.3390/biom11121903
Mohammed MY, Kareem KY. 2022. A comparison study of probiotic, postbiotic and prebiotic on performance and meat quality of broilers. Tikrit Journal for Agricultural Sciences, 22, 24–32. https://doi.org/10.25130/tjas.22.4.4
Moradi M, Mardani K, Tajik H. 2019. Characterization and application of postbiotics of Lactobacillus spp. on Listeria monocytogenes in vitro and in food models. LWT – Food Science and Technology, 111, 457–464. https://doi.org/10.1016/j.lwt.2019.05.072
Moreira F, Orlandi C, Risco CA, Mattos R, Lopes F, Thatcher WW. 2001. Effects of presynchronization and bovine somatotropin on pregnancy rates to a timed artificial insemination protocol in lactating dairy cows. Journal of Dairy Science, 84, 1646–1659. https://doi.org/10.3168/jds.S0022-0302(01)74600-0
Muñoz C, Wills DA, Yan T. 2017. Effects of dietary active dried yeast (Saccharomyces cerevisiae) supply at two levels of concentrate on energy and nitrogen utilisation and methane emissions of lactating dairy cows. Animal Production Science, 57, 656. https://doi.org/10.1071/AN15356
Mwangi FW, Charmley E, Gardiner CP, Malau-Aduli BS, Kinobe RT, Malau-Aduli AEO. 2019. Diet and genetics influence beef cattle performance and meat quality characteristics. Foods, 8, 648. https://doi.org/10.3390/foods8120648
Nakamura S, Kuda T, An C, Kanno T, Takahashi H, Kimura B. 2012. Inhibitory effects of Leuconostoc mesenteroides 1RM3 isolated from narezushi, a fermented fish with rice on Listeria monocytogenes infection to Caco-2 cells and A/J mice. Anaerobe, 18, 19–24. https://doi.org/10.1016/j.anaerobe.2011.11.006
Nguyen Tien Thanh, Chwen, L.T., Ling, F.H., Bejo, H.M., Azhar, K., 2010. Inhibitory activity of metabolites produced by strains of Lactobacillus plantarum isolated from Malaysian fermented food. International Journal of Probiotics and Prebiotics, 5, 37–44.
Oguey C, Thayer MT. 2024. Sow reproductive and progeny growth performance when fed Pichia guilliermondii yeast postbiotic: systematic review and meta-analysis. Translational Animal Science, 8, 137 https://doi.org/10.1093/tas/txae137
Olagaray KE, Sivinski SE, Saylor BA, Mamedova LK, Sauls-Hiesterman JA, Yoon I, Bradford BJ. 2019. Effect of Saccharomyces cerevisiae fermentation product on feed intake parameters, lactation performance, and metabolism of transition dairy cattle. Journal of Dairy Science, 102, 8092–8107. https://doi.org/10.3168/jds.2019-16315
Otto JR, Freeman MJ, Malau-Aduli BS, Nichols PD, Lane PA, Malau-Aduli AEO. 2014. Reproduction and fertility parameters of dairy cows supplemented with omega-3 fatty acid-rich canola oil. Annual Research and Reviews in Biology, 4, 1611–1636. https://doi.org/10.9734/ARRB/2014/7689
Pellegrino MS, Frola ID, Natanael B, Gobelli D, Nader-Macias MEF, Bogni CI. 2019. In vitro characterization of lactic acid bacteria isolated from bovine milk as potential probiotic strains to prevent bovine mastitis. Probiotics and Antimicrobial Proteins, 11, 74–84. https://doi.org/10.1007/s12602-017-9383-6
Phesatcha K, Phesatcha B, Chunwijitra K, Wanapat M, Cherdthong A. 2021. Changed rumen fermentation, blood parameters, and microbial population in fattening steers receiving a high concentrate diet with Saccharomyces cerevisiae improve growth performance. Veterinary Science, 8, 294. https://doi.org/10.3390/vetsci8120294
Plaizier JC, Khafipour E, Li S, Gozho GN, Krause DO. 2012. Subacute ruminal acidosis (SARA), endotoxins and health consequences. Animal Feed Science and Technology, 172, 9–21. https://doi.org/10.1016/j.anifeedsci.2011.12.004
Ponnampalam EN, Bekhit AED, Bruce H, Scollan ND, Muchenje V, Silva P, Jacobs JL. 2019. Production strategies and processing systems of meat. In: Sustainable Meat Production and Processing. Elsevier, pp. 17–44. https://doi.org/10.1016/B978-0-12-814874-7.00002-X
Poppy GD, Rabiee AR, Lean IJ, Sanchez WK, Dorton KL, Morley PS. 2012. A meta-analysis of the effects of feeding yeast culture produced by anaerobic fermentation of Saccharomyces cerevisiae on milk production of lactating dairy cows. Journal of Dairy Science, 95, 6027–6041. https://doi.org/10.3168/jds.2012-5577
Price KL, Totty HR, Lee HB, Utt MD, Fitzner GE, Yoon I, Ponder MA, Escobar J. 2010. Use of Saccharomyces cerevisiae fermentation product on growth performance and microbiota of weaned pigs during Salmonella infection. Journal of Animal Science, 88, 3896–3908. https://doi.org/10.2527/jas.2009-2728
Qadis AQ, Goya S, Ikuta K, Yatsu M, Kimura A, Nakanishi S, Sato S. 2014. Effects of a bacteria-based probiotic on ruminal pH, volatile fatty acids and bacterial flora of Holstein calves. Journal of Veterinary Medical Science, 76, 877–885. https://doi.org/10.1292/jvms.14-0028
Qiao J, Chen R, Wang M, Bai R, Cui X, Liu Y, Wu C, Chen C. 2021. Perturbation of gut microbiota plays an important role in micro/nanoplastics-induced gut barrier dysfunction. Nanoscale, 13, 8806–8816. https://doi.org/10.1039/D1NR00038A
Rabelo CHS, Basso FC, Lara EC, Jorge LGO, Härter CJ, Mari LJ, Reis RA. 2017. Effects of Lactobacillus buchneri as a silage inoculant or probiotic on in vitro organic matter digestibility, gas production and volatile fatty acids of low dry‐matter whole‐crop maize silage. Grass and Forage Science, 72, 534–544. https://doi.org/10.1111/gfs.12273
Retallick KM, Faulkner DB, Rodriguez-Zas SL, Nkrumah JD, Shike DW. 2013. Relationship among performance, carcass, and feed efficiency characteristics, and their ability to predict economic value in the feedlot. Journal of Animal Science, 91, 5954–5961. https://doi.org/10.2527/jas.2013-6156
Ríus AG, Kaufman JD, Li MM, Hanigan MD, Ipharraguerre IR. 2022. Physiological responses of Holstein calves to heat stress and dietary supplementation with a postbiotic from Aspergillus oryzae. Scientific Reports, 12, 1587. https://doi.org/10.1038/s41598-022-05505-3
Rojas-Downing MM, Nejadhashemi AP, Harrigan T, Woznicki SA. 2017. Climate change and livestock: Impacts, adaptation, and mitigation. Climate Risk Management, 16, 145–163. https://doi.org/10.1016/j.crm.2017.02.001
Sanchez NCB, Young TR, Carroll JA, Corley JR, Rathmann RJ, Johnson BJ. 2014. Yeast cell wall supplementation alters the metabolic responses of crossbred heifers to an endotoxin challenge. Innate Immunity, 20, 104–112. https://doi.org/10.1177/1753425913482152
Şanlı ER. 2024. Usability of postbiotics in ruminant nutrition and health. Turkish Journal of Agriculture – Food Science and Technology, 12, 1104–1109. https://doi.org/10.24925/turjaf.v12i6.1104-1109.6710
Sasaki Y, Uematsu M, Kitahara G, Osawa T. 2016. Reproductive performance of Japanese Black cattle: Association with herd size, season, and parity in commercial cow-calf operations. Theriogenology, 86, 2156–2161. https://doi.org/10.1016/j.theriogenology.2016.07.007
Scott E, De Paepe K, van de Wiele T. 2022. Postbiotics and their health modulatory biomolecules. Biomolecules, 12, 1640. https://doi.org/10.3390/biom12111640
Sevin S, Karaca B, Haliscelik O, Kibar H, OmerOglou E, Kiran F. 2021. Postbiotics secreted by Lactobacillus sakei EIR/CM-1 isolated from cow milk microbiota, display antibacterial and antibiofilm activity against ruminant mastitis-causing pathogens. Italian Journal of Animal Science, 20, 1302–1316. https://doi.org/10.1080/1828051X.2021.1958077
Shabunin SV, Nezhdanov AG, Mikhalev VI, Pask NV, Prokulevich VA, Potapovich MI, Gricuk VA, Volkova IV. 2019. Interferon-tau and formation of pregnancy in cows. Sel’skokhozyaistvennaya Biologiya, 54, 246–258. https://doi.org/10.15389/agrobiology.2019.2.259eng
Sharma RK, Chauhan K, Pathak AK, Khan N. 2020. Postbiotics for animal health and performance. Intas Polivet, 21, 288–294.
Smith SB, Crouse JD. 1984. Relative contributions of acetate, lactate and glucose to lipogenesis in bovine intramuscular and subcutaneous adipose tissue. Journal of Nutrition, 114, 792–800. https://doi.org/10.1093/jn/114.4.792
Spaggiari L, Sala A, Ardizzoni A, De Seta F, Singh DK, Gacser A, Blasi E, Pericolini E. 2022. Lactobacillus acidophilus, L. plantarum, L. rhamnosus, and L. reuteri cell-free supernatants inhibit Candida parapsilosis pathogenic potential upon infection of vaginal epithelial cells monolayer and in a trans-well co-culture system in vitro. Microbiology Spectrum, 10. https://doi.org/10.1128/spectrum.02696-21
Štofilová J, Kvaková M, Kamlárová A, Hijová E, Bertková I, Guľašová Z. 2022. Probiotic-based intervention in the treatment of ulcerative colitis: Conventional and new approaches. Biomedicines, 10, 2236. https://doi.org/10.3390/biomedicines10092236
Štolcová M, Bartoň L, Řehák D. 2024. Milk components as potential indicators of energy status in early lactation Holstein dairy cows from two farms. Animal, 18, 101235. https://doi.org/10.1016/j.animal.2024.101235
Suybeng B, Charmley E, Gardiner CP, Malau-Aduli BS, Malau-Aduli AEO. 2019. Methane emissions and the use of Desmanthus in beef cattle production in Northern Australia. Animals, 9, 542. https://doi.org/10.3390/ani9080542
Swyers KL, Wagner JJ, Dorton KL, Archibeque SL. 2014. Evaluation of Saccharomyces cerevisiae fermentation product as an alternative to monensin on growth performance, cost of gain, and carcass characteristics of heavy-weight yearling beef steers. Journal of Animal Science, 92, 2538–2545. https://doi.org/10.2527/jas.2013-7559
Tartrakoon W, Charoensook R, Incharoen T, Numthuam S, Pechrkong T, Onoda S, Shoji G, Brenig B. 2023. Effects of heat-killed Lactobacillus plantarum L-137 Supplementation on growth performance, blood profiles, intestinal morphology, and immune gene expression in pigs. Veterinary Science, 10, 87. https://doi.org/10.3390/vetsci10020087
Thanh NT, Chwen LT, Foo HL, Hair-Bejo M, Kasim AB. 2010. Inhibitory activity of metabolites produced by strains of Lactobacillus plantarum isolated from Malaysian fermented food. International Journal of Probiotics and Prebiotics, 5, 37–44. http://psasir.upm.edu.my/id/eprint/15232
Thatcher, W., Guzeloglu, A., Meikle, A., Kamimura, S., Bilby, T., Kowalski, A., Badinga, L., Pershing, R., Bartolome, J., Santos, J., 2019. Regulation of embryo survival in cattle. In: Reproduction in Domestic Ruminants Volume 5, Bioscientifica Proceedings Reproduction Supplement 61, 253-266. https://doi.org/10.1530/biosciprocs.5.019
Thibessard A, Borges F, Fernandez A, Gintz B, Decaris B, Leblond-Bourget N. 2004. Identification of Streptococcus thermophilus CNRZ368 genes involved in defense against superoxide stress. Applied and Environmental Microbiology, 70, 2220–2229. https://doi.org/10.1128/AEM.70.4.2220-2229.2004
Thomas M, Serrenho RC, Puga SO, Torres JM, Puga Sair Ordaz, Stangaferro M. 2023. Effect of feeding a Saccharomyces cerevisiae fermentation product to Holstein cows exposed to high temperature and humidity conditions on milk production performance and efficiency – A pen-level trial. Journal of Dairy Science, 106, 4650–4665. https://doi.org/10.3168/jds.2022-22516
Thorakkattu P, Khanashyam AC, Shah K, Babu KS, Mundanat AS, Deliephan A, Deokar GS, Santivarangkna C, Nirmal NP. 2022. Postbiotics: Current trends in food and pharmaceutical industry. Foods, 11, 3094. https://doi.org/10.3390/foods11193094
Thorsteinsson M, Vestergaard M. 2020. Performance and health of young rosé veal calves supplemented with yeast (Saccharomyces cerevisiae) and a postbiotic from Lactobacillus acidophilus. Journal of Animal and Feed Science, 29, 115–124. https://doi.org/10.22358/jafs/124040/2020
Thu TV, Loh TC, Foo HL, Yaakub H, Bejo MH. 2011. Effects of liquid metabolite combinations produced by Lactobacillus plantarum on growth performance, faeces characteristics, intestinal morphology and diarrhoea incidence in postweaning piglets. Tropical Animal Health and Production, 43, 69–75. https://doi.org/10.1007/s11250-010-9655-6
Tripathi MK, Karim SA. 2011. Effect of yeast cultures supplementation on live weight change, rumen fermentation, ciliate protozoa population, microbial hydrolytic enzymes status and slaughtering performance of growing lamb. Livestock Science, 135, 17–25. https://doi.org/10.1016/j.livsci.2010.06.007
Trzeciak P, Herbet M. 2021. Role of the intestinal microbiome, intestinal barrier and psychobiotics in depression. Nutrients, 13, 927. https://doi.org/10.3390/nu13030927
Tsai TC, Kim HJ, Wang X, Bass BE, Frank JW, Maxwell CV. 2016. Effect of Saccharomyces cerevisiae fermentation product supplementation in late gestation and lactation on sow and litter performance, milk components, and fecal Clostridium perfringens. Journal of Animal Science, 94, 134–134. https://doi.org/10.2527/msasas2016-286
Tsai WH, Chou CH, Chiang YJ, Lin CG, Lee CH. 2021. Regulatory effects of Lactobacillus plantarum-GMNL6 on human skin health by improving skin microbiome. International Journal of Medical Sciences, 18, 1114–1120. https://doi.org/10.7150/ijms.51545
Tufarelli V, Puvača N, Glamočić D, Pugliese G, Colonna MA. 2024. The most important metabolic diseases in dairy cattle during the transition period. Animals, 14, 816. https://doi.org/10.3390/ani14050816
Urli S, Corte PF, Crociati M, Baufeld A, Monaci M, Stradaioli G. 2023. Impact of microplastics and nanoplastics on livestock health: An emerging risk for reproductive efficiency. Animals, 13, 1132. https://doi.org/10.3390/ani13071132
Vicente F, Campo-Celada M, Menéndez-Miranda M, García-Rodríguez J, Martínez-Fernández A. 2024. Effect of postbiotic supplementation on nutrient digestibility and milk yield during the transition period in dairy cows. Animals, 14(16), 2359; https://doi.org/10.3390/ani14162359
Vinderola G, Sanders ME, Salminen S. 2022. The concept of postbiotics. Foods, 11, 1077. https://doi.org/10.3390/foods11081077
Wagner JJ, Engle TE, Belknap CR, Dorton KL. 2016. Meta-analysis examining the effects of Saccharomyces cerevisiae fermentation products on feedlot performance and carcass traits. The Professional Animal Scientist, 32, 172–182. https://doi.org/10.15232/pas.2015-01438
Walczak AP, Kramer E, Hendriksen PJM, Tromp P, Helsper JPFG, van der Zande M, Rietjens IMCM, Bouwmeester H. 2015. Translocation of differently sized and charged polystyrene nanoparticles in in vitro intestinal cell models of increasing complexity. Nanotoxicology, 9, 453–461. https://doi.org/10.3109/17435390.2014.944599
Wang J, Tan Z, Peng J, Qiu Q, Li M. 2016. The behaviours of microplastics in the marine environment. Marine Environmental Research 113, 7–17. https://doi.org/10.1016/j.marenvres.2015.10.014
Wang Y, Zhao Y, Tang X, Nan X, Jiang L, Wang H, Liu J, Yang L, Yao J, Xiong B. 2024. Nutrition, gastrointestinal microorganisms and metabolites in mastitis occurrence and control. Animal Nutrition, 17, 220–231. https://doi.org/10.1016/j.aninu.2024.01.010
Wei Y, Wang J, Liu Z, Pei J, Brennan C, Abdel-Aty AM. 2022. Isolation and characterization of bacteriocin-producing Lacticaseibacillus rhamnosus XN2 from Yak yoghurt and its bacteriocin. Molecules, 27, 2066. https://doi.org/10.3390/molecules27072066
Wenz JR, Barrington GM, Garry FB, McSweeney KD, Dinsmore RP, Goodell G, Callan RJ. 2001. Bacteremia associated with naturally occurring acute coliform mastitis in dairy cows. Journal of the American Veterinary Medical Association, 219, 976–981. https://doi.org/10.2460/javma.2001.219.976
Wilson DJ, Grohn YT, Bennett GJ, González RN, Schukken YH, Spatz J. 2008. Milk production change following clinical mastitis and reproductive performance compared among J5-vaccinated and control dairy cattle. Journal of Dairy Science, 91, 3869–3879. https://doi.org/10.3168/jds.2008-1405
Wu J, Zhao Y, Wang X, Kong L, Johnston LJ, Lu L, Ma X. 2022. Dietary nutrients shape gut microbes and intestinal mucosa via epigenetic modifications. Critical Reviews in Food Science and Nutrition, 62, 783–797. https://doi.org/10.1080/10408398.2020.1828813
Xie S, Zhou A, Wei T, Li S, Yang B, Xu G, Zou J. 2021. Nanoplastics induce more serious microbiota dysbiosis and inflammation in the gut of adult Zebrafish than microplastics. Bulletin of Environmental Contamination and Toxicology, 107, 640–650. https://doi.org/10.1007/s00128-021-03348-8
Xu S, Jia X, Liu Y, Pan X, Chang J, Wei W, Lu P, Petry D, Che L, Jiang X, Wang J, Wu D. 2023. Effects of yeast-derived postbiotic supplementation in late gestation and lactation diets on performance, milk quality, and immune function in lactating sows. Journal of Animal Science, 101. https://doi.org/10.1093/jas/skad201
Yee MSL, Hii LW, Looi CK, Lim WM, Wong SF, Kok YY, Tan BK, Wong CY, Leong CO. 2021. Impact of microplastics and nanoplastics on human health. Nanomaterials, 11, 496. https://doi.org/10.3390/nano11020496
Yin K, Wang Y, Zhao H, Wang D, Guo M, Mu M, Liu Y, Nie X, Li B, Li J, Xing M. 2021. A comparative review of microplastics and nanoplastics: Toxicity hazards on digestive, reproductive and nervous system. Science of The Total Environment, 774, 145758. https://doi.org/10.1016/j.scitotenv.2021.145758
Yin L, Chen B, Xia B, Shi X, Qu K. 2018. Polystyrene microplastics alter the behavior, energy reserve and nutritional composition of marine jacopever (Sebastes schlegelii). Journal of Hazardous Materials, 360, 97–105. https://doi.org/10.1016/j.jhazmat.2018.07.110
Yitbarek BM. 2019. Livestock and livestock product trends by 2050: Review. International Journal of Animal Research, 4, 1–20. https://escipub.com/international-journal-of-animal-research/
Yun B, Oh S, Griffiths MW. 2014. Lactobacillus acidophilus modulates the virulence of Clostridium difficile. Journal of Dairy Science, 97, 4745–4758. https://doi.org/10.3168/jds.2014-7921
Zaleska B, Milewski S, Ząbek K. 2015. Impact of Saccharomyces cerevisiae supplementation on reproductive performance, milk yield in ewes and offspring growth. Archives Animal Breeding 58, 79–83. https://doi.org/10.5194/aab-58-79-2015
Zomeño C, Gispert M, Čandek-Potokar M, Mörlein D, Font-i-Furnols M. 2023. A matter of body weight and sex type: Pig carcass chemical composition and pork quality. Meat Science, 197, 109077. https://doi.org/10.1016/j.meatsci.2022.109077
Disclaimer/Publisher Note: The statements, opinions, institutional affiliations, data contained in all publications, and all responsibilities for accuracy are solely those of the individual author(s) and contributor(s) and not of MARCIAS AUSTRALIA and AJAVAS/or the Editor(s). MARCIAS AUSTRALIA and AJAVAS/or the Editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.

