Fermented diets influence broiler intestinal microbiota
Pride Hodzi1,2* Tonderai Mutibvu2 Godfrey Bernard Nyamushamba3 Soul Washaya1
- Gary Magadzire School of Agriculture, Great Zimbabwe University, P.O. Box 1235, Masvingo, Zimbabwe
- Department of Livestock Sciences, University of Zimbabwe, P.O. Box 167 Mount Pleasant, Harare, Zimbabwe
- Faculty of Agriculture and Environmental Science, Bindura University of Science Education, P. Bag 1020, Bindura, Zimbabwe
Article Information
- Date Received: 16/03/2026
- Date Revised: 0/04/2026
- Date Accepted: 12/04/2026
- Date Published Online: 27/04/2026
Copyright: © 2026 The Authors. Published by MARCIAS AUSTRALIA, 32 Champion Drive, Rosslea, Queensland 4812, Australia. This is an open access publication under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/). Citation: Hodzi P, Mutibvu T, Nyamushamba GB, Washaya S (2026). Fermented diets influence broiler intestinal microbiota. Aust J Agric Vet Anim Sci (AJAVAS), 2(2), 100018 https://doi.org/10.64902/ajavas.2026.100018
Abstract
The intestinal microbiota plays a critical role in broiler health, nutrient metabolism, immune function, and overall productivity. Its composition is influenced by multiple factors, with diet being the primary driver alongside genetics, digesta passage rate, and rearing environment. Amid growing restrictions on antimicrobial growth promoters and increasing emphasis on sustainable production, fermented diets have emerged as a viable nutritional strategy in broiler nutrition. Furthermore, fermentation serves as a valuable tool for valorizing agricultural by-products and developing alternative broiler feed ingredients. Research has shown that fermented feed enhances growth performance, improves gut health, and beneficially modulates the intestinal microbiota, and hence microbiome. This review synthesizes current research on how fermented feeds, including fermented cereals, brans, legumes, pulses, pomaces, and complete rations, influence the composition, diversity, and functional dynamics of the broiler intestinal microbiota. We discuss the chicken gastrointestinal tract, microbiota, fermentation of broiler feed, the mechanisms through which fermented diets interact with the gastrointestinal environment, emphasizing their role in improving gut barrier integrity, immune responses, and nutrient absorption. By integrating recent findings, this paper highlights the potential of fermented feeds as a viable, microbiome-targeted intervention to support sustainable broiler production while addressing contemporary challenges in poultry health and nutrition.
Keywords
Broiler chicken, fermented feed, gut health, intestinal microbiome, antimicrobial growth promoter alternatives
Highlights
- Broiler intestinal microbiome is significantly influenced by diet.
- Fermented feed ingredients used in broiler nutrition supply beneficial bioactive compounds, including short chain fatty acids (SCFAs), probiotics, and bacteriocins.
- Fermented feeds improve the broiler intestinal microbiome by enhancing beneficial microbial populations and suppressing pathogens.
1.0 Introduction
Optimising gut health is paramount for sustainable and efficient broiler production (Kogut, 2019; Pan and Yu, 2014). The gut microbiota plays a critical role in broiler health (Sun et al., 2022a), nutrient metabolism (Aruwa and Sabiu, 2024), immune function (Wickramasuriya et al., 2022), and overall productivity (Fathima et al., 2022; Gao et al., 2024). As diet is the primary environmental driver shaping this microbial community, specific feed components exert profound selective pressures that determine its structure and function, thereby influencing host health (Pan and Yu, 2014). Consequently, dietary strategies to modulate the microbiota are a central focus of modern nutritional research (Kogut, 2019; Predescu et al., 2024).
Common feed ingredients used in broiler nutrition, including whole cereal grains, oilseeds, and their by-products, contain antinutritional factors (ANFs), such as tannins, saponins, phytates, gossypol, trypsin inhibitors, and non-starch polysaccharides, which impair nutrient digestibility and absorption in the avian GIT (Abbas., 2020; Samtiya et al., 2020; Nalluri and Karri, 2021). Furthermore, these compounds can induce gut inflammation and compromise intestinal integrity, exacerbating the constant challenge posed by enteric pathogens such as Salmonella and Clostridium perfringens in intensive production systems (Pan and Yu, 2014). Intensive poultry production faces the persistent challenge of enteric pathogens such as Salmonella and Clostridium perfringens, creating a critical need to move beyond traditional antibiotic growth promoters (Fancher et al., 2020). This has spurred a continuous search for safe and effective feed additives or processing methods that enhance gut health and bird performance (Salim et al., 2018; Fancher et al., 2020; Muneeb et al., 2024). Amid growing restrictions on AGPs and an increasing emphasis on sustainable production, fermented diets have emerged as a particularly promising nutritional strategy in modern broiler nutrition (Rahman et al., 2022; Pewan et al., 2025).
Feed fermentation, a traditional bioprocessing, is being re-evaluated with modern science to enable targeted modifications that yield desirable products such as organic acids, bacteriocins, enzymes, or prebiotic compounds (Nan et al., 2022; Mao et al., 2023; Dastar et al., 2025; Hodzi et al., 2025; Katu et al., 2025). Fermentation enhances broiler feed quality by breaking down complex compounds such as non-starch polysaccharides (e.g., cellulose, xylan, β glucans), phytate, tannins, and other anti-nutritional factors, making nutrients more digestible and increasing their bioavailability for absorption by intestinal epithelial cells (Haryati Supriyati et al., 2015). A primary benefit is the significant reduction in the concentration of ANF (Sokrab et al., 2014; Predescu et al., 2024; Cardoso Dal Pont et al., 2020). Feed fermentation also elevates concentrations of beneficial components, including probiotics (Soren et al., 2024; Hodzi et al., 2025; Katu et al., 2025). Additionally, fermented feed acts as a direct source of health-promoting metabolites, including short-chain fatty acids (SCFAs), which regulate intestinal blood flow, stimulate enterocyte growth and proliferation, modulate mucin production, and influence local immune responses (Liu et al., 2023). The production of SCFAs lowers the luminal pH, creating an environment that selectively inhibits acid-sensitive pathogenic and spoilage microorganisms while favoring acid-tolerant commensals, particularly Lactobacillus species (Guo et al., 2021). Thus, fermented feed primarily influences the poultry gut microbiota by directly introducing beneficial microorganisms and metabolites, which suppress pathogens and improve the gut environment for health-promoting bacteria (Guo et al., 2021; Pan and Yu, 2014; Kang et al., 2025).
While promising, the effects of fermented feed on the broiler gut microbiome are complex and can vary. A comprehensive synthesis of current knowledge on how fermented diets specifically reshape the broiler intestinal microbiota is needed. This article explores the current scientific evidence on how fermented diets (fermented cereals, brans, seed meals, fruit pomaces, legumes, pulses or complete feeds) shape the composition, diversity, and functional capacity of the broiler intestinal microbiome. We analyze the interplay between fermented diets, the avian gastrointestinal tract, and the gut microbiota. By synthesizing recent research findings, this review aims to provide a clear understanding of the potential of fermented feeds as a microbiome-targeted nutritional strategy, offering insights for researchers, nutritionists, and producers seeking to advance sustainable poultry production.
Review methodology:
To compile this review, a thorough and structured literature search was conducted to identify and synthesise peer-reviewed studies examining the effects of fermented diets on broiler intestinal microbiota, gut morphology, and related parameters. Relevant scholarly publications were identified using electronic databases, including Google Scholar, Web of Science, Scopus, and PubMed. In addition to the scientific scholarly electronic databases, Google Search, Research Gate, and Research Square were also used to obtain published articles relevant to the review. The search was performed using combinations of keywords applied to titles, abstracts, and full texts where appropriate, with primary terms including “broiler”, “gut morphology”, “intestinal microbiota”, and “fermented feed”, and Boolean operators (AND, OR) were used to refine the search (e.g., “broiler”, “fermented feed” AND “intestinal microbiota”). The review considered peer reviewed original research articles and review papers published predominantly after 2014, with a few older, highly relevant exceptions included for foundational context, and only studies published in English were included. The focus was on broiler chickens, although a limited number of studies addressing poultry more broadly (e.g., layers, turkeys) were retained when they provided relevant mechanistic insights. A total of 1,000 articles were initially screened, comprising approximately 950 original research articles and 50 review papers, which were categorized according to the following thematic areas: intestinal morphology, intestinal microbiota, fermented feed ingredients in broiler nutrition, and effects of fermented feeds on the broiler intestinal microbiota. Articles that did not directly address the relationship between fermented diets and broiler intestinal health were excluded, resulting in a final selection of 120 articles. The remaining literature was critically evaluated, and relevant data were extracted, summarized, and synthesized narratively following the approach described by Brignardello-Petersen et al. (2025).
2.0 Broiler GIT morphology
The GIT of the broiler chicken is a specialised and highly efficient system (Dibner and Richards, 2004) . Morphologically, as shown in Figure 1, it consists of the oesophagus, crop, proventriculus, gizzard, small intestine (duodenum, jejunum, and ileum), caeca, and large intestine (Dibner and Richards, 2004; Maki et al, 2019). Notably shorter than the mammalian digestive system, it processes feed rapidly, with a rapid feed transit time of approximately 3.5 hours or less (Wilkinson et al., 2016). The ceca are the critical exception to this fast passage and serve as a fermentation chamber with a much slower turnover, making them the primary habitat for a dense and highly diverse microbial community (Maki et al, 2019; Katu et al., 2025). Other segments of the broiler GIT, including the esophagus, crop, proventriculus, and gizzard, small intestine and large intestine, also host distinct microbial communities (Figure 1). These microorganisms play essential roles in supporting the growth, health, immunity, and productive performance of broilers.
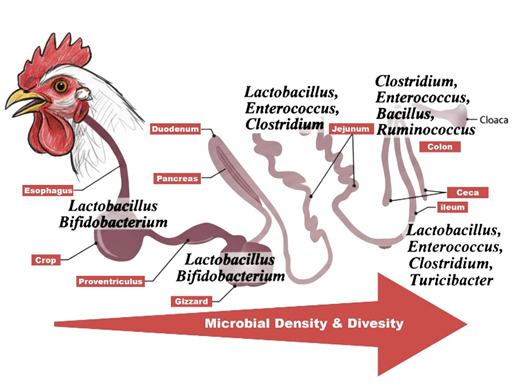
Figure 1. The broiler GIT and its predominant microbial residents (Made using Adobe Photoshop, Adobe Inc, 2019)
The gut mucosa lines the inner surface of the avian GIT, as shown in Figure 2.
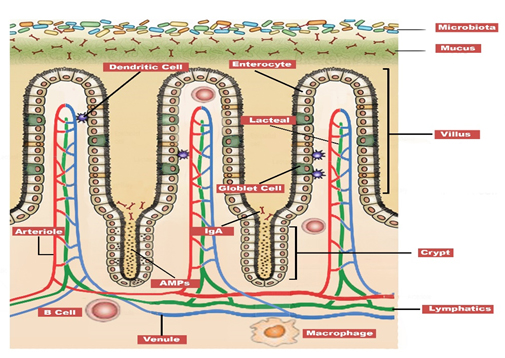
Figure 2. The broiler intestinal morphology showing microbial biofilm, mucus layer and intestinal integrity (Made using Adobe Photoshop, Adobe Inc, 2019)
This mucosa acts as both a physical and immunological barrier, primarily composed of a gel-like mucus layer secreted by goblet cells, a biofilm of microorganisms, their metabolites, and secretory immunoglobulin A (sIgA) (Pelaseyed et al., 2014; Alemao et al., 2021). The mucus itself is structured in two strata: an outer, loose layer that harbors the gut microbiota, and an inner, compact layer that is largely impervious to bacteria, serving as the first line of defense against infection (Pelaseyed et al., 2014; Jia et al., 2019).
In vitro studies have shown that chicken intestinal mucus reduces C. jejuni virulence through inhibiting its adhesion and invasion of epithelial cells (Byrne et al., 2007; Alemka et al., 2010). The mucus layer is anchored to a central, single-cell layer of specialized epithelial cells. Intestinal epithelial cells produce antimicrobial peptides like β-defensins, which disrupt the membranes of pathogens (Jia et al., 2019). The epithelial cells are bound together by tight junction proteins (ETJP), that regulate the passage of ions and molecules through the paracellular pathway (Awad et al., 2017). The epithelial layer is supported by the underlying lamina propria, a layer rich in immune cells, connective tissue, and blood vessels, (Vielmuth, 2021).
Cellular components of the innate immune system, such as macrophages and heterophils, reside in the lamina propria and are recruited to sites of infection to eliminate invaders via phagocytosis (Chen et al., 2022). Nutrients and other compounds cross this epithelial barrier via two principal routes. The transcellular pathway moves large, antigenic, or lipophilic substances through the epithelial cells via transporters, endocytosis, or diffusion (Hansen et al., 2024). Conversely, the paracellular pathway allows small ions and hydrophilic molecules (<600 Da) to pass between cells, a process tightly regulated by the tight junctions (Wu et al., 2023; Hansen et al., 2024). The small intestine is chiefly responsible for the digestion and absorption of nutrients (El Sabry and Yalcin, 2023). Digestion and nutrient absorption occur at the level of the intestinal villi and crypts. The morphological health of this surface is gauged by villus height (VH), crypt depth (CD), and their ratio (VH/CD), where longer villi and shallower crypts indicate a greater surface area for absorption and are directly linked to enhanced growth performance (Katu et al., 2025).
Ultimately, broiler intestinal health is a broad and dynamic concept reliant on the intricate interplay of three core components, which are nutrition, intestinal morphology, and the gut microbiota (Wickramasuriya et al., 2022; Ducatelle et al., 2023). These elements work in concert to maintain the functional and defensive balance of the GIT lumen, ensuring efficient nutrient movement from the lumen into systemic circulation and directly underpinning overall performance.
3.0 Microbiota and microbiome
The terms microbiota and microbiome are distinct, though often erroneously used as synonyms. The microbiota refers specifically to the community of living microorganisms, including bacteria, fungi, viruses, and protozoa, that inhabit a defined environment such as the intestine (Berg et al., 2020). In the context of poultry, the phrase “intestinal microbiota” typically denotes the bacterial population, which represents the dominant and functionally most significant component (Bailey, 2011; Maki et al., 2019; Wickramasuriya et al., 2022). Synonyms such as communal bacteria or gut microbiota are also appropriate.
In contrast, the microbiome is a broader, functional organ. It encompasses not only the microbiota (the organisms themselves) but also their collective genetic material (genes and genomes), their metabolic products, and their dynamic interactions with the host environment (Berg et al., 2020). Thus, analysing the microbiome is inherently more complex than cataloging the microbiota alone, as it involves understanding the system’s functional potential and activity.
4.0 Broiler intestinal microbiome
Advances in DNA sequencing, from initial Sanger methods to modern next-generation sequencing (NGS), have enabled a comprehensive characterization of the poultry intestinal microbiome (Pan and Yu, 2014). This ecosystem is remarkably biodiverse, hosting at least 500 bacterial phylotypes or an estimated one million bacterial genes, a genetic repertoire 40 to 50 times larger than the chicken genome itself (Kogut, 2019). Crucially, over 90-95% of these microbes, particularly within the cecum, are unculturable in the laboratory (Sergeant et al., 2014; Kogut, 2019) and are thus accessible only through molecular biological approaches like 16S rRNA gene sequencing (Deng et al., 2025).
While 13 bacterial phyla have been identified, the community is overwhelmingly dominated (>90%) by three: Firmicutes, Bacteroidetes, and Proteobacteria (Wei et al., 2013). Lactobacillus spp. is the most dominant genus in the ileum and jejunum, comprising over 99% of bacteria (Yeoman et al., 2012; Kogut et al., 2019). The distribution of gut microbiota varies by section as shown in Figure 1. The crop exhibits the greatest diversity of Lactobacillus, while the cecum is the most densely colonized region (1010 to 1012 CFU/g) and hosts the highest overall bacterial diversity (Abbas et al., 2007; Jian et al., 2025). Katu et al. (2025) reported that cecal microbiota is dominated by two major phyla, which are Gram-positive Firmicutes (including families such as Ruminococcaceae and Lachnospiraceae) and Gram-negative Bacteroidetes (including Rikenellaceae and Bacteroidaceae). Ultimately, the composition of this intricate microbiome is shaped by a combination of genetic, age-related, dietary, and environmental factors, including digesta passage rate and rearing conditions (Maki et al., 2019; Sun et al., 2015; da Silva et al., 2024).
Pathogenic gut microbiota detrimentally impact the avian host through direct competition for nutrients, such as energy and protein, in the small intestine and gizzard (Fathima et al., 2022). Additionally, intestinal pathogens compromise host physiology by generating toxic metabolites and degrading bile acids, leading to suppressed growth and reduced fat digestibility (Pan and Yu, 2014). Conversely, commensal bacteria help maintain gut function, modulate the immune system, digest nutrients, and support overall intestinal homeostasis (Pan and Yu, 2014; Maki et al., 2019; da Silva et al., 2024). A healthy gut microbiome is composed of high abundance of commensals such as Firmicutes (e.g., Lactobacillus spp) and Bifidobacteria, alongside low levels of pathogenic bacteria such as E. coli, Salmonella, Clostridium spp and Bacteroides (Suriano et al., 2022; Sun et al., 2022). This balanced microbial community stimulates the bird’s immune system and contributes to host nutrient metabolism. In the presence of a healthy microbiota, the intestinal lining responds by increasing both mucin production and epithelial cell turnover (da Silva et al., 2024). These mechanisms lubricate the gastrointestinal tract and prevent microbial invasion of the host epithelium (Pan and Yu, 2014).
5.0 Microbiota colonisation and succession
Microbial colonisation of the chick gastrointestinal tract begins immediately after hatch and progresses in a predictable, staged manner, culminating in a stable, complex community by approximately 15–22 days of age, as shown in Figure 3. Starting from a largely sterile state, the tract is rapidly inoculated by microorganisms from the environment, including the incubator, feed, water, and handling (Proszkowiec-Weglarz et al., 2022). Within the first three days post-hatch, facultative anaerobes such as Streptococcus and Escherichia rapidly establish, lowering the gut’s redox potential by consuming oxygen and creating conditions suitable for obligate anaerobes (Maki et al., 2019). These pioneering communities are subsequently displaced by anaerobic members of the Firmicutes phylum, including Ruminococcus and Lachnospiraceae. By day 10, the appearance of slower-growing anaerobes like Romboutsia spp. signifies a shift towards greater diversity and evenness, with age being the primary driver of compositional change (Jurburg et al., 2019).

Figure 3. Microbial colonisation and succession in the broiler GIT (Made using Adobe Photoshop, Adobe Inc, 2019)
Colonisation patterns further vary by gut segment as shown in Figure 1. Low-pH regions (crop, proventriculus, gizzard) are typically dominated by Lactobacillus, whereas the small intestine hosts sparser populations of genera such as Lactobacillus, Escherichia, Enterococcus, and Clostridium (da Silva et al., 2014).
The process culminates in a stable “climax community,” most developed within the ceca, which becomes dominated by the phyla Firmicutes, including Clostridium, Enterococcus, Bacillus, and Ruminococcus (da Silva et al., 2014; Maki et al., 2019). This mature, metabolically specialized microbiota is essential for host digestion, immune maturation, and pathogen resistance. Delays in microbiota succession impair the beneficial role of commensal gut microbes, heightening the susceptibility of chicks to enteric diseases such as necrotic enteritis (Pan and Yu, 2014; Maki et al., 2019). This imbalance, termed dysbiosis, triggers inflammation and disrupts digestive and immune functions, ultimately resulting in significant production losses (Teirlynck et al., 2011; Maki et al., 2019). To address this, the poultry industry employs competitive exclusion strategies. A study by Angelakis and Raoult, (2010) showed that intragastrically administering Lactobacillus spp microbial cultures to day-old chicks helps rapidly establish a higher weight gain, healthy and protective gut microbiome.
6.0 Microbial-microbe interactions in the avian GIT
Bacterial populations coordinate their behavior through quorum sensing, a cell-to-cell communication system mediated by secreted chemical signals called auto-inducers (Rattray et al., 2022). This mechanism regulates collective behaviors, including virulence and biofilm formation, and facilitates interspecies and even host-bacteria communication (Coolahan and Whalen, 2025). As in other microbial communities, different members of the gut microbiome interact in various ways, including competition, cooperation, and antagonism. Competition for nutrients and attachment sites is common among bacterial populations in the avian GIT. Beneficial species (e.g., Lactobacillus, Bifidobacteria) occupy adhesion sites (due to their ability to adhere to and metabolize mucin), forming a biofilm which blocks the attachment and colonization by pathogenic bacteria such as Salmonella and Clostridium spp, a phenomenon known as “competitive exclusion” (Zhao, and Maynard, 2022; Peña et al., 2025). For instance, Svetoch et al. (2011) have shown that Lactobacillus salivarius strains isolated from the chicken gastrointestinal tract secrete bacteriocins active against pathogens like Salmonella, Enteritidis, and Campylobacter jejuni.
The production of bacteriostatic and bactericidal substances by gut microbes represents another key form of microbial interaction. Beneficial bacteria break down carbohydrates to produce SCFA such as lactic acid, acetic acid and butyric acid (Peng et al., 2024; Teng et al., 2023). These reduce the intestinal pH, making the environment hostile for pathogenic bacteria (Ali et al., 2022). In vitro studies have demonstrated that lactic acid bacteria ferment carbohydrates to produce lactic acid, which inhibits the growth of pathogens such as E. coli, Salmonella, Typhimurium, and C. perfringens (Hinton et al., 1992; Murry et al., 2004; Pan and Yu, 2014). Furthermore, an in vivo study demonstrated a negative correlation between SCFA concentrations and the abundance of the Enterobacteriaceae family in the ceca of broilers (van Der Wielen et al., 2000).
Certain bacteria produce microbial peptides, known as bacteriocins, which can selectively inhibit the growth of competing bacteria. For example, strains of Lactobacillus salivarius isolated from the chicken gastrointestinal tract produce bacteriocins active against both Gram-negative and Gram-positive bacteria, including Salmonella Enteritidis and Campylobacter jejuni (Stern et al., 2006; Svetoch et al., 2011; Pan and Yu, 2014). Similarly, Teo and Tan, (2005), and Shin et al. (2008) demonstrated that bacteriocins from broiler chicken isolates of Enterococcus faecium, Pediococcus pentosaceus, and Bacillus subtilis inhibit Clostridium perfringens and Listeria monocytogenes. The ability to produce bacteriocins is a key trait in probiotic selection, as it directly targets common pathogens. Horizontal gene transfer (HGT) also occurs among avian gut microbiota (Goldenfeld and Woese, 2007). Notably, this process includes the exchange of virulence genes between poultry enteric pathogens, which can directly enhance the pathogenicity of the recipient bacteria (Johnson et al., 2010). Predominant commensal gut microorganisms often possess specific traits such as bacteriocin production or mucin utilization which enable them to outcompete pathogens and thrive in the GIT (Pan and Yu, 2014). Pathogens can acquire these advantageous traits, through horizontal gene transfer (van Reenen and Dicks, 2011; Juhas, 2015; Oladeinde et al., 2021). This can enhance a pathogen’s competitiveness and survival.
7.0 Interactions between gut microbiome and host
A complex web of bidirectional interactions exists between the poultry host and its intestinal microbiome. This symbiotic relationship is fundamentally characterized by a mutual exchange of nutrients and by the profound regulatory effects of the microbiota on the host’s intestinal development, physiological processes, and immune system maturation (Aruwa and Sabiu, 2024; Wickramasuriya et al., 2022; Fathima et al., 2022; Gao et al., 2024). The positive interactions with the avian host come mainly from beneficial bacteria such as Firmicutes (e.g Lactobacillus spp) and Bifidobacterium.
7.1. Nutritional interactions
Most readily digestible dietary carbohydrates are digested and absorbed by the host in the proximal gut, leaving indigestible polysaccharides, oligosaccharides, and residual sugars for bacterial fermentation in the distal regions. Intestinal bacteria hydrolyze these complex carbohydrates into simple sugars, which are then fermented to produce SCFAs, primarily acetate, propionate, and butyrate (Ali et al., 2022). SCFA production increases as young birds develop. In broilers, cecal acetate, propionate, and butyrate are undetectable at one day old (van der Wielen et al., 2000). These SCFAs are absorbed across the cecal epithelium via passive diffusion and enter various host metabolic pathways. Butyrate, in particular, serves as a crucial energy source for intestinal epithelial cells (Zhang et al., 2025; Yuan et al., 2025). Beyond energy provision, SCFAs regulate intestinal blood flow, stimulate enterocyte growth and proliferation, modulate mucin production, and influence local immune responses (Liu et al., 2023.
Gut bacteria contribute to host nitrogen metabolism by catabolizing uric acid (from urine that retrograde-peristalsis moves into the ceca) into ammonia, which the host can use for amino acid synthesis (Hajra et al., 2024; Moniaci, 2017). While some dietary nitrogen is incorporated into bacterial protein, this resource is largely lost in feces because most bacterial digestion occurs in the ceca, which cannot absorb protein (Pan and Yu, 2014; Moniaci, 2017). However, chickens can access this bacterial protein through coprophagy, which is possible when they are housed on hard floors (Pan and Yu, 2014). The poultry gut microbiome also synthesizes vitamins, notably B vitamins (Uebanso et al., 2020), but like bacterial protein, most of these are excreted unless reclaimed through coprophagy; this is evidenced by the higher vitamin requirements of cage-housed birds. In a reciprocal relationship, the host provides nutrients to the microbiota, primarily in the form of mucins secreted by gut cells, which serve as a carbon, nitrogen, and energy source for commensal bacteria such as Bacteroides and Bifidobacterium and Akkermansia muciniphila (Corfield, 2018; Suriano et al., 2022). While mucin degradation is well-documented in other species, these bacterial groups are also present in poultry, suggesting a similar metabolic relationship likely exists.
7.2. Effects of microbiome on broiler intestinal morphology and physiology
The gut microbiome significantly influences broiler intestinal development and structure. Research using germ-free (GF) models has demonstrated that GF chickens possess a smaller intestinal mass with thinner intestinal walls compared to conventionally raised birds (Furuse and Okumura, 1994; Gabriel and Mallet, 2006). This developmental stimulation is partly attributed to microbially produced short-chain fatty acids (SCFAs), which promote enterocyte growth and proliferation (Le Blay et al., 2000; Blottière et al., 2003). Supporting this mechanism, studies show that broilers fed fermentable carbohydrates, which enhance microbial SCFA production, exhibit improved gut morphology (Qaisrani et al., 2015; Liao et al., 2020).
Beyond development, the microbiome critically shapes intestinal morphology (Bayer et al., 2021). Birds with a depleted or absent microbiota, such as GF models, display shorter villi and shallower crypts (Gabriel et al., 2006). Supplementing the diet with probiotic species like Lactobacillus acidophilus, Bacillus subtilis, and Saccharomyces cerevisiae has been shown to improve duodenal villus architecture and increase the ileal villus height-to-crypt depth ratio (Chae et al., 2012; Hernández-Coronado et al., 2025). However, pathogenic disruptions can severely damage this morphology by causing necrotic enteritis. For example, infections with Eimeria spp. or C. perfringens cause significant reductions in villus height and the villus height-to-crypt depth ratio (Golder et al., 2011; Goo et al., 2023). Furthermore, the microbiome modulates intestinal function, as evidenced by the higher activity of key digestive enzymes like intestinal alkaline phosphatase in conventional birds compared to their GF counterparts (Palmer et al., 1983).
7.3. Microbiome and avian host immunity
The gut microbiome is recognized as a key determinant of health and functions as an integral immune “organ” in chickens (Kogut and Santin, 2020; Sun et al., 2022). To establish an infection, enteric pathogens must first adhere to and breach the intestinal epithelial barrier. In healthy birds, this process is actively impeded by the resident commensal bacteria. These microbial communities densely colonize the intestinal mucosa, forming a protective biofilm (Katu et al., 2025; da Silva et al., 2024). This layer of dense and complex microbial communities can effectively block the attachment and subsequent colonization by most invading enteric pathogens. Some commensal bacteria in the gut, such as Lactobacillus salivarius produce bacteriocins, to inhibit the growth of competing microbes selectively (Maki et al., 2019; Svetoch et al., 2011; Predescu et al., 2024).
Beyond this physical barrier, the gut microbiome interacts with the host’s innate immune system to shape subsequent adaptive immune responses (Kogut et al., 2020). For example, administration of a probiotic containing Lactobacillus acidophilus, Bifidobacterium bifidum, and Streptococcus faecalis has been shown to increase systemic antibody responses to sheep red blood cells, elevate intestinal IgG against tetanus toxoid, and boost serum IgG and IgM reactive to both tetanus toxoid and Clostridium perfringens α-toxin (Pan and Yu, 2014). The precise mechanism remains unclear, but it is hypothesized that probiotics may stimulate the production of Th2 cytokines such as IL-4 and IL-10, thereby promoting antibody-mediated immunity (Kang and Im, 2015). The gut microbiome also significantly influences cell-mediated immunity. Research using germ-free, conventional, and gnotobiotic chickens has demonstrated that the complexity of the enteric microbiome profoundly affects the gut T cell repertoire (Mwangi et al., 2010). Furthermore, different Lactobacillus species can induce distinct cytokine expression in T cells in chicken cecal tonsils, contributing to intestinal immune homeostasis (Brismin et al., 2012). It is important to emphasize that immune modulation is not solely the function of administered probiotics or reactions to specific pathogens. The native community of commensal bacteria is also a major immunoregulatory agent (Brisbin et al., 2008). Future research must focus on identifying which commensal species are immunologically significant and elucidating their specific mechanisms of action to fully harness their potential for enhancing poultry health.
7.4. Interactions between gut microbiome and avian diet
Diet stands as the primary environmental driver shaping the intestinal microbiome in poultry, with specific feed components exerting profound and selective pressures on microbial community structure, function, and, consequently, host health. The journey of feed through the gastrointestinal tract activates a cascade of microbial interactions. Diets rich in viscous, non-starch polysaccharides (NSPs), such as those derived from wheat, barley, or rye, increase digesta viscosity, slow intestinal transit, and reduce nutrient digestibility (Pan and Yu, 2014). This altered luminal environment creates an ideal niche for the proliferation of Clostridium perfringens, directly predisposing birds to necrotic enteritis. In contrast, corn-based diets, lower in these NSPs, do not trigger the same pathogenic bloom. Beyond pathogen dynamics, the carbohydrate profile fundamentally shapes the commensal landscape; for instance, a standard corn-soybean ration favors Lactobacillus agilis type R5, while a wheat middlings-based diet favours the type R1.
The influence of protein is equally significant. The source matters: while soybean meal is a staple, using fermented cottonseed meal as a protein alternative has been shown to increase beneficial Lactobacilli and decrease coliforms in the broiler cecum (Ashayerizadeh et al., 2024). Conversely, diets high in animal protein, such as fishmeal, enrich the hindgut with undigested protein, providing a substrate that favors C. perfringens and elevates necrotic enteritis risk (Drew and Syed, 2004). Furthermore, the dietary fat source modulates this ecosystem; broilers fed animal fat (lard/ tallow) harbor greater ileal C. perfringens populations than those fed soy oil, demonstrating that lipid composition is a key microbial determinant (Knarreborg et al., 2002; Pan and Yu, 2014). Strategically, the poultry industry employs feed additives to directly modulate this complex community toward a healthier state. Enzymes like xylanase and β-glucanase are being used to break down NSPs, and create conditions that favor beneficial lactic acid bacteria over adversaries like E. coli (Pan and Yu, 2014; Kim et al., 2025). Some interventions operate on a subtler level, interfering with bacterial communication. The use of fermented feed, for example, can exert a profound and targeted influence on the gut microbial ecosystem, delivering a consortium of beneficial effects.
8. The dawn of fermented feed ingredients in broiler nutrition
The adoption of fermented feed ingredients marks a paradigm shift in broiler nutrition. This modern application of an ancient technique is a strategic biotechnological response to the converging demands for antibiotic-free meat, enhanced feed safety, and sustainable production. Its primary drivers are the industry’s search for effective antibiotic alternatives, the need to improve feed efficiency, and the opportunity to upcycle agricultural waste.
In response to global restrictions on in-feed antimicrobial growth promoters, the poultry industry has prioritized the search for effective, natural alternatives to sustain gut health and productivity (Salim et al., 2018; Rahman et al., 2022). Fermented feed has risen as a leading solution in this endeavor. This process not only fosters a favorable intestinal environment but also directly introduces beneficial microorganisms, such as Lactobacillus and Bifidobacterium, into the gastrointestinal tract (Predescu et al., 2024). These microbes benefit the avian host through positive nutritional contributions and by supporting intestinal development, physiological function, and immune system maturation (Sun et al., 2022; da Silva et al., 2024; Predescu et al., 2024). Another key driver to fermented feedstuffs is the enhancement of feed efficiency and value. Feed ingredients of plant origin contain tannins, phytic acid, and protease inhibitors (Abbas, 2020; Tadele, 2015; Erdaw and Beyene, 2018). These disrupt nutrient digestion, absorption and bioavailability in broilers (Akande et al., 2010; Tadele et al., 2015; Zhang et al., 2022; Bloot et al., 2023; Diribi and Fromsa 2025). Fermentation with fungal or bacterial microbes produces specialized enzymes such as tannases, phytases, cellulases and xylanases which breakdown the ANF, thereby enhancing nutrient availability to broilers, hence feed efficiency and value (Motta et al., 2013; Sokrab et al., 2014; Predescu et al., 2024; Hodzi et al., 2025).
In alignment with SDG 12, there is a growing focus on upcycling industrial waste to mitigate its environmental impact, which has led to innovations like the fermentation of these materials for livestock nutrition (Mnisi et al., 2022; Bibi et al., 2023; He and Cui., 2025). The fermentation process serves as a transformative biotechnology, converting low-value by-products such as fruit pomaces, cereal brans, and vegetable residues into high-quality, nutritious feed components (Wang et al., 2017; Mnisi et al., 2022; Katu et al., 2025). This practice not only diverts organic matter from landfills, reducing greenhouse gas emissions and pollution, but also establishes a circular economy within the agri-food sector. It adds tangible value to side streams, enhancing resource efficiency and creating a more sustainable and resilient production model.
The use of fermented feed in broiler nutrition is not merely an additive but a systemic upgrade, significantly improving growth performance metrics like feed conversion ratio and average daily gain (Gungor et al., 2021a; Cheng et al., 2022; Predescu et al., 2024; Hodzi et al., 2025; Dastar et al., 2025; Katu et al., 2025). Beyond mere growth, it elevates meat quality by positively influencing attributes such as fatty acid profiles and reducing cholesterol (Cao et al., 2012; Sun et al., 2022; Kim and Kang, 2016; Ding et al., 2021). Crucially, fermented feed acts directly on the gastrointestinal system, promoting optimal gut morphology through increased villus height and a healthier villus-to-crypt ratio (Qu et al., 2025; Xu et al., 2025; Katu et al., 2025; Gungor et al., 2021a). This structural improvement fosters a more robust gut barrier. Simultaneously, it beneficially modulates the gut microbiota, enriching populations of beneficial bacteria (e.g., Lactobacillus) while suppressing pathogenic ones (Gungor et al., 2021a; Li et al., 2020; Predescu et al., 2024; Katu et al., 2025) which collectively strengthens overall gut health, immune function, and nutrient absorption.
9. Fermenting broiler feeds
9.1. The process
The industry employs several core techniques to ferment feed ingredients, primarily liquid state fermentation (LSF), solid-state fermentation (SSF), and ensiling (Predescu et al., 2024). Liquid fermentation yields a nutrient-dense slurry rich in organic acids and live microbes, while SSF offers practical advantages like greater product stability, lower energy consumption, and reduced wastewater (Chen, 2013; Xu et al., 2023). Ensiling typically utilizes fresh, moist plant materials that naturally support vigorous microbial activity. A critical principle across all methods is the use of defined starter cultures, such as Bacillus strains or Aspergillus niger, to ensure a consistent and safe process, as spontaneous fermentation risks the growth of undesirable pathogens. Aspergillus niger, is valued for its production of a broad spectrum of enzymes including hemicellulases, hydrolases, pectinases, proteases, amylases, lipases, and tannases (Pinto et al., 2021, Katu et al., 2025), while various Bacillus strains, are favoured for their spore-forming ability which ensures survival through feed processing and the harsh gastrointestinal tract environment (Ramlucken et al., 2020; Bahaddad et al., 2023; Katu et al., 2025). For efficacy, research suggests a target concentration of live microorganisms in the final fermented biomass between 10⁵ and 10⁹ colony-forming units per gram (CFU/g) (Dai et al., 2020). During fermentation process, the composition of raw materials, the formulation of the starter culture, and the specific conditions of temperature, humidity, and duration during the fermentation itself should be meticulously controlled (Peng et al., 2022; Hodzi et al., 2025). Strict post-production quality control is equally vital to maintain the feed’s safety and efficacy.
9.2. The outcomes
Fermented feed offers a diverse array of benefits as previously shown in Figure 3. The benefits are linked to improved gut microbiota, which is responsible for maintaining overall intestinal homeostasis. Fermentation enhances broiler feed quality by breaking down complex compounds, making nutrients more digestible and increasing their bioavailability for absorption by intestinal epithelial cells (Haryati Supriyati et al., 2015). A primary benefit is the significant reduction of anti-nutritional factors (ANFs) like tannins, phytates, and gossypol, which otherwise compromise gut integrity by inducing inflammation and mucosal damage (Sokrab et al., 2014; Predescu et al., 2024; Cardoso Dal Pont et al., 2020). Many ingredients common in broiler nutrition, including whole cereal grains, oilseeds, and their by-products, contain antinutritional factors (ANFs), such as tannins, saponins, phytates, gossypol, trypsin inhibitors, and non-starch polysaccharides (Abbas., 2020; Samtiya et al., 2020; Nalluri and Karri, 2021). These compounds adversely affect the bioavailability of nutrients across various feed components. Tannins, for example, form stable complexes with both dietary proteins and digestive enzymes like trypsin and amylase, directly blocking the breakdown and assimilation of proteins and carbohydrates (Akande et al., 2010). Similarly, phytic acid acts as a potent chelator, binding essential minerals such as calcium, zinc, and iron into insoluble salts that are indigestible to the bird, while also interfering with starch metabolism (Tadele et al., 2015; Zhang et al., 2022; Bloot et al., 2023). Protease inhibitors present another barrier by competitively and irreversibly binding to the active sites of protein-digesting enzymes, effectively halting the crucial process of protein digestion and amino acid release (Tadele, 2015; Akande et al., 2010; Diribi and Fromsa 2025). The microbial fermentation process serves as a powerful form of “external pre-digestion” that systematically dismantles these complex compounds, facilitating efficient nutrient digestion and absorption (Haryati Supriyati et al., 2015; Katu et al., 2025).
Microbial fermentation reduces the fiber (including cellulose, hemicellulose, pectin, and lignin) content of plant cereals and cereal by-products, significantly improving feed digestibility and palatability (Katu et al., 2025). This is particularly important for monogastric animals like pigs and poultry, which lack the enzymes that degrade these polysaccharides. Fermentation of rice bran with rumen microbes significantly reduced crude fiber and acid detergent fiber by 13% and 7%, respectively (Debi et al., 2022). Enzyme pre-treatment can also be done before microbial fermentation to enhance the outcomes. For instance, Zhu et al. (2021) demonstrated that pretreating rapeseed meal (RSM) with fiber-degrading enzymes (cellulase and pectinase) prior to fermentation with a mixed culture of lactobacilli (Limosilactobacillus reuteri, Lactiplantibacillus plantarum, and Lactobacillus johnsonii) significantly reduces crude fiber content. The enzymatic activity of Bacillus species, notably B. amyloliquefaciens, is essential for fiber breakdown (Katu et al., 2025). By producing enzymes such as α-amylases, β-glucanases, and proteases, they convert complex fibers into simple sugars and other metabolites, thereby improving the nutritional value of fiber-rich materials (Priest et al., 1987).
Concomitantly, the process elevates concentrations of beneficial components, including probiotics (Soren et al., 2024; Hodzi et al., 2025; Katu et al., 2025). Probiotics enhance commensal community in the GIT, which have protective effects through two primary modes of action. The first is metabolic antagonism, driven by the continual conversion of carbohydrates into SCFAs, which sustains a low-pH barrier inimical to many harmful microbes (Guo et al., 2021). The second is direct microbial warfare; these beneficial bacteria synthesize and secrete a battery of antimicrobial compounds, including specialized bacteriocins, which can selectively target and inhibit intestinal pathogens (Pan and Yu, 2014; Predescu et al., 2024). This combination of environmental modification and targeted inhibition allows the commensal microbiome to effectively limit colonization by invaders, thereby enhancing intestinal health and resilience. Additionally, fermented feed acts as a direct source of health-promoting metabolites, including SCFAs and essential vitamins synthesized by beneficial fermentative bacteria (Katu et al., 2025). SCFAs also aid in extending feed shelf life by inhibiting spoilage molds and bacteria (Katu et al., 2025). SCFAs will result in reduced luminal pH, which selectively inhibits acid-sensitive pathogenic and spoilage microorganisms while favoring acid-tolerant commensals, particularly Lactobacillus species (Guo et al., 2021). SCFAs, particularly butyrate, serve as a crucial energy source for intestinal epithelial cells (Zhang et al., 2025; Yuan et al., 2025). SCFAs also regulate intestinal blood flow, stimulate enterocyte growth and proliferation, modulate mucin production, and influence local immune responses (Liu et al., 2023.
The combined effects of reduced ANFs, increased nutrient availability, and direct microbial benefits translate to improved intestinal morphology, microbiota and function. Fermented feed primarily influences the poultry gut microbiota by directly introducing beneficial microorganisms and metabolites, thereby suppressing pathogens and improving the gut environment for health-promoting bacteria (Guo et al., 2021; Pan and Yu, 2014; Kang et al., 2025). This foundational gut health directly drives superior broiler growth performance, evidenced by enhanced feed intake, increased body weight gain, and more efficient feed conversion (Soren et al., 2024; Predescu et al., 2024).
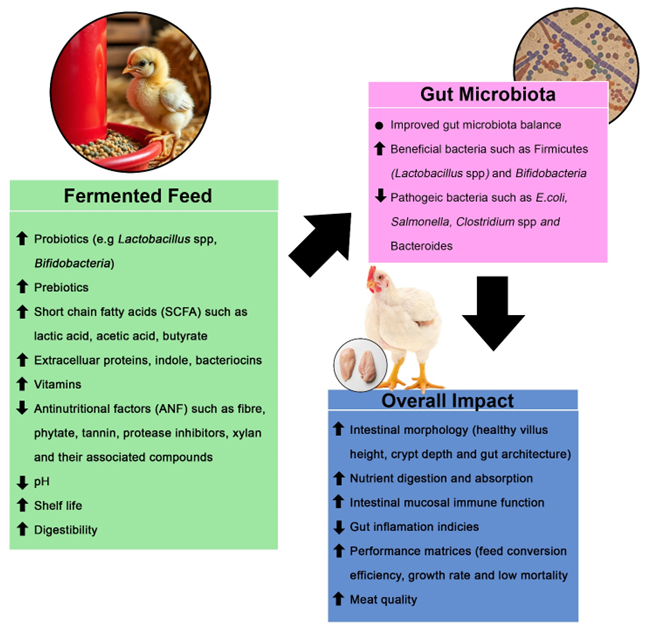
Figure 4. Fermented feed improves overall broiler growth performance and product quality (Made using Adobe Photoshop, Adobe Inc, 2019)
10. Scientific evidence showing effects of fermented feeds on broiler intestinal microbiome
Although fermented feed has been widely studied for its impacts on growth, health, and product quality, its influence on the broiler gut microbiome has received limited attention, resulting in inconsistent conclusions. A growing body of evidence, however, confirms that fermented feeds can significantly modulate microbial communities in the broiler gut. As summarised in Table 1, studies employing various fermentation agents, conditions, and inclusion levels have demonstrated these effects.
Table 1. Fermented feedstuffs commonly used in broiler diets and their effects on gut microbiome
| Feed Category | Feedstuff | Fermentation Type & Microbes | Effect on Gut Microbiota | References |
|---|---|---|---|---|
| Legumes | Soybean | Bacillus amyloliquefaciens, Lactobacillus acidophilus, Saccharomyces cerevisiae; 37°C, 24 h; SSF | ↑ Lachnospiraceae, Lachnoclostridium, Gastranaerophilales, Lactobacillus; ↓ Escherichia-Shigella, Clostridiales | Li et al., 2020 |
| Cereals | Corn, soybean, wheat (6:2:2) | Lactobacillus casei; SSF | ↑ Lactobacillaceae; ↓ Delsulfobacterota, Desulfovibriona, Negativicutes | Peng et al., 2022 |
| Wheat | Lactobacillus plantarum; 36°C, 4 h; LSF | ↑ Lactobacillus spp.; no change in E. coli & total bacteria | Kim & Kang, 2016 | |
| Brans | Corn, soybean, wheat bran | Bacillus spp., Lactobacillus spp., Saccharomyces cerevisiae | ↑ Parasutterella, Butyricicoccus, Erysipelotrichaceae | Liu et al., 2021 |
| Wheat bran | Laetiporus sulphureus; Bacillus amyloliquefaciens; Saccharomyces cerevisiae; SSF | ↑ Lactobacillaceae; ↓ Proteobacteria; ↑ ileal LAB (10%) | Lin & Lee, 2020; Teng et al., 2017 | |
| Rice bran | Lactobacillus plantarum, Saccharomyces cerevisiae, Bacillus subtilis; SSF | No significant change in Lactobacillus, E. coli, Salmonella | Kang et al., 2015 | |
| Seed Meals | Cottonseed | Bacillus subtilis, Saccharomycetes; 7 d; LSF | ↑ Cecal Lactobacilli | Wang et al., 2017 |
| Palm kernel | Paenibacillus polymyxa; 9 d; SSF | ↑ LAB; ↓ Enterobacteriaceae | Alshelmani et al., 2016; 2021 | |
| Rapeseed | Lactobacillus acidophilus, Bacillus subtilis, Aspergillus niger; SSF | ↑ LAB; ↓ ileal coliforms | Dastar et al., 2025 | |
| Grape seed | Saccharomyces cerevisiae; LSF | ↑ Shannon & Simpson indices; ↑ Firmicutes; ↓ Bacteroidetes | Nan et al., 2022 | |
| Leaf Meals | Dandelion | — | ↓ Diversity indices; ↓ Bacteroidota, Bacteroides, Alistipes, Verrucomicrobiota; ↑ Firmicutes | Mao et al., 2023 |
| Fruit Pomaces | Banana peels | — | ↓ Ileal coliform population | Sugiharto et al., 2019 |
| Grape | Saccharomyces cerevisiae; Aspergillus niger; SSF | ↑ Firmicutes; ↓ Bacteroidetes; ↓ Clostridium perfringens | Nan et al., 2022; Gungor et al., 2021a | |
| Pomegranate | Aspergillus niger; SSF | ↓ Clostridium perfringens | Gungor et al., 2021b | |
| Sour cherry | Aspergillus niger; SSF | ↑ Cecal Lactobacillus spp. | Gungor & Erener, 2020 | |
| Vegetable Pomaces | Tomato pomace | Aspergillus niger; SSF | No significant microbial changes | Gungor et al., 2024 |
SSF – Solid State Fermentation; LSF – Liquid State Fermentation
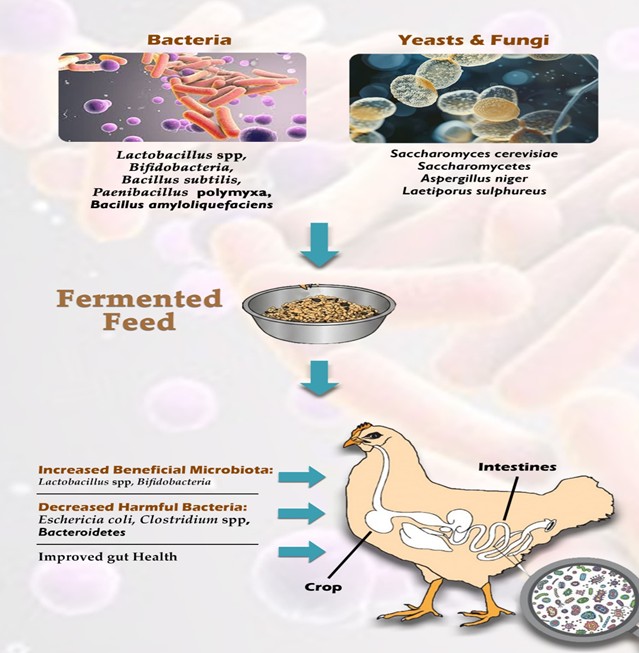
Figure 5. The microorganisms used to ferment the broiler feeds and their impact on bird’s microbiota. Made using Adobe Photoshop, Adobe Inc (2019)
In one study, fermented rapeseed meal (RSM) increased cecal and colonic Lactobacilli counts compared to both control diets and unfermented rapeseed meal (Dastar et al., 2025). Similarly, a 25% inclusion of fermented soybean meal elevated cecal abundances of beneficial genera such as Lachnospiraceae, Lachnoclostridium, Gastranaerophilales, and Lactobacillus, while reducing populations of Escherichia-Shigella and Clostridiales (Li et al., 2020). This pattern indicates that fermented feed can reshape the gut microbiome by promoting commensal bacteria and suppressing pathogenic species. Furthermore, a study using fermented wheat bran at 5% inclusion increased the family Lactobacillaceae and suppressed the phylum Proteobacteria (Lin and Lee 2020). Likewise, a diet containing a blend of fermented brans (maize bran, wheat bran, and soybean bran) enhanced the abundance of beneficial bacteria, including genus Parasutterella, Butyricicoccus, and Erysipelotrichaceae (Liu et al., 2021). In contrast, Kang et al. (2015) reported that the inclusion of 10 g/kg fermented rice bran did not significantly alter the cecal populations of Lactobacillus, Escherichia coli, and Salmonella. This, however, is a low inclusion level relative to other studies and may have been insufficient to induce a measurable change.
This principle of microbiota modulation also applies to fermented seed meals and seed kernels. It was reported that 17.9% inclusion of fermented cottonseed meal significantly increased cecal Lactobacilli counts in broilers (Wang et al., 2017). Similarly, fermented rapeseed meal significantly increased the LAB population in the crop, while reducing coliform counts in the ileum (Dastar et al., 2025). Additionally, Nan et al. (2022) reported that 4 % and 6% inclusion of grapeseed meal increased the Shannon and Simpson indices of intestinal microbiota. The same study also showed a reduction in the relative cecal abundance of Bacteroidetes. In a separate study by Alshelmani et al. (2016), they showed that a 15% inclusion of fermented palm kernel cake significantly increased lactic acid bacteria and decreased the Enterobacteriaceae. The consistency in findings regarding microbiota modulation by fermented seed meals and seed kernels demonstrates that this intervention is a reliable strategy for enhancing the gut microbiome in broilers.
As part of the valorization of industrial waste into broiler feed, fruit pomace has also been shown to alter the broiler intestinal microbiome. A study by Gungor et al. (2021a) demonstrated that 15 g/kg inclusion of fermented grape pomace (FTP) reduced the cecal Clostridium perfringens count in broilers. Fermented banana (Sugiharto et al., 2019), Pomegranate (Gungor et al., 2021b), and sour cherry (Gungor and Erner, 2020) all altered broiler gut microbiota. Hodzi et al. (2025) also summarized the impacts of fermented fruit pomaces on the intestinal microbiota of broiler chickens. Fermented tomato pomace (FTP) did not alter cecal Lactobacillus spp., Enterococcus spp., E. coli, C. jejuni, S. aureus, and C. perfringens counts at 10 g/kg inclusion (Gungor et al., 2024), likely due to a low inclusion level. In contrast, Mao et al. (2023) showed that even minimal inclusions (0.5 and 1g/kg) of fermented dandelion (FD) induced significant microbial changes by increasing the abundance of Firmicutes while reducing the abundance of Verrucomicrobiota. The same inclusion level also decreased alpha-diversity indices (observed species, Shannon, Chao1, and ACE) and lowered the abundance of Bacteroidota, Bacteroides, and Alistipes. This discrepancy suggests that the observed difference in microbial modulation is driven not solely by inclusion level but also by the inherent bioactive potency of the specific fermented ingredient.
11. Conclusion
Fermented feed components, including prebiotics and short-chain fatty acids, modulate specific microbial populations to foster a balanced intestinal microbiota in broilers. Achieving optimal feed quality and safety requires a tailored fermentation process, informed by substrate characteristics, careful selection of fermentation microorganisms and conditions, as well as routine nutritional assessments. Collectively, the reviewed literature demonstrates that fermented feed beneficially alters the broiler intestinal microbiota. This modulation consistently enhances the abundance of beneficial bacteria, such as Lactobacillus spp., while suppressing potentially harmful populations, including Clostridia spp. The resulting microbial shifts contribute to improved nutrient absorption, stronger disease resistance, and enhanced overall growth performance. Discrepancies in reported outcomes can often be attributed to variations in inclusion levels and the intrinsic properties of the fermented substrate. Future research should therefore focus on refining strategies for the effective integration of fermented feeds into broiler diets. This necessitates a deeper investigation into how the type and composition of the raw material, alongside the specific microorganisms used in fermentation, influence the final physiological and microbial outcomes in broiler production.
Author Contributions: Conceptualisation: Tonderai Mutibvu; Writing – Original draft preparation: Pride Hodzi; Writing – Review and editing: Soul Washaya, Godfrey Bernard Nyamushamba, Tonderai Mutibvu, Pride Hodzi; Validation: Soul Washaya, Godfrey Bernard Nyamushamba, Tonderai Mutibvu, Pride Hodzi; Resources: Pride Hodzi; Visualisation: Soul Washaya, Godfrey Bernard Nyamushamba, Tonderai Mutibvu. All authors have read and agreed to the published version of the manuscript.
Funding: This research received no external funding.
Acknowledgments: Authors gratefully acknowledge the Great Zimbabwe University, University of Zimbabwe and Bindura University of Science Education.
Conflicts of Interest: The authors declare no conflicts of interest.
Artificial Intelligence: AI was not used for this original research article.
References
Abbas Hilmi HT, Surakka A, Apajalahti J, Saris PE. 2007. Identification of the most abundant Lactobacillus species in the crop of 1-and 5-week-old broiler chickens. Applied and Environmental Microbiology, 73(24), 7867-7873. https://doi.org/10.1128/AEM.01128-07
Abbas G. 2020. An augmented review about anti-nutrients and toxins of feed stuff and their control strategies, a step toward sustainable resource utilization. International Journal of Agriculture Innovations and Research, 8(4), 2319-1473. https://ijair.org/index.php/issues?view=publication&task=show&id=1323
Afonso CL, Afonso AM. 2023. Next-generation sequencing for the detection of microbial agents in avian clinical samples. Veterinary Sciences, 10(12), 690. https://doi.org/10.3390/vetsci10120690
Akande KE, Doma UD, Agu HO, Adamu HM. 2010. Major antinutrients found in plant protein sources: Their effect on nutrition. Pakistan Journal of Nutrition 9 (8), 827-832. https://www.researchgate.net/publication/49965865_Major_Antinutrients_Found_in_Plant_Protein_Sources_Their_Effect_on_Nutrition
Alemao CA, Budden KF, Gomez HM, Rehman SF, Marshall JE, Shukla SD, Hansbro PM. 2021. Impact of diet and the bacterial microbiome on the mucous barrier and immune disorders. Allergy, 76(3), 714-734. https://doi.org/10.1111/all.14548
Alemka A, Whelan S, Gough R, Clyne M, Gallagher ME, Carrington SD, Bourke B. 2010. Purified chicken intestinal mucin attenuates Campylobacter jejuni pathogenicity in vitro. Journal of Medical Microbiology, 59, 898-903. http://dx.doi.org/10.1099/jmm.0.019315-0
Ali Q, Ma S, La S, Guo Z, Liu B, Gao Z, Shi Y. 2022. Microbial short-chain fatty acids: a bridge between dietary fibers and poultry gut health -a review. Animal Bioscience, 35(10), 1461. https://doi.org/10.5713/ab.21.0562
Alshelmani MI, Kaka U, Abdalla EA, Humam AM, Zamani HU. 2021. Effect of feeding fermented and non-fermented palm kernel cake on the performance of broiler chickens: a review. World’s Poultry Science Journal, 77(2), 377-388. https://doi.org/10.1080/00439339.2021.1910472
Alshelmani MI, Loh TC, Foo HL, Sazili AQ, Lau WH. 2016. Effect of feeding different levels of palm kernel cake fermented by Paenibacillus polymyxa ATCC 842 on nutrient digestibility, intestinal morphology, and gut microflora in broiler chickens. Animal Feed Science and Technology, 216, 216-224. https://doi.org/10.1016/j.anifeedsci.2016.03.019
Angelakis E, Raoult D. 2010. The increase of Lactobacillus species in the gut flora of newborn broiler chicks and ducks is associated with weight gain. PLoS One, 5(5), e10463. https://doi.org/10.1371/journal.pone.0010463
Aruwa CE, Sabiu S. 2024. Interplay of poultry–microbiome interactions–influencing factors and microbes in poultry infections and metabolic disorders. British Poultry Science, 65(5), 523-537. https://doi.org/10.1080/00071668.2024.2356666
Aruwa CE, Pillay C, Nyaga MM, Sabiu S. 2021. Poultry gut health–microbiome functions, environmental impacts, microbiome engineering and advancements in characterization technologies. Journal of Animal Science and Biotechnology, 12(1), 119. https://doi.org/10.1186/s40104-021-00640-9
Ashayerizadeh A, Jazi V, Sharifi F, Toghyani M, Mohebodini H, Kim IH, Roura E. 2024. Fermented but not irradiated cottonseed meal has the potential to partially substitute soybean meal in broiler chickens. Animals, 14(19), 2797. https://doi.org/10.3390/ani14192797
Awad WA, Hess C, Hess M. 2017. Enteric pathogens and their toxin-induced disruption of the intestinal barrier through alteration of tight junctions in chickens. Toxins, 9(2), 60. https://doi.org/10.3390/toxins9020060
Bahaddad SA, Almalki MHK, Alghamdi OA, Sohrab SS, Yasir M, Azhar EI, Chouayekh H. 2023. Bacillus species as direct-fed microbial antibiotic alternatives for monogastric production. Probiotics Antimicrobes Proteins, 15, 1–16. https://doi.org/10.1007/s12602-022-09909-5
Bailey R. 2011. Intestinal microbiota and the pathogenesis of dysbacteriosis in broiler chickens (Doctoral dissertation, University of East Anglia). https://ueaeprints.uea.ac.uk/id/eprint/33034/1/2010BaileyRAPhD.pdf
Berg G, Rybakova D, Fischer D, Cernava T, Vergès MCC, Charles T, Schloter M. 2020. Microbiome definition re-visited: Old concepts and new challenges. Microbiome, 8(1), 103. https://doi.org/10.1186/s40168-020-00875-0
Bibi F, Ilyas N, Saeed M, Shabir S, Shati AA, Alfaifi MY, Sayyed RZ. 2023. Innovative production of value-added products using agro-industrial wastes via solid-state fermentation. Environmental Science and Pollution Research, 30(60), 125197-125213. https://doi.org/10.1007/s11356-023-28765-6
Bloot APM, Kalschne DL, Amaral JAS, Baraldi IJ, Canan C. 2023. A review of phytic acid sources, obtention, and applications. Food Reviews International, 39(1), 73-92. https://doi.org/10.1080/87559129.2021.1906697
Blottière HM, Buecher B, Galmiche JP, Cherbut C. 2003. Molecular analysis of the effect of short-chain fatty acids on intestinal cell proliferation. Proceedings of the Nutrition Society, 62, 101-106. http://dx.doi.org/10.1079/PNS2002215
Brignardello-Petersen R, Santesso N, Guyatt GH. 2025. Systematic reviews of the literature: an introduction to current methods. American Journal of Epidemiology, 194(2), 536-542. https://doi.org/10.1093/aje/kwae232
Brisbin JT, Gong J, Sharif S. 2008. Interactions between commensal bacteria and the gut-associated immune system of the chicken. Animal Health Research Reviews, 9(1), 101. https://doi.org/10.10.1017/S146625230800145X
Brisbin JT, Parvizi P, Sharif S. 2012. Differential cytokine expression in T-cell subsets of chicken caecal tonsils co-cultured with three species of Lactobacillus. Beneficial Microbes, 3, 205-210 http://dx.doi.org/10.3920/BM2012.0014
Byrne CM, Clyne M, Bourke B. 2007. Campylobacter jejuni adhere to and invade chicken intestinal epithelial cells in vitro. Microbiology, 153, 561-569. http://dx.doi.org/10.1099/mic.0.2006/000711-0
Cao FL, Zhang XH, Yu WW, Zhao LG, Wang T. 2012. Effect of feeding fermented Ginkgo biloba leaves on growth performance, meat quality, and lipid metabolism in broilers. Poultry Science, 91(5), 1210-1221. https://doi.org/10.3382/ps.2011-01886
Cardoso Dal Pont G, Farnell M, Farnell Y, Kogut MH. 2020. Dietary factors as triggers of low-grade chronic intestinal inflammation in poultry. Microorganisms, 8(1), 139. https://doi.org/10.3390/microorganisms8010139
Chae B, Ingale S, Kim J, Kim K, Sen S, Lee S, Khong C, Kim EK, Kwon IK. 2012. Effect of dietary supplementation of probiotics on performance, caecal microbiology and small intestinal morphology of broiler chickens. Animal Nutrition and Feed Technology, 12, 1-12. https://scispace.com/journals/animal-nutrition-and-feed-technology-3r4o3w8e/2012
Chen B, Li D, Leng D, Kui H, Bai X, Wang T. 2022. Gut microbiota and meat quality. Frontiers in Microbiology, 13, 951726. https://doi.org/10.3389/fmicb.2022.951726
Chen H. 2013. Modern solid state fermentation. Netherlands: Springer, 599. https://doi.org/10.1007/978-94-007-6043-1
Chen R, Chen J, Xiang Y, Chen Y, Shen W, Wang W, He X. 2022. Differential modulation of innate antiviral profiles in the intestinal Lamina propria cells of chickens infected with infectious bursal disease viruses of different virulence. Viruses, 14(2), 393. https://doi.org/10.3390/v14020393
Coolahan M, Whalen KE. 2025. A review of quorum-sensing and its role in mediating interkingdom interactions in the ocean. Communications Biology, 8(1), 179. https://doi.org/10.1038/s42003-025-07608-9
Corfield AP. 2018. The interaction of the gut microbiota with the mucus barrier in health and disease in humans. Microorganisms, 6(3), 78. https://doi.org/10.3390/microorganisms6030078
da Silva WA, dos Santos Nascimento JC, Rabello CBV, de Souza LFA, Ribeiro AG, da Silva DA, de Araújo Santos AC. 2024. The role of intestinal microbiome in the health and production of commercial poultry: A review. In: Agricultural sciences unveiled: exploring the dynamics of farming and sustainability (Chapter 8 pp.67-78). Atena Editora Publishers, Brazil. https://doi.org/10.22533/at.ed.9662430048
Dai Z, Cui L, Li J, Wang B, Guo L, Wu Z, Zhu W, Wu G. 2020. Fermentation Techniques in Feed Production; Elsevier eBooks: Amsterdam, The Netherlands; pp. 407–429. https://doi.org/10.1016/B978-0-12-817052-6.00024-0
Dastar B, Ashayerizadeh A, Sharifi F, Jazi V. 2025. Replacement of soybean meal with fermented rapeseed meal in broiler diets: impacts on growth performance, gut health, and nutrient digestibility. Poultry Science, 104(11), 105616. https://doi.org/10.1016/j.psj.2025.105616
Debi MR, Wichert BA, Liesegang A. 2022. Anaerobic fermentation of rice bran with rumen liquor for reducing their fiber components to use as chicken feed. Heliyon, 8(4). https://doi.org/10.1016/j.heliyon.2022.e09275
Deng ZC, Cao KX, Huang YX, Peng Z, Zhao L, Yi D, Sun LH. 2025. Comprehensive cultivation of the broiler gut microbiota guides bacterial isolation from chickens. Science China Life Sciences, 68(3), 836-845. https://doi.org/10.1007/s11427-024-2735-8
Díaz-Sánchez S, Hanning I, Pendleton S, D’Souza D. 2013. Next-generation sequencing: The future of molecular genetics in poultry production and food safety. Poultry Science, 92(2), 562-572. https://doi.org/10.3382/ps.2012-02741
Dibner JJ, Richards JD. The digestive system: challenges and opportunities. 2004. Journal of Applied Poultry Research, 13(1), 86-93. https://doi.org/10.1093/japr/13.1.86
Ding Y, Jiang X, Yao X, Zhang H, Song Z, He X, Cao R. 2021. Effects of feeding fermented mulberry leaf powder on growth performance, slaughter performance, and meat quality in chicken broilers. Animals, 11(11), 3294. https://doi.org/10.3390/ani11113294
Diribi M, Fromsa I. 2025. A review on livestock feed antinutrients: impact on productivity and mitigation strategies. Russian Journal of Agricultural and Socio-Economic Sciences, 166(10), 50-62. https://rjoas.com/issue-2025-10/article_05.pdf
Drew MD, Syed NA, Goldade BG, Laarveld B, Van Kessel AG. 2004. Effects of dietary protein source and level on intestinal populations of Clostridium perfringens in broiler chickens. Poultry Science, 83, 414-420. https://doi.org/10.1093/ps/83.3.414
Ducatelle R, Goossens E, Eeckhaut V, Van Immerseel F. 2023. Poultry gut health and beyond. Animal Nutrition, 13, 240-248. https://doi.org/10.1016/j.aninu.2023.03.005
El Sabry MI, Yalcin S. 2023. Factors influencing the development of gastrointestinal tract and nutrient transporters’ function during the embryonic life of chickens—A review. Journal of Animal Physiology and Animal Nutrition, 107(6), https://doi.org/10.1111/jpn.13852
Erdaw MM, Beyene WT. 2018. Anti-nutrients reduce poultry productivity: influence of trypsin inhibitors on pancreas. Asian Journal of Poultry Science, 12(1), 14-24. https://doi.org/10.3923/ajpsaj.2018.14.24
Fancher CA, Zhang L, Kiess AS, Adhikari PA, Dinh TT, Sukumaran AT. 2020. Avian pathogenic Escherichia coli and Clostridium perfringens: Challenges in no antibiotics ever broiler production and potential solutions. Microorganisms, 8(10), 1533. https://doi.org/10.3390/microorganisms8101533
Fathima S, Shanmugasundaram R, Adams D, Selvaraj RK. 2022. Gastrointestinal microbiota and their manipulation for improved growth and performance in chickens. Foods, 11(10), 1401. https://doi.org/10.3390/foods11101401
Feng J, Liu X, Xu ZR, Wang YZ, Liu JX. 2007. Effects of fermented soybean meal on digestive enzyme activities and intestinal morphology in broilers. Poultry Science, 86, 1149–1154. https://doi.org/10.1093/ps/86.6.1149
Furuse M, Okumura J. 1994. Nutritional and physiological characteristics in germ-free chickens. Comparative Biochemistry Physiology A Physiology, 109, 547-556. http://dx.doi.org/10.1016/0300-9629(94)90193-7
Gabriel I, Lessire M, Mallet S, Guillot JF. 2006. Microflora of the digestive tract: Critical factors and consequences for poultry. Worlds Poultry Science Journal, 62, 499-512. https://doi.org/10.1017/S0043933906001115
Gao G, Jiao Y, Kwok LY, Zhong Z. 2024. Gut Microbiome-host genetics co-evolution shapes adiposity by modulating energy and lipid metabolism in selectively bred broiler chickens. Animals, 14(22), 3174. https://doi.org/10.3390/ani14223174
Goldenfeld N, Woese C. 2007. Biology’s next revolution. Nature, 445, 369. http://dx.doi.org/10.1038/445369a
Golder HM, Geier MS, Forder RE, Hynd PI, Hughes RJ. 2011. Effects of necrotic enteritis challenge on intestinal micro-architecture and mucin profile. British Poultry Science, 52, 500-506. http://dx.doi.org/10.1080/00071668.2011.587183
Goo D, Choi J, Ko H, Choppa VSR, Liu G, Lillehoj HS, Kim WK. 2023. Effects of Eimeria maxima infection doses on growth performance and gut health in dual-infection model of necrotic enteritis in broiler chickens. Frontiers in Physiology, 14, 1269398. https://doi.org/10.3389/fphys.2023.1269398
Gungor E, Erener G. 2020. Effect of dietary raw and fermented sour cherry kernel (Prunus cerasus L.) on digestibility, intestinal morphology and caecal microflora in broiler chickens. Poultry Science, 99(1), 471-478. https://doi.org/10.3382/ps/pez538
Gungor E, Altop A, Erener G. 2021. Effect of raw and fermented grape pomace on the growth performance, antioxidant status, intestinal morphology, and selected bacterial species in broiler chicks. Animals, 11(2), 364. https://doi.org/10.3390/ani11020364
Gungor E, Altop A, Erener G. 2024. Effect of fermented tomato pomace on the growth performance, antioxidant capacity, and intestinal microflora in broiler chickens. Animal Science Journal, 95(1), e13885. https://doi.org/10.1111/asj.13885
Guo L, Lv J, Liu Y, Ma H, Chen B, Hao K, Min Y. 2021. Effects of different fermented feeds on production performance, cecal microorganisms, and intestinal immunity of laying hens. Animals, 11(10), 2799. https://doi.org/10.3390/ani11102799
Hajra D, Kirthivasan N, Chakravortty D. 2024. Symbiotic synergy from sponges to humans: Microflora–host harmony is crucial for ensuring survival and shielding against invading pathogens. ACS Infectious Diseases, 10(2), 317-336. https://doi.org/10.1021/acsinfecdis.3c00554
Hansen ME, Ibrahim Y, Desai TA, Koval M. 2024. Nanostructure-mediated transport of therapeutics through epithelial barriers. International Journal of Molecular Sciences, 25(13), 7098. https://doi.org/10.3390/ijms25137098
Haryati Supriyati T, Susanti T, Susana IWR. 2015. Nutritional value of rice bran fermented by Bacillus amyloliquefaciens and humic substances and its utilization as a feed ingredient for broiler chickens. Asian-Australasian Journal of Animal Science 28, 231–238. http://dx.doi.org/10.5713/ajas.14.0039
He D, Cui C. 2025. Fermentation of organic wastes for feed protein production: Focus on agricultural residues and industrial by-products tied to agriculture. Fermentation, 11(9), 528. https://doi.org/10.3390/fermentation11090528
Hernández-Coronado AC, Cervantes M, González F, Valle A, Arce N, Vásquez N, Morales A. 2025. Effect of probiotic supplementation on productive performance and epithelial intestinal integrity of broiler chickens exposed to heat stress. Tropical Animal Health and Production, 57(5), 235. https://doi.org/10.1007/s11250-025-04488-3
Hinton A Jr, Spates GE, Corrier DE, Hume ME, Deloach JR, Scanlan CM. 1991. In vitro inhibition of Salmonella typhimurium and Escherichia coli 0157: H7 by an anaerobic gram-positive coccus isolated from the cecal contents of adult chickens. Journal of Food Protection, 54 (7), 496-501.
https://doi.org/10.4315/0362-028X-54.7.496
Jia Y, Si W, Hong Z, Qu M, Zhu N, Liu S, Li G. 2019. Toll-like receptor 2-mediated induction of avian β-defensin 9 by Lactobacillus rhamnosus and its cellular components in chicken intestinal epithelial cells. Food and Agricultural Immunology, 30(1), 398-417. https://doi.org/10.1080/09540105.2019.1593325
Jian Z, Wu H, Yan S, Li T, Zhao R, Zhao J, Dou T. 2025. Species and functional composition of cecal microbiota and resistance gene diversity in different Yunnan native chicken breeds: A metagenomic analysis. Poultry Science, 104(7), 105138. https://doi.org/10.1016/j.psj.2025.105138
Johnson TJ, Thorsness JL, Anderson CP, Lynne AM, Foley SL, Han J, Fricke WF, McDermott PF, White DG, Khatri M. 2010. Horizontal gene transfer of a ColV plasmid has resulted in a dominant avian clonal type of Salmonella enterica serovar Kentucky. PLoS One, 5, e15524. http://dx.doi.org/10.1371/journal.pone.0015524
Juhas M. 2015. Horizontal gene transfer in human pathogens. Critical Reviews in Microbiology, 41(1), 101-108. https://doi.org/10.3109/1040841X.2013.804031
Jurburg SD, Brouwer MS, Ceccarelli D, van der Goot J, Jansman AJ, Bossers A. 2019. Patterns of community assembly in the developing chicken microbiome reveal rapid primary succession. Microbiologyopen, 8(9), e00821. https://doi.org/10.1002/mbo3.821
Kang HJ, Im SH. 2015. Probiotics as an immune modulator. Journal of Nutritional Science and Vitaminology, 61(Supplement), S103-S105. https://doi.org/10.3177/jnsv.61.S103
Kang HK, Kim JH, Kim CH. 2015. Effect of dietary supplementation with fermented rice bran on the growth performance, blood parameters and intestinal microflora of broiler chickens. European Poultry Science, 79, 1-11. https://doi.org/10.1399/eps.2015.112
Katu JK, Tóth T, Varga L. 2025. Enhancing the nutritional quality of low-grade poultry feed ingredients through fermentation: A review. Agriculture, 15(5), 476. https://doi.org/10.3390/agriculture15050476
Katu JK, Tóth T, Ásványi B, Hatvan Z, Varga L. 2025. Effect of Fermented Feed on Growth Performance and Gut Health of Broilers: A Review. Animals, 15(13), 1957. https://doi.org/10.3390/ani15131957
Khan TJ, Hasan MN, Azhar EI, Yasir M. 2019. Association of gut dysbiosis with intestinal metabolites in response to antibiotic treatment. Human Microbiome Journal, 11, 100054. https://doi.org/10.1016/j.humic.2018.11.004
Kim CH, Kang HK. 2016. Effects of fermented barley or wheat as feed supplement on growth performance, gut health and meat quality of broilers. European Poultry Science, 80, 2016, 1-11 https://doi.org/10.1399/eps.2016.162
Kim E, Choct M, Fickler A, Pasquali GA, Hall L, Crowley TM, Sharma NK. 2025. Supplementation of β-mannanase alone or in combination with xylanase and β-glucanase enhanced growth performance, non-starch polysaccharide degradation, and gastrointestinal environment of broilers offered wheat-based diets. Animal Nutrition, 23, 429-437. https://doi.org/10.1016/j.aninu.2025.05.008
Knarreborg A, Simon MA, Engberg RM, Jensen BB, Tannock GW. 2002. Effects of dietary fat source and subtherapeutic levels of antibiotic on the bacterial community in the ileum of broiler chickens at various ages. Applied Environmental Microbiology, 68, 5918- 5924. http://dx.doi.org/10.1128/AEM.68.12.5918-5924.2002
Kogut MH. 2019. The effect of microbiome modulation on the intestinal health of poultry. Animal Feed Science and Technology, 250, 32-40. https://doi.org/10.1016/j.anifeedsci.2018.10.008
Kogut MH, Lee A, Santin E. 2020. Microbiome and pathogen interaction with the immune system. Poultry Science, 99(4), 1906-1913. https://doi.org/10.1016/j.psj.2019.12.011
Le Blay G, Blottière HM, Ferrier L, Le Foll E, Bonnet C, Galmiche JP, Cherbut C. 2000. Short-chain fatty acids induce cytoskeletal and extracellular protein modifications associated with modulation of proliferation on primary culture of rat intestinal smooth muscle cells. Digestive Diseases and Sciences, 45, 1623-1630. http://dx.doi.org/10.1023/A:1005529414765
Lee SH, Hosseindoust A, Laxman Ingale S, Rathi PC, Yoon SY, Choi JW, Kim JS. 2020. Thermostable xylanase derived from Trichoderma citrinoviride increases growth performance and non-starch polysaccharide degradation in broiler chickens. British Poultry Science, 61(1), 57-62. https://doi.org/10.1080/00071668.2019.1673316
Li Y, Guo B, Wu Z, Wang W, Li C, Liu G, Cai H. 2020. Effects of fermented soybean meal supplementation on the growth performance and cecal microbiota community of broiler chickens. Animals, 10(6), 1098. https://doi.org/10.3390/ani10061098
Lin WC, Lee TT. 2020. Effects of Laetiporus sulphureus-fermented wheat bran on growth performance, intestinal microbiota and digesta characteristics in broiler chickens. Animals, 10(9), 1457. https://doi.org/10.3390/ani10091457
Liu XF, Shao JH, Liao YT, Wang LN, Jia Y, Dong PJ, Zhang X. 2023. Regulation of short-chain fatty acids in the immune system. Frontiers in Immunology, 14, 1186892. https://doi.org/10.3389/fimmu.2023.1186892
Liu Y, Feng J, Wang Y, Lv J, Li J, Guo L, Min Y. 2021. Fermented corn-soybean meal mixed feed modulates intestinal morphology, barrier functions and caecal microbiota in laying hens. Animals, 11, 3059. https://doi.org/10.3390/ani11113059
Lundberg R, Scharch C, Sandvang D. 2021. The link between broiler flock heterogeneity and cecal microbiome composition. Animal Microbiome, 3(1), 54. https://doi.org/10.1186/s42523-021-00110-7
Maki JJ, Klima CL, Sylte MJ, Looft T. 2019. The microbial pecking order: utilization of intestinal microbiota for poultry health. Microorganisms, 7(10), 376. https://doi.org/10.3390/microorganisms7100376
Mao J, Wang Y, Duan T, Yin N, Dong C, Ren X, Qi J. 2023. Effect of fermented dandelion on productive performance, meat quality, immune function, and intestinal microbiota of broiler chickens. BMC Veterinary Research, 19(1), 178. https://doi.org/10.1186/s12917-023-03751-9
Mnisi CM, Mhlongo G, Manyeula F. 2022. Fruit pomaces as functional ingredients in poultry nutrition: A review. Frontiers in Animal Science, 3, 883988. https://doi.org/10.3389/fanim.2022.883988
Moniaci P. 2017. Assessment of the impact of different feeding strategies on chicken gastrointestinal tract by shotgun metagenomic sequencing to fight colonization by potential foodborne pathogens. PhD Thesis, University of Bologna, Italy. https://amsdottorato.unibo.it/id/eprint/7953/1/Ph.D._Thesis_Paola_Moniaci.pdf
Motta F, Andrade C, Santana M. 2013. A Review of Xylanase Production by the Fermentation of Xylan: Classification. Sustainable degradation of lignocellulosic biomass: Techniques, applications and commercialization. London, UK: IntechOpen, 251-276. https://doi.org/10.5772/53544
Muneeb M, Khan EU, Ahmad S, Naveed S, Ali M, Qazi MA, Abdollahi, MR. 2024. An updated review on alternative strategies to antibiotics against necrotic enteritis in commercial broiler chickens. World’s Poultry Science Journal, 80(3), 821-870. https://doi.org/10.1080/00439339.2024.2330934
Muramatsu T, Takasu O, Okumura J. 1993. Research note: fructose feeding increases lower gut weights in germ-free and conventional chicks. Poultry Science, 72, 1597-1600. http://dx.doi.org/10.3382/ps.0721597
Murry A Jr., Hinton A Jr., Morrison H. 2004. Inhibition of growth of Escherichia coli, Salmonella typhimurium, and Clostridia perfringens on chicken feed media by Lactobacillus salivarius and Lactobacillus plantarum. International Journal of Poultry Science, 3, 603-607; http://dx.doi.org/10.3923/ijps.2004.603.607
Mwangi WN, Beal RK, Powers C, Wu X, Humphrey T, Watson M, Bailey M, Friedman A, Smith AL. 2010. Regional and global changes in TCR alphabeta T cell repertoires in the gut are dependent upon the complexity of the enteric microflora. Developmental & Comparative Immunology, 34, 406-417 http://dx.doi.org/10.1016/j.dci.2009.11.009
Nalluri N, Karri VR. 2021. Grain legumes and their by-products: As a nutrient rich feed supplement in the sustainable intensification of commercial poultry industry. Sustainable Agriculture Reviews 51: Legume Agriculture and Biotechnology 2, 51-96. https://doi.org/10.1007/978-3-030-68828-8_3
Nan S, Yao M, Zhang X, Wang H, Li J, Niu J, Nie C. 2022. Fermented grape seed meal promotes broiler growth and reduces abdominal fat deposition through intestinal microorganisms. Frontiers in Microbiology, 13, 994033. https://doi.org/10.3389/fmicb.2022.994033
Oladeinde A, Abdo Z, Press MO, Cook K, Cox NA, Zwirzitz B, Ritz C. 2021. Horizontal gene transfer is the main driver of antimicrobial resistance in broiler chicks infected with Salmonella enterica serovar Heidelberg. Msystems, 6, e00729-21. https://doi.org/10.1128/mSystems.00729-21 https://journals.asm.org/doi/pdf/10.1128/msystems.00729-21
Palmer MF, Rolls BA. 1983. The activities of some metabolic enzymes in the intestines of germ-free and conventional chicks. British Journal of Nutrition, 50, 783-790. http://dx.doi.org/10.1079/BJN19830149
Pan D, Yu Z. 2014. Intestinal microbiome of poultry and its interaction with host and diet. Gut Microbes, 5(1), 108-119. http://dx.doi.org/10.4161/gmic.26945
Pelaseyed T, Bergström JH, Gustafsson JK, Ermund A, Birchenough, GM, Schütte A, Hansson GC. 2014. The mucus and mucins of the goblet cells and enterocytes provide the first defense line of the gastrointestinal tract and interact with the immune system. Immunological Reviews, 260(1), 8-20.
Peña N, Lafuente I, Sevillano E, Feito J, Allendez G, Muñoz-Atienza E, Borrero J. 2025. Exploring the functional potential of the broiler gut microbiome using shotgun metagenomics. Genes, 16(8), 946. https://doi.org/10.3390/genes16080946
Peng W, Talpur MZ, Zeng Y, Xie P, Li J, Wang S, Wang L, Zhu X, Gao P, Jiang Q. 2022. Influence of fermented feed additive on gut morphology, immune status, and microbiota in broilers. BMC Veterinary Research, 18, 218. https://doi.org/10.1186/s12917-022-03322-4
Peng Z, Wang D, He Y, Wei Z, Xie M, Xiong T. 2024. Gut distribution, impact factor, and action mechanism of bacteriocin-producing beneficial microbes as promising antimicrobial agents in gastrointestinal infection. Probiotics and Antimicrobial Proteins, 16(5), 1516-1527. https://doi.org/10.1007/s12602-024-10222-6
Pewan SB, Kabantiyok D, Emennaa PE, Dawurung JS, Dawurung CJ, Duwil RK, Ezema C. 2025. Advancing Nigerian indigenous poultry health and production, use of probiotics as viable alternatives to antibiotics: A review. Antibiotics, 14(8), 846. https://doi.org/10.3390/antibiotics14080846
Pinto GAS, Leite SGF, Terzi SC, Couri S. 2001. Selection of tannase-producing Aspergillus niger strains. Brazilian Journal of Microbiology, 32, 24–26. https://doi.org/10.1590/S1517-83822001000100006
Predescu NC, Stefan G, Rosu MP, Papuc C. 2024. Fermented feed in broiler diets reduces the antinutritional factors, improves productive performances and modulates gut microbiome – A review. Agriculture, 14(10), 1752. https://doi.org/10.3390/agriculture14101752
Priest FG, Goodfellow M, Shute LA, Berkeley RCW. 1987 Bacillus amyloliquefaciens sp. nov., nom. rev. International Journal of Systematic Bacteriology, 37, 69–71. https://doi.org/10.1099/00207713-37-1-69
Proszkowiec-Weglarz M, Miska KB, Ellestad LE, Schreier LL, Kahl S, Darwish N, Shao J. 2022. Delayed access to feed early post-hatch affects the development and maturation of gastrointestinal tract microbiota in broiler chickens. BMC Microbiology, 22(1), 206. https://doi.org/10.1186/s12866
Qaisrani S N, Van Krimpen MM, Kwakkel RP, Verstegen MWA, Hendriks WH. 2015. Diet structure, butyric acid, and fermentable carbohydrates influence growth performance, gut morphology, and cecal fermentation characteristics in broilers. Poultry Science, 94(9), 2152-2164. https://doi.org/10.3382/ps/pev003
Qu H, Zan G, Li H, Wang X, Zhou J, Wang X, Yan H. 2025. Fermented feed promotes gut development by enhancing intestinal stem cell expansion via activation of the Wnt/β-catenin signaling pathway. Fermentation, 11(2), 52. https://doi.org/10.3390/fermentation11020052
Rahman MRT, Fliss I, Biron E. 2022. Insights in the development and uses of alternatives to antibiotic growth promoters in poultry and swine production. Antibiotics, 11(6), 766. https://doi.org/10.3390/antibiotics11060766
Ramlucken U, Lalloo R, Roets Y, Moonsamy G, van Rensburg CJ. 2020. Advantages of Bacillus-based probiotics in poultry production. Livestock Science, 241, 104215. https://doi.org/10.1016/j.livsci.2020.104215
Rattray JB, Thomas SA, Wang Y, Molotkova E, Gurney J, Varga JJ, Brown SP. 2022. Bacterial quorum sensing allows graded and bimodal cellular responses to variations in population density. MBio, 13(3), e00745-22. https://doi.org/10.1128/mbio.00745-22
Rukambile E, Sintchenko V, Muscatello G, Kock R, Alders R. 2019. Infection, colonization and shedding of Campylobacter and Salmonella in animals and their contribution to human disease: a review. Zoonoses and Public Health, 66(6), 562-578. https://doi.org/10.1111/zph.12611
Salim HM, Huque KS, Kamaruddin KM, Haque Beg A. 2018. Global restriction of using antibiotic growth promoters and alternative strategies in poultry production. Science Progress, 101(1), 52-75. https://doi.org/10.3184/003685018X15173975498947
Samtiya M, Aluko RE, Dhewa T. 2020. Plant food anti-nutritional factors and their reduction strategies: an overview. Food Production, Processing and Nutrition, 2(1), 6. https://doi.org/10.1186/s43014-020-0020-5
Sergeant MJ, Constantinidou C, Cogan TA, Bedford MR, Penn CW, Pallen, MJ. 2014. Extensive microbial and functional diversity within the chicken cecal microbiome. PLoS One, 9(3), e91941. https://doi.org/10.1371/journal.pone.0091941
Shin MS, Han SK, Ji AR, Kim KS, Lee WK. 2008. Isolation and characterization of bacteriocin-producing bacteria from the gastrointestinal tract of broiler chickens for probiotic use. Journal of Applied Microbiology, 105, 2203-2212. http://dx.doi.org/10.1111/j.1365-2672.2008.03935.x
Sokrab AM, Mohamed Ahmed IA, Babiker EE. 2014. Effect of fermentation on antinutrients, and total and extractable minerals of high and low phytate corn genotypes. Journal of Food Science and Technology, 51(10), 2608-2615. https://doi.org/10.1007/s13197-012-0787-8
Soren S, Mandal GP, Mondal S, Pradhan S, Mukherjee J, Banerjee D, Samanta I. 2024. Efficacy of Saccharomyces cerevisiae fermentation product and probiotic supplementation on growth performance, gut microflora and immunity of broiler chickens. Animals, 14(6), 866. https://doi.org/10.3390/ani14060866
Stern NJ, Svetoch EA, Eruslanov BV, Perelygin VV, Mitsevich EV, Mitsevich IP, Pokhilenko VD, Levchuk VP, Svetoch OE, Seal BS. 2006. Isolation of a Lactobacillus salivarius strain and purification of its bacteriocin, which is inhibitory to Campylobacter jejuni in the chicken gastrointestinal system. Antimicrobial Agents Chemotherapy, 50, 3111-3116. http://dx.doi.org/10.1128/AAC.00259-06
Sun H, Chen D, Cai H, Chang W, Wang Z, Liu G, Chen Z. 2022a. Effects of fermenting the plant fraction of a complete feed on the growth performance, nutrient utilization, antioxidant functions, meat quality, and intestinal microbiota of broilers. Animals, 12(20), 2870. https://doi.org/10.3390/ani12202870
Sun F, Chen J, Liu K, Tang M, Yang Y. 2022b. The avian gut microbiota: Diversity, influencing factors, and future directions. Frontiers in Microbiology, 13, 934272. https://doi.org/10.3389/fmicb.2022.934272
Suriano F, Nyström EE, Sergi D, Gustafsson JK. 2022. Diet, microbiota, and the mucus layer: The guardians of our health. Frontiers in Immunology, 13, 953196. https://doi.org/10.3389/fimmu.2022.953196
Svetoch EA, Eruslanov BV, Levchuk VP, Perelygin VV, Mitsevich EV, Mitsevich IP Stern NJ. 2011. Isolation of Lactobacillus salivarius 1077 (NRRL B-50053) and characterization of its bacteriocin, including the antimicrobial activity spectrum. Applied and Environmental Microbiology, 77(8), 2749-2754. https://doi.org/10.1128/AEM.02481-10
Tadele Y. 2015. Important anti-nutritional substances and inherent toxicants of feeds. Food Science and Quality Management, 36, 40-47. https://www.iiste.org/Journals/index.php/FSQM/article/view/19778/20140
Teirlynck E, Gussem MDE, Dewulf J, Haesebrouck F, Ducatelle R, Van Immerseel F. 2011. Morphometric evaluation of “dysbacteriosis” in broilers. Avian Pathology, 40(2), 139-144. https://doi.org/10.1080/03079457.2010.543414
Teng K, Huang F, Liu Y, Wang Y, Xia T, Yun F, Zhong J. 2023. Food and gut originated bacteriocins involved in gut microbe-host interactions. Critical Reviews in Microbiology, 49(4), 515-527. https://doi.org/10.1080/1040841X.2022.2082860
Teng PY, Chang CL, Huang CM, Chang SC, Lee TT. 2017. Effects of solid-state fermented wheat bran by Bacillus amyloliquefaciens and Saccharomyces cerevisiae on growth performance and intestinal microbiota in broiler chickens. Italian Journal of Animal Science, 16(4), 552-562. https://doi.org/10.1080/1828051X.2017.1299597
Teo AY, Tan HM. 2005. Inhibition of Clostridium perfringens by a novel strain of Bacillus subtilis isolated from the gastrointestinal tracts of healthy chickens. Applied Environmental Microbiology, 71, 4185-4190. http://dx.doi.org/10.1128/AEM.71.8.4185-4190.2005
Uebanso T, Shimohata T, Mawatari K, Takahashi A. 2020. Functional roles of B‐vitamins in the gut and gut microbiome. Molecular Nutrition & Food Research, 64(18), 2000426. https://doi.org/10.1002/mnfr.202000426
van der Wielen PW, Biesterveld S, Notermans S, Hofstra H, Urlings BA, van Knapen F. 2000. Role of volatile fatty acids in development of the cecal microflora in broiler chickens during growth. Applied and Environmental Microbiology, 66(6), 2536-2540. http://dx.doi.org/10.1128/AEM.66.6.2536-2540.2000
van Reenen CA, Dicks LM. 2011. Horizontal gene transfer amongst probiotic lactic acid bacteria and other intestinal microbiota: what are the possibilities? A review. Archives of Microbiology, 193(3), 157-168. https://doi.org/10.1007/s00203-010-0668-3
Vielmuth F. 2021. Anatomy of the oral mucosa. In: Schmidt, E. (eds) Diseases of the Oral Mucosa. Springer, Cham. https://doi.org/10.1007/978-3-030-82804-2_2
Wang Y, Deng Q, Song D, Wang W, Zhou H, Wang L, Li A. 2017. Effects of fermented cottonseed meal on growth performance, serum biochemical parameters, immune functions, antioxidative abilities, and cecal microflora in broilers. Food and Agricultural Immunology, 28(4), 725-738. https://doi.org/10.1080/09540105.2017.1311308
Wei S, Morrison M, Yu Z. 2013. Bacterial census of poultry intestinal microbiome. Poultry Science, 92(3), 671-683. https://doi.org/10.3382/ps.2012-02822
Wickramasuriya SS, Park I, Lee K, Lee Y, Kim WH, Nam H, Lillehoj HS. 2022. Role of physiology, immunity, microbiota, and infectious diseases in the gut health of poultry. Vaccines, 10(2), 172. https://www.mdpi.com/2076-393X/10/2/172
Wilkinson N, Hughes RJ, Aspden WJ, Chapman J, Moore RJ, Stanley D. 2016. The gastrointestinal tract microbiota of the Japanese quail, Coturnix japonica. Applied Microbiology and Biotechnology, 100(9), 4201-4209. https://doi.org/10.1007/s00253-015-7280-z
Wisselink HJ, Cornelissen JBWJ, Mevius DJ, Smits MA, Smidt H, Rebel JM. 2017). Antibiotics in 16-day-old broilers temporarily affect microbial and immune parameters in the gut. Poultry Science, 96(9), 3068-3078. https://doi.org/10.3382/ps/pex133
Wu D, Chen Q, Chen X, Han F, Chen Z, Wang Y. 2023. The blood–brain barrier: Structure, regulation and drug delivery. Signal Transduction and Targeted Therapy, 8(1), 217. https://doi.org/10.1038/s41392-023-01481-w
Xu C, Lu X, Ayalew H, Iqbal W, Wang Y, Yin X, Zhang H. 2025. Esterified butyric and valeric acids enhance broiler growth and intestinal health by modulating gut morphology and microbial profile. Journal of the Science of Food and Agriculture, 105(13), 7293-7309. https://doi.org/10.1002/jsfa.14441
Xu F, Wu H, Xie J, Zeng T, Hao L, Xu W, Lu L. 2023. The effects of fermented feed on the growth performance, antioxidant activity, immune function, intestinal digestive enzyme activity, morphology, and microflora of yellow-feather chickens. Animals, 13, 3545. https://doi.org/10.3390/ani13223545
Yeoman CJ, Chia N, Jeraldo P, Sipos M, Goldenfeld ND, White BA. 2012. The microbiome of the chicken gastrointestinal tract. Animal Health Research Reviews, 13(1), 89-99. https://doi.org/10.1017/S1466252312000138
Yu K, Choi I, Kim M, Pyung YJ, Lee JS, Choi Y, Yun CH. 2025. Florfenicol-induced dysbiosis impairs intestinal homeostasis and host immune system in laying hens. Journal of Animal Science and Biotechnology, 16(1), 56. https://doi.org/10.1186/s40104-025-01186-w
Yuan M, Gao K, Peng K, Bi S, Cui X, Liu Y. 2025. A review of nutritional regulation of intestinal butyrate synthesis: Interactions between dietary polysaccharides and proteins. Foods, 14(21), 3649. https://doi.org/10.3390/foods14213649
Zhang H, Sun J, Xie A, Yang H, Li Y, Mei Y, Liang Y. 2025. Enhancing butyrate synthesis and intestinal epithelial energy supply through mixed probiotic intervention in dextran sulfate sodium-induced colitis. Food Bioscience, 63, 105727. https://doi.org/10.1016/j.fbio.2024.105727
Zhang YY, Stockmann R, Ng K, Ajlouni S. 2022. Revisiting phytate-element interactions: implications for iron, zinc and calcium bioavailability, with emphasis on legumes. Critical Reviews in Food Science and Nutrition, 62(6), 1696-1712. https://doi.org/10.1080/10408398.2020.1846014
Zhao Q, Maynard CL. 2022. Mucus, commensals, and the immune system. Gut Microbes, 14(1), 2041342. https://doi.org/10.1080/19490976.2022.2041342
Zhu X, Wang L, Zhang Z, Ding L, Hang S. 2021. Combination of fiber-degrading enzymatic hydrolysis and lactobacilli fermentation enhances utilization of fiber and protein in rapeseed meal as revealed in simulated pig digestion and fermentation in vitro. Animal Feed Science and Technology, 278, 115001. https://doi.org/10.1016/j.anifeedsci.2021.115001
Disclaimer/Publisher’s Note: The statements, opinions, institutional affiliations, data contained in all publications, and all responsibilities for accuracy are solely those of the individual author(s) and contributor(s) and not of MARCIAS AUSTRALIA and AJAVAS/or the Editor(s). MARCIAS AUSTRALIA and AJAVAS/or the Editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.

