Focussed ultrasonographic assessment of rabbit post-anaesthetic gastrointestinal function in combination with feed intake and faecal output data
Matilda Fitzmaurice 1,3 Charlotte Nix 1 Dario d’Ovidio 2 Chiara Adami 1
- Department of Veterinary Medicine, University of Cambridge, Madingley Road, Cambridge, CB3 0ES, United Kingdom
- Private Practitioner, Arzano, NA, Italy
- Present address: The Ralph Veterinary Centre, Fourth Avenue Globe Business Park, Marlow, SL71YG, United Kingdom
Article Information
- Date Received: 13/05/2025
- Date Revised: 04/06/2025
- Date Accepted: 17/06/2025
- Date Published Online: 07/07/2025
Copyright: © 2025 The Authors. Published by MARCIAS AUSTRALIA, 32 Champion Drive, Rosslea, Queensland 4812, Australia. This is an open access publication under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Citation: Fitzmaurice M, Nix C, d’Ovidio D, Adami C (2025). Focussed ultrasonographic assessment of rabbit post-anaesthetic gastrointestinal function in combination with feed intake and faecal output data. Aust J Agric Vet Anim Sci (AJAVAS), 1(1), 100001
https://doi.org/10.64902/ajavas.2025.100001
Abstract
Objective assessment methods are essential to assess the effects of anaesthesia on rabbits’ gastrointestinal function. In this prospective case series, the digestive function of four pet rabbits undergoing general anaesthesia for elective orchiectomy was assessed using both abdominal ultrasonography (60 minutes pre-anaesthesia and 45-75 minutes post-anaesthesia) and quantification of their food intake and faecal output. The ultrasonographic outcome measures were duodenal and pyloric contractions, counted from a three-minute video recording for each site. A peristaltic contraction was defined as either unidirectional motion of luminal contents (sagittal plane) or a single rhythmic dilation and contraction of the lumen (transverse plane) lasting one to two seconds. Postoperative food intake was calculated by weighing the offered food using a precision scale. The faeces collected during the observation period were also weighed. Only one out of the four rabbits ate during the elapsed observation period from the end of the anaesthetic to abdominal ultrasonography, and none of them produced any faeces during this time. Abdominal ultrasonographic assessment could be completed within 15 minutes and was feasible with gentle restraint of the rabbits in sternal recumbency. Duodenal contractions were subjectively found to be more consistent and easier to detect and monitor than pyloric contractions. Based on the pilot findings of this case series, ultrasonographic assessment of duodenal contractions, together with monitoring of postoperative food intake and faecal output, may be a useful tool for the early detection of post-anaesthetic digestive dysfunction in rabbits, and potential identification of rabbits at higher risk of developing postoperative ileus.
Keywords
Duodenal contractions, general anaesthesia, ileus, Lagomorpha, monogastrics
Highlights
- Post-anaesthetic digestive dysfunction is common in rabbits, but difficult to assess
- Focussed ultrasonography may be added to food intake/faecal output measurement
- Duodenal contractions seem to be easier to detect and monitor than pyloric contractions
1.0 Introduction
Post-anaesthetic hyporexia and reduced defaecation are common in pet rabbits, affecting approximately one-third of anaesthetic cases (Lee et al., 2018). Additionally, ileus is known to be the fourth most common cause of death and the fifth most common cause of morbidity in pet rabbits in the United Kingdom (O’Neill et al., 2020). Anaesthetic drugs such as medetomidine and buprenorphine have been shown to affect gastrointestinal motility in rabbits, and may contribute to the development of ileus in this species (Martin-Flores et al., 2017; Schnellbacher et al., 2017; Deflers et al., 2018; Botman et al, 2020; Feldman et al., 2021). Peri-anaesthetic administration of gastrointestinal prokinetics is anecdotally common among practitioners managing rabbit anaesthesia. However, published evidence does not seem to support their clinical use (Di Girolamo et al., 2025; McCready et al., 2025), and one study failed to demonstrate that cisapride would mitigate gastrointestinal dysmotility caused by buprenorphine in this species (Feldman et al., 2017).
One reason for the paucity of research in this area may be the challenge of objectively assessing gastrointestinal function in the post-anaesthetic period. In clinical settings, assessment of post-anaesthetic gastrointestinal function in hospitalised rabbits typically relies on subjective behavioural observations, such as voluntary food intake and faecal output (Cooper et al., 2009; Martin-Flores et al., 2017; Mead et al., 2024). However, since hospitalisation is likely a significant stressor for rabbits, these behaviours may be inconsistent and difficult to quantify reliably.
Ultrasonographic assessment of gastrointestinal motility may offer a more objective and reproducible alternative for evaluating peri-anaesthetic gastrointestinal function. An objective and repeatable method for evaluating gastrointestinal motility could also enable earlier detection of ileus and more informed peri-anaesthetic management in clinical practice.
This pilot case series aimed to assess the feasibility of using focused abdominal ultrasonography, in combination with food intake and faecal output measurements, to evaluate post-anaesthetic gastrointestinal function in healthy pet rabbits undergoing elective orchiectomy. We hypothesised that this approach would yield clinically relevant information on pyloric and duodenal motility following surgery, and provide quantifiable parameters – specifically, the number of peristaltic contractions – for objective assessment.
2.0 Presentation
Data are reported as medians and ranges (minimum – maximum). Four healthy (ASA grade 1) client-owned male mixed breed pet rabbits (cases 1, 2, 3 and 4) aged 7 (5 – 9) months, weighing 2 (1.3-2.5) kg and with body Condition Score (BCS) of 3/5 (3/5 – 3.5/5) presented for elective orchiectomy were recruited for inclusion in this prospective case series. Exclusion criteria were a history of gastrointestinal dysmotility or dental disease, a body weight less than 0.8 kg or a BCS of either 1/5 or 5/5, based on the Pet Food Manufacturing Association (PFMA) chart (Rabbit Size-O-Meter, Pfma.org.uk).
3.0 History and clinical procedures
Following hospital admission on the morning of the procedure, rabbits were each housed individually in a small kennel, located in a quiet ward with no other species present. They were provided with free access to a hide and a known amount of food pellets, hay and water (via both bottle and bowl), all measured with a precision scale. All rabbits received 1 mg/kg of oral cisapride (Cisapride non-flavoured oral suspension, Summit Veterinary Pharmaceuticals, UK) 30 minutes prior to premedication, and were anaesthetised with the same protocol. Intramuscular (IM) premedication consisted of 50 mcg/kg of medetomidine (Sedator, Dechra Veterinary Products, UK), 30 mcg/kg of buprenorphine (Vetergesic, Ceva Animal Health, UK) and 5mg/kg of ketamine (Narketan, Vetoquinol, UK). Meloxicam (Metacam, Boehringer Ingelheim, UK) was administered subcutaneously at the dose of 0.6 mg/kg before the beginning of surgery (Miller et al., 2022). The effect of premedication was subjectively rated as good (Cases 2 – 4) to profound (Case 1) within 5 – 10 minutes of administration in all rabbits and none of them required additional sedation. Following pre- medication, oxygen was provided via a tight-fitting face mask as early as the patient was tolerant of this, and an intra- venous (IV) cannula was placed in the marginal ear vein. Anaesthesia was induced with IV alfaxalone (Alfaxan, Jurox, UK), titrated to effect to allow endotracheal intubation, in two rabbits (dose: 1.5 mg/kg for Case 1 and 0.75 mg/kg for Case 4). Two rabbits (Case 1 and Case 3) did not require IV alfaxalone prior to endotracheal intubation attempts. Topical lidocaine spray (Intubeaze, Dechra Veterinary Products, UK) was applied to the larynx (Longley, 2008), and 45 seconds later, otoscopic-guided intubation was attempted using an uncuffed endotracheal tube. A species-specific laryngeal mask (Rabbit V-gel, Jorgensen Laboratories, CO, USA) was inserted if intubation was unsuccessfully at- tempted for up to three minutes, or sooner than that if hypoxaemia (as detected with pulse-oximetry) or apnoea occurred. Otoscopic-guided endotracheal intubation was attempted in Cases 1 – 3, but was only successful in one rabbit (Case 3) within the allotted timeframe. Following administration of intravenous alfaxalone, Case 4 developed post-induction apnoea and was deemed to be too small for rapid otoscopic visualisation of the larynx; hence a V-gel was placed immediately, instead of commencing endotracheal intubation attempts. Anaesthesia was maintained with isoflurane (IsoFlo, Zoetis, UK) in oxygen via an Ayres T-piece breathing system. Rabbits were placed in reverse Trende- lenburg position for the duration of anaesthesia in order to reduce the weight of the abdominal organs on the diaphragm (Hickey et al. 2023). Intratesticular lidocaine (Lidocaine 2%, Hameln Pharma Ltd, UK) was administered prior to surgery at the dose of 1 mg/testicle (Serpieri et al. 2024). Physiological monitoring included sidestream capnography, pulse-oximetry, and measurement of non-invasive oscillometric blood pressure and oesophageal temperature (via a thermistor probe). Measured parameters were continuously monitored and recorded every five minutes on a paper record. Active warming was provided with a thermostatically controlled heat mat (HotDog, Burtons, UK). Intravenous fluid therapy (Vetivex (Hartmann’s) Solution, Dechra Veterinary Products, UK) was provided at a rate of 10 ml/kg/h. If required based on the observation of autonomic responses during surgical stimulation, rescue analgesia consisted of 0.5 mg/kg of IV ketamine (Narketan, Vetoquinol UK Ltd); this applied to one rabbit only (Case 3). Scrotal orchiectomy was performed in a standardised manner by one of three experienced veterinary surgeons, with a final year veterinary student acting as surgical assistant. Total surgical time was recorded. At the end of surgery, the rabbits were placed in sternal recumbency, isoflurane was discontinued and atipamezole (Atipam 5.0mg/ml; Dechra Veterinary Products, UK) was administered IM at the dose of 0.25 mg/kg (Kim et al., 2004). End-of-procedure oesophageal temperature and total duration of anaesthesia (from initiation of endotracheal intubation attempts to atipamezole administration) was recorded. Two rabbits exhibited mild hypothermia at the end of surgery (Cases 2 and 4; oesophageal temperatures 36.8C and 36.7C, respectively); hence active warming was continued during recovery until normothermia was re- stored. The total duration of anaesthesia was 67 (39 – 103) minutes, while surgical duration was 20 (10 – 45) minutes. All rabbits experienced an uncomplicated surgery and anaesthetic.
4.0 Assessment of post-anaesthesia food/water intake and faecal output
Food and water intake were quantified prior to ultrasonographic assessment, by subtracting the amount of food and water leftover in the kennel at the end of the postoperative observation period to that of the previously offered food and water. Urinary output was assessed qualitatively via direct observation of the litter trays, while faecal output was measured using a precision scale. The postoperative observation period was defined as the time elapsed from the end of anaesthesia (extubation of the trachea or removal of the V-gel) to removal of the rabbits from their kennels to allow abdominal ultrasonography. Only one rabbit (Case 1) was observed eating postoperatively; this was quantified as 2 g of pellets and 2 g of hay. In Cases 2, 3 and 4, eating or drinking was not observed, and weighing of food and water provisions revealed no measurable change in the amount of food or water provided. Postoperatively, no faecal or urinary output was detected in the kennel in any of the rabbits.
5.0 Diagnostic assessment: Ultrasonography
Abdominal ultrasonographic exam was performed in the conscious rabbits 30 minutes prior to administration of cisapride (approximately 30-60 minutes after hospital admission), and repeated 45 to 75 minutes after the end of anaesthesia (extubation of the trachea or removal of the V-gel). For all rabbits, the pre-anaesthetic ultrasound assessment took place between 10.30-11.30am, and the post-anaesthetic ultrasound assessment took place between 1.00-3.00pm. All ultrasound examinations were performed by the same specialist-in-training in veterinary diagnostic imaging (CN), under the supervision of aboard-certified specialist, using either a Phillips EPIQ Elite or Phillips Compact 5500 ultra- sound system (Philips Medical Systems International B.V., The Netherlands). The rabbits were positioned in sternal recumbency in order to minimise patient restraint and distress. The fur was not clipped prior to the ultrasonographic examination. Isopropyl alcohol and coupling gel were applied to the fur overlying the right craniolateral abdomen to provide acoustic coupling without the need for clipping. A high-frequency (18MHz) broadband linear array transducer (L18-5) was configured with standard B-mode ultrasound settings with manual adjustments made to depth, gain, and focal point to optimize image quality. For each patient the proximal duodenum was located through a right subcostal window with the array directed in a cranio-medial direction. The duodenum was imaged in either the sagittal or transverse plane depending on the size of the patient. B-mode still images and 3-minute video recordings of the proximal duodenum were obtained and stored in a local picture archiving and communication system (Figure 1). Peristaltic contractions were recorded over a single three-minute period, measured using a stopwatch. A peristaltic contraction was defined as either unidirectional motion of luminal contents (sagittal plane) or by a single rhythmic dilation and contraction of the lumen (transverse plane) lasting one to two seconds. The luminal contents comprised of a mixture of anechoic fluid and heterogeneously hyperechoic ingesta. To-and-fro motion of luminal contents without subsequent contraction or a churning appearance was not recorded as a duodenal peristaltic contraction. The period of three minutes was stopped and re-started if there was significant movement or breathing artifact preventing continuous evaluation. The ultrasound study was well tolerated in all rabbits, and each image acquisition period (pre- or post- anaesthesia) was less than 15 minutes in duration. Post-anaesthetic ultrasonography was performed in all four rabbits, whereas pre-anaesthetic ultrasonography was only feasible in Cases 2, 3 and 4 due to hospital scheduling constraints. It was possible to visualise both the pylorus and duodenum in Case 1, only the pylorus in Case 2, and only the duodenum in Cases 3 and 4. Contractions were counted over a three-minute period (Table 1). Visualisation of the pylorus was subjectively reported to be more challenging than visualisation of the duodenum.
Table 1.
Table 1. Number of pyloric and duodenal contractions observed via ultrasonography over a three-minute period in four rabbits pre- and post-anaesthesia for orchiectomy. A peristaltic contraction was defined as either unidirectional motion of luminal contents (sagittal plane) or a single rhythmic dilation and contraction of the lumen (transverse plane) lasting one to two seconds.
| Case | Pyloric contractions | Duodenal contractions | ||
| Pre-anaesthesia | Post-anaesthesia | Pre-anaesthesia | Post-anaesthesia | |
| 1 | NP | 6 | NP | 8 |
| 2 | 15 | 0 | NV | NV |
| 3 | NV | NV | 15 | 42 |
| 4 | NV | NV | 20 | 10 |
NV = not visualised; NP = not performed
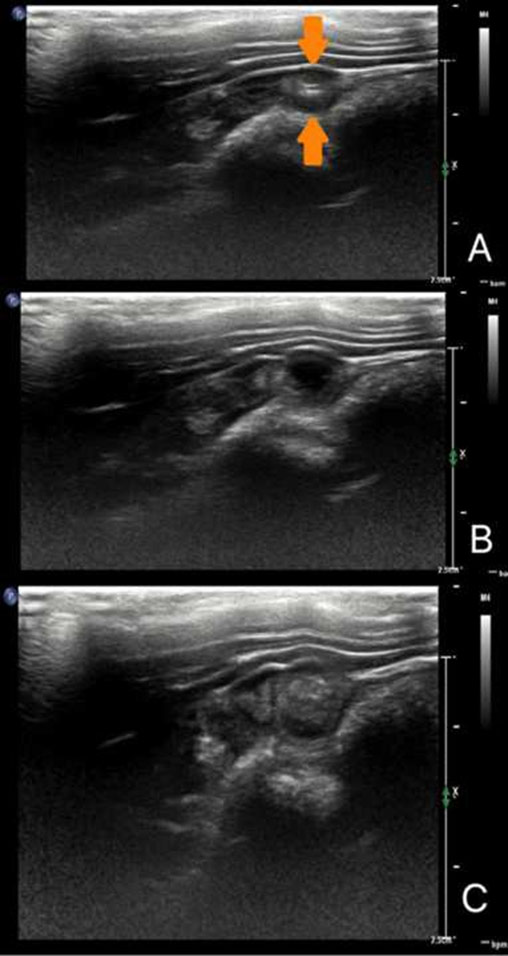
Figure 1. B-mode sagittal still ultrasound images of the duodenum in a rabbit pre-operatively demonstrating a peristaltic contraction (dilation followed by contraction). To the left is the cranial aspect of the patient and to the right is the caudal aspect of the patient. In image A, the duodenum is non-dilated, with the orange arrows delineating the walls of the duodenum and a small amount of hyperechoic gas delineating the lumen of the duodenum. In image B, the duodenum is dilated containing a bolus of intraluminal anechoic fluid. In image C, the duodenum is dilated with a bolus of heterogeneously hyperechoic ingesta. Following dilation of the duodenum, the bolus of ingesta or fluid moved ab-orally (to the right) resulting in contraction and return of the duodenum to an empty state, demonstrated in image A.
6.0 Discussion
This prospective case series assessed the feasibility of focussed ultrasonographic evaluation of pyloric and duodenal motility in hospitalised pet rabbits, with the purpose of evaluating their digestive function prior to post-anaesthetic discharge. Our pilot findings suggest that this approach is likely to be well tolerated by rabbits and that quantitative data pertaining to gastrointestinal contractions may be collected within a short timeframe of 15 minutes.
Ultrasonography has previously been utilised to assess gastrointestinal motility in rabbits pre- and post-administration of buprenorphine and a butorphanol-midazolam-ketamine combination (Deflers et al., 2018; Oura et al., 2019). It is worth noting that Deflers and colleagues used an experimental population of 15 New Zealand White rabbits. Given the diversity in breed, size, and handleability of pet rabbits seen in clinical practice, it is unclear whether this methodology would be directly translatable to a clinical setting. With respect to the methods developed by Oura and colleagues, this relied on accurate continuous doppler ultrasonographic evaluation of the peristaltic contractions, with the rabbits in dorsal recumbency and following extensive fur clipping in the conscious rabbits (Oura et al., 2019). Potential limitations of the aforementioned approach include that it may be time consuming, affected by breathing artefacts and poor tolerance by conscious rabbits – all of which may raise ethical concerns when applied to clinical patients, and may also negatively affect gastrointestinal motility by causing stress.
In this limited case series, we evaluated a quick and focussed ultrasound scan based on a few very specific outcome measures, which could be translated to future prospective clinical trials including a larger number of rabbits. Although visualisation of both the pylorus and duodenum within the 15 minutes time slot was not consistent in all rabbits, at least one of these gastrointestinal segments could always be detected and assessed in all cases. The duodenum was subjectively the most accessible and reliable part of the gastrointestinal tract to evaluate with the patient positioned in sternal recumbency from a right lateral subcostal window, suggesting that it may serve as a more practical target for assessment. This is in line with the results of a previous study, which also concluded that the peristaltic activity of rabbit stomachs could not be reliably assessed via ultrasonography, in contrast to the duodenum, for which peristaltic activity was consistently visualised (Oura et al., 2019). Focussing on duodenal contractions may therefore improve the time-efficiency and feasibility of post-anaesthetic ultrasonographic evaluation of gastrointestinal motility in clinical rabbits with minimal stress and restraint of the patient.
Marked variation in the frequency of gastrointestinal contractions was observed between cases and between timepoints, although the small sample size precludes further statistical analysis. Notably, a decrease in pyloric contractility was observed post-anaesthesia compared to baseline in Case 2, while duodenal contractility increased in Case 3 and decreased in Case 4. This variability may reflect true biological responses to anaesthesia, individual pharmacological sensitivity, variability in individual rabbit’s stress response to handling, or an inconsistent clinical effect of cisapride between subjects (Di Girolamo et al., 2025). Although there is little evidence supporting the peri-operative use of cisapride in rabbits (Di Girolamo et al., 2025; Feldman et al., 2021), pre-anaesthetic administration of a prokinetic to rabbits is standard practice at our institution, and was included to adhere to current ethical guidelines. Lastly, there was variation amongst cases regarding the intravenous dose of alfaxalone administered an induction agent. However, the effects of an intravenous bolus of alfaxalone are only expected to last for a few minutes (Gil et al., 2017) and, unlike opioids and alpha-2 agonists, alfaxalone is not known to produce significant effects on gastrointestinal motility. Therefore, we can expect the physiological effects of alfaxalone to be clinically negligible during the recovery period.
Behavioural indicators of gastrointestinal function (such as voluntary food intake and faecal output) were absent in all but one rabbit, despite ultrasonographic evidence of gastrointestinal motility in some cases. This highlights a potential lack of sensitivity that may be encountered if behavioural observations are used to infer gastrointestinal activity, and supports the value of objective imaging modalities in assessing gastrointestinal motility.
7.0 Limitations
Several limitations should be acknowledged. Firstly, the small sample size and incomplete pre-anaesthetic imaging limit the extent to which these results can be extrapolated to a broader population. It is important to emphasize that the findings of this report should be intended as pilot preliminary data, to be used as a basis to potentially design large clinical studies. Secondly, while the concept of using objective indicators of gastrointestinal motility is appealing, post-anaesthetic ultrasonographic exam, although quick, may only be applicable to clinical research and not to busy clinical settings, especially in rabbits that are prone to handling-related stress. Other potential limitations include that no period for patient acclimatisation to the hospital environment was permitted prior to the pre-anaesthetic ultrasound assessment, and that the postoperative observation time period was relatively short. However, increasing the duration of hospitalisation may contribute to stress in the rabbits, and would not be realistic in busy hospital settings.
8.0 Conclusion
Despite these limitations, this case series demonstrates that quick, focussed ultrasonographic assessment of gastrointestinal motility is feasible in conscious pet rabbits minimally restrained in sternal recumbency, and may be used as part of prospective clinical trials warranting further exploration of post-anaesthetic digestive dysfunction in this species.
Author Contributions: Conceptualization: Matilda Fitzmaurice, Dario d’Ovidio, Chiara Adami; Methodology: Matilda Fitzmaurice, Chiara Adami, Charlotte Nix; Investigation: Matilda Fitzmaurice, Charlotte Nix; Data Curation: Matilda Fitzmaurice, Charlotte Nix; Writing—Original Draft Preparation: Matilda Fitzmaurice, Chiara Adami; Writing—Review and Editing: Chiara Adami, Dario d’Ovidio. All authors have read and agreed to the published version of the manuscript.
Funding: This research received no external funding.
Ethics Approval Statement: This case series was conducted in accordance with the Declaration of Helsinki and approved by the Institutional (Department of Veterinary Medicine of the University of Cambridge) Ethics and Welfare Committee (project reference CR651; date of approval 14 December 2022).
Informed Consent Statement: Informed consent was obtained from the owners of all the pet rabbits included in this case series.
Data Availability Statement: All the relevant data acquired for this work are included in the published report.
Acknowledgments: The authors would like to thank the First Opinion Service of the Queen Veterinary School Hospital for providing the clinical cases object of this report.
Conflicts of Interest: The authors declare no conflicts of interest.
Artificial Intelligence: AI was not used for this case series article.
References
Botman J, Hontoir, F, Gustin P, Cambier C, Gabriel F, Dugdale A, Vandeweerd JM. 2020. Postanaesthetic effects of ketamine midazolam and ketamine–medetomidine on gastrointestinal transit time in rabbits anaesthetised with isoflurane. Veterinary Record, 186(8), 249. https://doi.org/10.1136/vr.105491
Cooper CS, Metcalf-Pate KA, Barat CE, Cook JA, Scorpio DG. 2009. Comparison of side effects between buprenorphine and meloxicam used postoperatively in Dutch belted rabbits (Oryctolagus cuniculus). Journal of the American Association for Laboratory Animal Science, 48(3), 279–85. https://pubmed.ncbi.nlm.nih.gov/19476717/
Di Girolamo N, Maranville RE, Pathak D, Womble W, Hanzlicek A, Sypniewski L, Brandão J. 2025. The 4 prokinetic drugs metoclopramide, cisapride, pyridostigmine, and capromorelin do not increase fecal output or food intake in healthy New Zealand rabbits (Oryctolagus cuniculus). Journal of American Veterinary Medical Association, 2, 1-7. https://doi.org/10.2460/javma.25.01.0040.
Feldman ER, Singh B, Mishkin NG, Lachenauer ER, Martin-Flores M, Daugherity EK. 2021. Effects of cisapride, buprenorphine, and their combination on gastrointestinal transit in New Zealand White Rabbits. Journal of the American Association for Laboratory Animal Science, 60(2), 221–228. https://doi.org/10.30802/aalas-jaalas-20-000064
Gil AG, Silván G, Villa A, Illera JC. 2012. Heart and respiratory rates and adrenal response to propofol or alfaxalone in rabbits. Veterinary Record, 170(17), 444–444. https://doi.org/10.1136/vr.100573
Hickey C, Guedes AG, Kennedy MJ, Tearney CC, Yamamoto K, Wendt-Hornickle E. 2023. Effect of the reverse Trendelenburg position on the incidence of gastroesophageal reflux in dogs anesthetized for elective stifle surgery. Veterinary Anaesthesia and Analgesia, 50(4), 333-340. https://doi.org/10.1016/j.vaa.2023.04.002
Kim MS, Jeong SM, Park JH, Nam TC, Seo KM. 2004. Reversal of medetomidine-ketamine combination anaesthesia in rabbits by atipamezole. Experimental Animals, 53(5), 423–428. https://doi.org/10.1538/expanim.53.423
Lee HW, Machin H, Adami C. 2018. Peri-anaesthetic mortality and nonfatal gastrointestinal complications in pet rabbits: A retrospective study on 210 cases. Veterinary Anaesthesia and Analgesia, 45(4), 520–528. https://doi.org/10.1016/j.vaa.2018.01.010
Longley L. 2008. Rabbit anaesthesia. In: Anaesthesia of Exotic Pets . Longley, L. (Editor) p. 43. W. B. Saunders, New York, USA: ISBN 9780702028885. https://doi.org/10.1016/B978-0-7020-2888-5.50004-6, Chapter 3.
Martin-Flores M, Singh B, Walsh CA, Brooks EP, Taylor L, Mitchell LM. 2017. Effects of buprenorphine, methylnaltrexone, and their combination on gastrointestinal transit in healthy New Zealand White rabbits. Journal of the American Association for Laboratory Animal Science, 56(2), 155–159. https://pmc.ncbi.nlm.nih.gov/articles/PMC5361040/
McCready JE. 2025. Therapies in exotic animal gastroenterology. Veterinary Clinics of North America Exotic Animal Practice, 28 (2), 453-483. https://doi.org/10.1016/j.cvex.2024.11.011
Mead SA, Allen MJ, Elsayed SAH, Gittel CS. 2024. Evaluating the efficacy of a peripheral nerve simulator-guided brachial plexus block in rabbits undergoing orthopaedic surgery compared to systemic analgesia. Veterinary Sciences, 11(5), 213. https://doi.org/10.3390/vetsci11050213
Miller AL, Clarkson JM, Quigley C, Neville V, Krall C, Geijer-Simpson A, Flecknell PA, Leach MC. 2022. Evaluating pain and analgesia effectiveness following routine castration in rabbits using behavior and facial expressions. Frontiers in Veterinary Science, 9,782486. https://doi.org/10.3389/fvets.2022.782486
O’Neill DG, Craven HC, Brodbelt DC, Church DB, Hedley J. 2020. Morbidity and mortality of domestic rabbits (Oryctolagus cuniculus) under primary veterinary care in England. Veterinary Record, 186(14), 451–451. https://doi.org/10.1136/vr.105592
Oura TJ, Graham JE, Knafo SE, Aarsvold S, Gladden JN, Barton BA. 2019. Evaluation of gastrointestinal activity in healthy rabbits by means of duplex Doppler ultrasonography. American Journal of Veterinary Research, 80(7), 657–662. https://doi.org/10.2460/ajvr.80.7.657
Rabbit Size-O-Meter [Internet]. Pfma.org.uk. 2017 [cited 16 June 2019]. Available from: https://www.ukpetfood.org/spotlight-on-obesity/how-to-identify-if-a-pet-needs-help/rabbit-size-o-meter-new.html
Schnellbacher RW, Divers SJ, Comolli JR, Beaufrère H, Maglaras CH, Andrade N, Barbur LA, Rosselli DD, Stejskal M, Barletta M, Mayer J, Rodriguez P, Quandt JE. 2017. Effects of intravenous administration of lidocaine and buprenorphine on gastrointestinal tract motility and signs of pain in New Zealand White rabbits after ovariohysterectomy. American Journal of Veterinary Research, 78(12),1359–1371. https://doi.org/10.2460/ajvr.78.12.1359
Serpieri M, Bonaffini, G, Ottino C, Quaranta G, Mauthe von Degerfeld M. 2024. Effects of intratesticular lidocaine in pet rabbits undergoing orchiectomy. Animals, 7;14(4):551. https://doi.org/doi:10.3390/ani14040551
Disclaimer/Publisher’s Note: The statements, opinions, institutional affiliations, data contained in all publications, and all responsibilities for accuracy are solely those of the individual author(s) and contributor(s) and not of MARCIAS AUSTRALIA and AJAVAS/or the Editor(s). MARCIAS AUSTRALIA and AJAVAS/or the Editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.

