Hypertrophic cardiomyopathy in cats at Procare Veterinary Hospital
Xuan Thanh Nguyen1 Kinh Van La1 Loan Vu Thuy Hong Nguyen1*
- Faculty of Veterinary Medicine and Animal Science, HUTECH University, Binh Thanh District, Ho Chi Minh City 717000, Vietnam
Article Information
- Date Received: 17/01/2026
- Date Revised: 11/04/2006
- Date Accepted: 26/04/2026
- Date Published Online: 27/04/2006
Copyright: © 2025 The Authors. Published by MARCIAS AUSTRALIA, 32 Champion Drive, Rosslea, Queensland 4812, Australia. This is an open access publication under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Citation: Nguyen XT, La KV, Nguyen LVTH (2025). Hypertrophic cardiomyopathy in cats at Procare Veterinary Hospital. Aust J Agric Vet Anim Sci (AJAVAS), 1(3), 100011
https://doi.org/10.64902/ajavas.2025.100011
Abstract
Hypertrophic cardiomyopathy (HCM) is the most common myocardial disease in cats and can progress to congestive heart failure. This study was conducted at Procare Veterinary Hospital from December 2024 to December 2025 to investigate the prevalence, clinical characteristics, diagnostic indices, and treatment outcomes of HCM in cats. Among 2,195 cats presented for examination, 167 cases (7.61%) were suspected of having heart disease; 30 cats (17.96%) were definitively diagnosed with HCM by echocardiography. HCM was most frequently observed in British Shorthair cats (66.66%), followed by mixed-breed cats (20.00%); sex was not an independent risk factor. Cats younger than 6 years accounted for 83.33%, and those weighing under 5 kg accounted for 73.33% (p < 0.01). Clinically, tachycardia and/or a gallop rhythm was the most common finding (34.67%), whereas cardiac murmurs were less frequent (8.00%); gallop rhythm represented 80.00% of all cardiac auscultation abnormalities. Echocardiographic parameters showed that interventricular septal thickness in diastole (IVSd) and the left atrium-to-aorta ratio (LA/Ao) increased significantly with disease stage, with LA/Ao reaching 2.55 in stage C. Vertebral heart score (VHS) gradually increased from 8.06 ± 0.15 in stage B1 to 9.83 ± 0.47 in stage C. Treatment outcomes were highly favorable in early stages (100% in B1; 94.44% in B2) but poor in the heart failure stage (16.67% in stage C), highlighting the importance of early screening by echocardiography to improve the prognosis of feline HCM. This study concludes that treatment is most effective when the disease is detected in the early or pre–heart failure stages, supplements scientific data on feline cardiovascular disease, supports early screening and the development of appropriate follow-up strategies in clinical veterinary practice in Vietnam. Based on the study findings, a key recommendation for future application by clinicians dealing with this condition in their practices is that echocardiography is essential for definitive diagnosis of HCM because clinical signs and cardiac auscultation findings have limited diagnostic specificity.
Keywords:
Hypertrophic cardiomyopathy; cat; echocardiography; treatment outcome
Highlights
- HCM is the most common myocardial disease in cats and may progress to congestive heart failure.
- Echocardiographic and radiographic indices (IVSd, LA/Ao, VHS) increase significantly with disease stage.
- Treatment is highly effective in early stages but poor in heart failure, hence the importance of early screening.
1.0 Introduction
Hypertrophic cardiomyopathy (HCM) is the most common acquired myocardial disease in cats, characterised by abnormal thickening of the left ventricular myocardium, particularly the interventricular septum and the left ventricular free wall. This results in reduced ventricular compliance, increased left atrial pressure, diastolic dysfunction, and a high risk of congestive heart failure, pulmonary edema, or arterial thromboembolism (Han et al., 2019). In the early stages, the disease is often asymptomatic; as it progresses, clinical signs such as dyspnea, lethargy, anorexia, or syncope usually appear late, making early detection difficult, and many cases are only diagnosed at an advanced stage or after sudden death (Payne et al., 2013).
Recent epidemiological studies in Europe report that the prevalence of HCM ranges from 8–15% in the general cat population and may reach 25–30% in certain pure breeds such as Maine Coon or Ragdoll (Sukumolanan et al., 2022). Ironside et al. (2021) demonstrated that left atrial enlargement and elevated NT-proBNP concentrations are two independent prognostic indicators for the risk of developing heart failure or thrombosis in preclinical HCM cats. These parameters can be monitored by Doppler echocardiography, electrocardiography, and blood biochemistry testing, thereby supporting early identification of high-risk cases.
In Vietnam, in-depth studies on feline cardiovascular diseases remain limited, particularly epidemiological studies and analyses of risk factors associated with HCM. As cat ownership is becoming increasingly popular in major urban areas, the lack of domestic scientific data has created difficulties in establishing systematic screening, diagnostic, and monitoring protocols. In clinical practice in Ho Chi Minh City, at several private veterinary facilities such as Procare Veterinary Hospital, numerous cats showing myocardial hypertrophy have been detected using imaging modalities and ancillary diagnostic tests; however, these cases have not yet been systematically collected and analysed to evaluate disease prevalence and its association with factors such as breed, age, sex, and body weight. Therefore, conducting the present study is necessary and practically significant. This study aims to investigate the prevalence, clinical characteristics, diagnostic indices, and treatment outcomes of HCM in cats.
2.0 Materials and methods
2.1. Study Site, Animals, Housing, and Experimental Design
This prospective study was conducted from December 2024 to December 2025 at Procare Veterinary Hospital, Phu Nhuan District, Ho Chi Minh City. Cats presented to Procare Veterinary Hospital during the study period were enrolled, including both cats with clinical signs suggestive of cardiac disease and cats undergoing routine health examinations. Eligible cats were between 1 and 12 years of age, regardless of sex or breed. The exclusion criteria were: Cats with secondary myocardial hypertrophy caused by systemic diseases such as hyperthyroidism, systemic hypertension, or chronic kidney disease. These were excluded to ensure inclusion of primary hypertrophic cardiomyopathy (HCM) cases only (Liu et al., 1984).
2.2. Data Collection
This was a prospective study in which all cats were systematically evaluated and recorded using a standardised medical record form in order to document clinical findings, medical history, and risk factors associated with hypertrophic cardiomyopathy (HCM).
2.2.1. General information
For each cat, the following baseline data were recorded: pet name, age, breed, sex, neuter status, body weight, and vaccination status.
2.2.2. Clinical signs and physical examination
Clinical signs recorded included tachypnea or dyspnea, especially at rest; open-mouth or labored breathing; decreased activity, lethargy, and increased sleeping; reduced appetite or anorexia; syncope or sudden collapse; and acute hindlimb paralysis with severe pain, suggestive of arterial thromboembolism (ATE). In addition to typical cardiovascular signs, general systemic manifestations were recorded, including unexplained weight loss, reduced interaction with owners, and changes in daily routine or resting behavior.
2.2.3. Medical history and risk factors
Medical history included previously diagnosed cardiac disease, history of dyspnea, pulmonary edema, or pleural effusion, and previous episodes of acute hindlimb paralysis suspected to be thromboembolic in origin.
2.2.4. Diagnostic classification of HCM
Based on history, clinical cardiovascular–respiratory examination, and risk indicators, cats suspected of HCM were classified to guide ancillary testing, disease staging, and data analysis, following ACVIM recommendations and published feline cardiology studies (Wess et al., 2010).
Group A (High-risk but clinically normal): Cats without clinical signs, presented for routine check-up, pre-anesthetic screening, or cardiac screening; belonging to high-risk breeds (e.g., Maine Coon, Ragdoll, British Shorthair, Persian, Scottish Fold) or adult/older cats. These cats showed no obvious abnormalities on auscultation but were considered at risk for preclinical HCM.
Groups B1 and B2 (Preclinical HCM): Cats without signs of congestive heart failure but with abnormal cardiac findings such as gallop rhythm, tachycardia, mild systolic murmur, or family history of heart disease. These cats may already show early structural changes but no clinical heart failure. Echocardiography was indicated to measure interventricular septal thickness (IVSd), left ventricular posterior wall thickness (LVPWd), and left atrial size (LA/Ao), with periodic follow-up.
Group C (HCM with congestive heart failure): Cats with clinical signs of CHF, including tachypnea, dyspnea, open-mouth breathing, increased resting respiratory rate, pulmonary crackles or decreased lung sounds, and radiographic evidence of pulmonary edema or pleural effusion. These cats required prompt stabilization before advanced diagnostics.
Group D (End-stage or complicated HCM): Cats with advanced disease, poor or no response to standard therapy, recurrent or refractory CHF, or severe complications such as recurrent arterial thromboembolism or repeated acute pulmonary edema. This group represented the end stage of HCM and was analyzed separately because of its distinct prognosis and treatment response (Gersh et al., 2011).
2.2.5. Radiographic evaluation
Thoracic radiography was performed after clinical assessment of respiratory status. Cats with severe dyspnea were stabilized before imaging. Right lateral and ventrodorsal views were routinely obtained; dorsoventral views were added when deemed necessary by the clinician in charge. Vertebral heart size (VHS) was measured and interpreted according to feline reference ranges (normal 6.9–8.1). Radiographs were used to assess cardiac silhouette and detect signs of pulmonary edema or pleural effusion to guide management of congestive heart failure.
2.2.6. Echocardiographic evaluation
Cats were examined in a quiet environment without routine sedation to avoid hemodynamic alterations. Hair was clipped at the right and left parasternal regions, skin cleaned, and ultrasound gel applied. Cats were positioned mainly in right lateral recumbency, with left lateral or dorsal recumbency when needed.
Transthoracic echocardiography included 2-D, M-mode, and color Doppler imaging.
2-D echocardiography: Used to evaluate overall cardiac morphology, chamber size, and ventricular wall thickness. Interventricular septal thickness in diastole (IVSd) and left ventricular posterior wall thickness in diastole (LVPWd) were measured in short-axis views at papillary muscle level.
M-mode echocardiography: Used to assess wall motion and systolic function, detect systolic anterior motion (SAM) of the mitral valve, measure left ventricular diameters in systole and diastole, and calculate functional indices. Left atrial size was assessed by the LA/Ao ratio at the aortic short-axis view (LA/Ao < 1.5: normal; ≥1.5–1.6: suggestive of left atrial enlargement). Cats were diagnosed with HCM when IVSd or LVPWd ≥ 6 mm in adult cats, in the absence of secondary causes of myocardial hypertrophy such as systemic hypertension or hyperthyroidism, consistent with ACVIM guidelines (Wess et al., 2010).
2.3. Statistical analysis
Data was recorded for each individual cat and analysed using Minitab 17 for Windows (Minitab 2026. Minitab Statistical Software. PA, USA). Quantitative variables were presented as descriptive statistics (n, mean ± SD, CV). Means were compared using analysis of variance (F-test) followed by Tukey’s post-hoc test. Proportions were compared using the Chi-square (χ²) test. Differences were considered statistically significant at p ≤ 0.05.
3.0 Results
3.1. Prevalence of Cats Suspected of HCM and Cats Diagnosed with HCM
The results of the survey on the proportion of cats suspected of having hypertrophic cardiomyopathy (HCM) and those definitively diagnosed with HCM at Procare Veterinary Hospital are presented in Fig. 1.
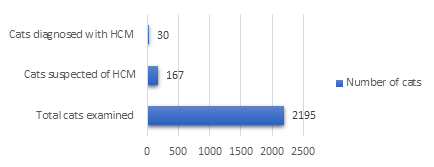
The results in Fig. 1 show that among 2,195 cats presented to Procare Veterinary Hospital during the study period, 167 cases were recorded as suspected of hypertrophic cardiomyopathy (HCM) based on clinical signs, cardiac murmurs, or suspicious findings during routine examination, accounting for 7.61% of all cases. Among the 167 suspected cases, 30 cats were definitively diagnosed with HCM, accounting for 17.96%.
3.2. Factors affecting the prevalence of HCM
The distribution of HCM by cat breed and disease stage is presented in Table 1.
Table 1. Prevalence of HCM by Cat Breed (n = 30)
| Breed | Stage B1 | Stage B2 | Stage C | Total | P-value | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Number (n) | Percentage (%) | Number (n) | Percentage (%) | Number (n) | Percentage (%) | Number (n) | Percentage (%) | |||
| Maine Coon | 0 | 0.00 | 2 | 11.11 | 0 | 0.00 | 2 | 6.67 | M-A: 0.000; | |
| British Shorthair | 6 | 100 | 9 | 50.00 | 5 | 83.33 | 20 | 66.66 | M-B: 1.000; | |
| Bengal | 0 | 0.00 | 2 | 11.11 | 0 | 0.00 | 2 | 6.67 | M-L: 0.000; | |
| Mixed-breed | 0 | 0.00 | 5 | 27.78 | 1 | 16.67 | 6 | 20.00 | A-B: 0.000; A-L: 0.000; B-L: 0.254 |
|
| Total | 6 | 100 | 18 | 100 | 6 | 100 | 30 | 100 | ||
Note: M = Maine Coon; A = British Shorthair; B = Bengal; L = Mixed-breed
Table 1 shows marked differences in disease distribution among breeds. British Shorthair cats accounted for the highest proportion of HCM (66.66%), followed by mixed-breed cats (20.00%), while Maine Coon and Bengal cats each represented 6.67%. Notably, British Shorthair cats in this study were present in all three disease stages (B1, B2, and C),. Maine Coon and Bengal cats accounted for only 6.67% of total HCM cases and were recorded exclusively in stage B2, with no cases in stages B1 or C. Mixed-breed cats accounted for 20.00% of HCM cases.
Table 2. Prevalence of HCM by sex of cat across disease stages
| Sex | Stage B1 | Stage B2 | Stage C | Total | P-value | ||||
|---|---|---|---|---|---|---|---|---|---|
| Number (n) | Percentage (%) | Number (n) | Percentage (%) | Number (n) | Percentage (%) | Number (n) | Percentage (%) | ||
| Male | 3 | 50.00 | 8 | 44.44 | 3 | 50.00 | 14 | 46.67 | 0.606 |
| Female | 3 | 50.00 | 10 | 55.56 | 3 | 50.00 | 16 | 53.33 | |
| Total | 6 | 100 | 18 | 100 | 6 | 100 | 30 | 100 | |
Table 2 shows the distribution of HCM stage in male and female cats. Male and female cats accounted for 46.67% and 53.33% of HCM prevalence, respectively (p > 0.05), indicating a relatively even distribution with no statistically significant difference by sex. In stage B1, the distribution was identical (50.00% each). Although in stage B2, females seemed slightly more frequent (55.56%) than males (44.44%), this difference was not statistically significant, hence negligible. In stage C, the proportions were equal (50.00%), indicating that disease progression is not influenced by sex.
Table 3. Prevalence of HCM by age across disease stages
| Age (years) | Stage B1 | Stage B2 | Stage C | Total | P-value | ||||
|---|---|---|---|---|---|---|---|---|---|
| Number (n) | Percentage (%) | Number (n) | Percentage (%) | Number (n) | Percentage (%) | Number (n) | Percentage (%) | ||
| <6 | 5 | 83.33 | 15 | 83.33 | 5 | 83.33 | 25 | 83.33 | 0.000 |
| ≥6 | 1 | 16.67 | 3 | 16.67 | 1 | 16.67 | 5 | 16.67 | |
| Total | 6 | 100 | 18 | 100 | 6 | 100 | 30 | 100 | |
Table 3 shows that cats younger than 6 years accounted for a significantly higher proportion (83.33%) compared with cats older than 6 years (16.67%) (p < 0.01), indicating that age is clearly associated with HCM prevalence in this population. Notably, cats < 6 years in this study were present in all stages (B1, B2, and C)
Table 4. Prevalence of HCM by body weight across disease stages
| Body weight (kg) | Stage B1 | Stage B2 | Stage C | Total | P-value | ||||
|---|---|---|---|---|---|---|---|---|---|
| Number (n) | Percentage (%) | Number (n) | Percentage (%) | Number (n) | Percentage (%) | Number (n) | Percentage (%) | ||
| <5 | 5 | 83.33 | 14 | 77.78 | 3 | 50.00 | 22 | 73.33 | 0.000 |
| ≥5 | 1 | 16.67 | 4 | 22.22 | 3 | 50.00 | 8 | 26.67 | |
| Total | 6 | 100 | 18 | 100 | 6 | 100 | 30 | 100 | |
Table 4 shows that cats weighing < 5 kg accounted for a significantly higher proportion of HCM cases (73.33%) than cats > 5 kg (26.67%) (p < 0.01), indicating a clear association between body weight and HCM prevalence in this population. In cats < 5 kg, HCM was common in all stages: 83.33% in B1 (5/6), 77.78% in B2 (14/18), and 50.00% in C (3/6). In cats ≥ 5 kg, the proportion increased in stage C (50.00%) compared with earlier stages.
3.3. Frequency of clinical signs in cats with HCM
The results in Table 5 present the clinical signs of hypertrophic cardiomyopathy (HCM) across three disease stages. Statistical analysis showed that the distribution of clinical signs differed significantly among stages (p < 0.01. Tachycardia and/or gallop rhythm was the most common clinical sign, accounting for 34.67% of all recorded signs and appearing in all three stages. Respiratory signs, including tachypnea, dyspnea, cyanosis, and abdominal breathing, accounted for 33.33% and were observed in all three disease stages. Seizures were recorded in only one case (5.56%) and occurred in stage C.
Table 5. Frequency of clinical signs in cats with HCM
| Clinical sign | Stage B1 | Stage B2 | Stage C | Total | P-value | ||||
|---|---|---|---|---|---|---|---|---|---|
| Number (n) | Percentage (%) | Number (n) | Percentage (%) | Number (n) | Percentage (%) | Number (n) | Percentage (%) | ||
| Tachycardia / gallop rhythm |
4 | 28.57 | 17 | 39.53 | 5 | 27.78 | 26 | 34.67 | |
| Tachypnea / syncope / cyanosis / abdominal breathing |
4 | 28.57 | 15 | 34.89 | 6 | 33.33 | 25 | 33.33 | |
| Seizures | 0 | 0.00 | 0 | 0.00 | 1 | 5.56 | 1 | 1.33 | 0.000 |
| Lethargy / reduced activity / exercise intolerance |
4 | 28.57 | 9 | 20.93 | 4 | 22.22 | 17 | 22.67 | |
| Heart murmur | 2 | 14.29 | 2 | 4.65 | 2 | 11.11 | 6 | 8.00 | |
| Total | 14 | 100 | 43 | 100 | 18 | 100 | 75 | 100 | |
Lethargy, reduced activity, and exercise intolerance accounted for 22.67%, and were most frequently recorded in stage B1 (28.57%). Heart murmurs accounted for only 8.00% of all signs—a low but noteworthy proportion.
3.4. Classification of heart sounds by disease stage
Table 6 shows the distribution of two types of cardiac sounds detected during clinical auscultation in cats with hypertrophic cardiomyopathy (HCM) across three disease stages. The results showed that gallop rhythm had a high detection rate (80.00%), whereas heart murmurs accounted for only 20.00%. Gallop rhythm was the most frequently recorded cardiac sound in this study and was present in all three disease stages. The highest prevalence of gallop rhythm was observed in stage B2 (88.89%). In contrast, heart murmurs accounted for only 20.00% of all cases.
| Heart Sound | Stage B1 | Stage B2 | Stage C | Total | P-value | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Number (n) | Percentage (%) | Number (n) | Percentage (%) | Number (n) | Percentage (%) | Number (n) | Percentage (%) | ||||
| Heart murmur | 2 | 33.33 | 2 | 11.11 | 2 | 33.33 | 6 | 20.00 | 0.000 | ||
| Gallop rhythm | 4 | 66.67 | 16 | 88.89 | 4 | 66.67 | 24 | 80.00 | |||
| Total | 6 | 100 | 18 | 100 | 6 | 100 | 30 | 100 | |||
3.5. Radiographic and Echocardiographic Indices in Cats Across ACVIM Stages
Table 7. Radiographic VHS and echocardiographic indices in cats with HCM by ACVIM stages
| Stage | Statistic | Radiographic Index | Echocardiographic Indices | ||
|---|---|---|---|---|---|
| VHS | LA/Ao | LVPWd | IVSd | ||
| B1 | n | 6 | 6 | 6 | 6 |
| X̅ | 8.06b | 1.62 | 0.61 | 0.63 | |
| SD | 0.15 | 0.04 | 0.02 | 0.01 | |
| Min | 7.80 | 1.58 | 0.59 | 0.62 | |
| Max | 8.20 | 1.68 | 0.65 | 0.65 | |
| B2 | n | 18 | 18 | 18 | 18 |
| X̅ | 8.43b | 1.85 | 0.63 | 0.70 | |
| SD | 0.32 | 0.20 | 0.06 | 0.07 | |
| Min | 8.10 | 1.60 | 0.55 | 0.61 | |
| Max | 9.20 | 2.36 | 0.79 | 0.87 | |
| C | n | 6 | 6 | 6 | 6 |
| X̅ | 9.83b | 2.55 | 0.70 | 0.91 | |
| SD | 0.47 | 0.70 | 0.10 | 0.18 | |
| Min | 9.30 | 1.55 | 0.61 | 0.62 | |
| Max | 10.50 | 3.48 | 0.90 | 1.15 | |
| P | 0.000 | 0.000 | 0.052 | 0.000 | |
Table 7 shows that VHS increased progressively with disease stage, with mean values of 8.06 ± 0.15 in stage B1, 8.43 ± 0.32 in stage B2, and 9.83 ± 0.47 in stage C. This difference was highly statistically significant (p < 0.01), indicating a strong association between VHS and disease progression. Pathophysiologically, HCM is characterized by left ventricular myocardial hypertrophy accompanied by secondary left atrial enlargement. Although VHS does not directly reflect myocardial wall thickness, it allows assessment of overall cardiac silhouette size on thoracic radiographs. Therefore, VHS tends to increase more markedly in later stages, when left atrial enlargement plays a major role in altering cardiac silhouette morphology. In stage B1, VHS ranged from 7.80 to 8.20, close to the normal reference range in healthy cats. This is consistent with Sousa et al. (2025), who reported that thoracic radiography has low sensitivity for detecting early-stage HCM, because myocardial hypertrophy is not yet sufficient to cause obvious changes in cardiac silhouette. In stage B2, when hemodynamic alterations and cardiac remodeling become more evident, VHS increased slightly, in parallel with echocardiographic changes such as increased LA/Ao ratio and interventricular septal thickness. However, VHS may still not exceed clear diagnostic thresholds, highlighting its limitation in screening pre–heart failure HCM. In stage C, VHS increased markedly, with a mean of 9.83 and values up to 10.5 in some cases. This corresponds to the congestive heart failure stage, when severe left atrial enlargement, pulmonary edema, or pleural effusion markedly alter cardiac silhouette appearance. This finding is consistent with reports by Ironside et al. (2021), in which high VHS values are considered a typical radiographic sign of heart failure in cats with HCM.
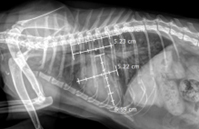
Fig. 2. Vertebral Heart Score (VHS) Measurement in a Cat with Congestive Heart Failure, Showing a VHS of 12
Regarding echocardiographic indices, statistically significant differences were observed for IVSd and LA/Ao, while LVPWd showed differences that did not reach statistical significance. For the LA/Ao ratio, a marked increase was observed across disease stages and was highly significant. This reflects the pathophysiology of HCM: left ventricular hypertrophy → reduced compliance → increased filling pressure → progressive left atrial enlargement over time. The LA/Ao value in stage C (2.55) greatly exceeded the reference threshold in the ACVIM consensus statement (2020), indicating chronically elevated filling pressures and high risk of pulmonary oedema and heart failure. This is consistent with Fuentes et al. (2020), who identified LA/Ao as the strongest predictor of congestive heart failure in HCM.
LVPWd but did not reach statistical significance (p = 0.052). This may be because more cats in the study exhibited an interventricular septal hypertrophy phenotype and were preferentially examined and diagnosed. According to Wess (2010), LVPWd is often not the predominant site of hypertrophy in early to moderate HCM. For IVSd, this index increased clearly across all three stages, and differences among stages were highly significant (p < 0.01). Specifically, from B1 to B2 there was a mild increase (0.63 to 0.70), and from B2 to C there was a marked increase (0.70 to 0.91). This indicates that interventricular septal hypertrophy was the dominant morphological pattern of HCM in this study population. This finding is consistent with Fuentes et al. (2020), who reported that IVSd > 0.9 cm is strongly associated with heart failure and arterial thromboembolism (ATE). A hallmark of HCM is asymmetric myocardial hypertrophy, most prominent in the interventricular septum. British Shorthair cats, in particular, tend to show a septal hypertrophy phenotype rather than predominant posterior wall thickening.
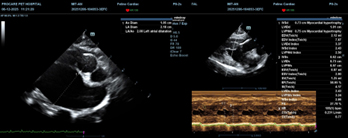
(A): LA/Ao = 2.08 (severe left atrial enlargement); (B): Interventricular septum and left ventricular free wall > 0.6 cm.
3.6. Treatment Outcomes of HCM According to Disease Stage
Table 8 shows a clear difference in treatment response rates across the stages of hypertrophic cardiomyopathy (HCM).
Table 8. Treatment outcomes of HCM by disease stage
| Stage | B1 | B2 | C | Total |
|---|---|---|---|---|
| Cases with successful outcome | 6 | 17 | 1 | 24 |
| Success rate (%) | 100 | 94.44 | 16.67 | 80.00 |
| Cases with poor outcome | 0 | 1 | 5 | 6 |
| Failure rate (%) | 0.00 | 5.56 | 83.33 | 20.00 |
| Total Cases | 6 | 18 | 6 | 30 |
In stage B1, myocardial hypertrophy is mild, and the results showed a 100% treatment response rate at this stage. In stage B2, myocardial hypertrophy is more pronounced as the treatment response rate remained high (94.44%). In contrast, treatment outcomes were poor in stage C, with a response rate of only 16.67% and an increase the failure rate (83.33%).
4.0 Discussion
The observation in Figure 1 based on clinical signs, cardiac murmurs, or suspicious findings during routine examination of suspected HCM cases presented to Procare Veterinary Hospital during the study period accounted for 7.61% of all cases, and is comparable to the study by Payne et al. (2015) on the prevalence of cats with clinical signs suggestive of heart disease in urban pet populations. They reported that approximately 8–10% of cats presented for examination had cardiac murmurs, but only 20–40% of these were truly affected by HCM when screened by echocardiography. Payne et al. (2015) also reported that not all cats with cardiac murmurs or suspicious signs actually have HCM. Cardiac murmurs may result from other causes such as hyperdynamic blood flow, stress, mild aortic stenosis, or extracardiac conditions (Fuentes et al., 2020). Therefore, the importance of echocardiography in establishing a definitive diagnosis of HCM is clearly demonstrated.
The marked differences in disease distribution among breeds observed in Table 1 where the British Shorthair cats accounted for the highest proportion of HCM is consistent with the findings of Granström et al. (2011), who reported that British Shorthairs had a prevalence of HCM comparable to or higher than Maine Coons in some clinical surveys. Notably, all three disease stages B1, B2, and C were all present in British Shorthair cats in this study, a finding that aligns with that of Fuentes et al. (2020), who suggested that British Shorthair cats tend to develop concentric left ventricular hypertrophy and may show faster disease progression than some other breeds. Our observation in the present study where Maine Coon and Bengal cats accounted for only 6.67% of total HCM cases and were recorded exclusively in only stage B2, contrasts with the lower proportion reported by Granström et al. (2011) and Payne et al. (2013), who identified Maine Coon as one of the breeds at highest risk for HCM. This discrepancy may be related to the lower number of Maine Coon and Bengal cats presented to Procare Veterinary Hospital, differences in breed composition of the owned-cat population in Ho Chi Minh City and the lack of routine cardiac screening by owners. Trehiou-Sechi et al. (2012) reported that mixed-breed cats still carry a risk of developing HCM, although generally lower than that of purebred cats. However, in the present study, the proportion of HCM in mixed-breed cats may be explained by the fact that mixed-breed cats constitute a large proportion of the cat population in Vietnam, and many may carry phenotypic or genetic traits derived from high-risk breeds such as British Shorthair, Scottish Fold, or British Longhair.
The non-significant sex differences portrayed in Table 2 where male and female cats recorded a relatively even distribution conform with previous studies suggesting that although early structural changes (e.g., ventricular wall thickness, left atrial size) become evident at this stage, but sex has not been identified as a decisive factor (Liew et al., 2017). This balanced distribution is consistent with other international studies like that of Paige (2009) which concluded that sex is not an independent risk factor for feline HCM. In terms of age, there was a clear association between HCM prevalence and age since cats younger than 6 years accounted for a significantly higher proportion compared with cats older than 6 years (Table 3). This is consistent with Kittleson et al. (2015) who reported that the average age of HCM onset in large studies ranges from 2–7 years and that first onset in older cats is uncommon. Whilst some breeds, such as Maine Coon and Ragdoll, may show early onset at 1–4 years, Fox et al. (2011) noted that HCM is not a disease specific to geriatric cats, but is more common in young and middle-aged cats, with 70–80% diagnosed before 7 years of age. The fact that cats < 6 years in this study were represented in all stages B1, B2, and C indicates that HCM can progress rapidly to congestive heart failure at a young age. The lack of routine cardiac screening in young cats may contribute to late detection, when clinical signs are already evident. These results confirm that cats under 6 years are a high-risk group for HCM and challenge the common perception that heart disease occurs mainly in older cats.
Body weight was a significant source of variation in HCM prevalence (Table 4) with cats weighing < 5 kg accounting for a significantly higher proportion of HCM cases (73.33%) than cats > 5 kg, indicating a clear association between body weight and HCM prevalence in this population. Most affected cats in this study were of low to medium body weight. This matches the population structure, in which breeds such as British Shorthair, Scottish Fold, and mixed-breed cats—typically weighing 3–5 kg—were predominant. This is consistent with Petrushko et al. (2022), who reported a higher prevalence of HCM in small- to medium-sized breeds compared with large breeds. The proportional increase in stage C compared with earlier stages in cats ≥ 5 kg may be related to overweight or obesity increasing afterload and myocardial workload, thereby promoting disease progression (Partington, 2024). The presence of large breeds such as Maine Coon may also have influenced this pattern. However, body weight is not considered a primary causal risk factor for HCM. The differences observed here likely reflect breed distribution and body conformation in the studied population rather than a direct pathogenic role of body weight in HCM.
The distribution of clinical signs (Table 5) differed significantly among stages, accurately reflecting the natural progression of HCM. The fact that tachycardia and/or gallop rhythm was the most common clinical sign in all three HCM stages can be explained by myocardial hypertrophy reducing left ventricular compliance, stimulating sympathetic activity and leading to a compensatory increase in heart rate (Gersh et al. 2011). On cardiac auscultation, the presence of S3/S4 gallop sounds is often associated with impaired left ventricular filling (Liew et al. 2017). This finding is consistent with Gaia de Sousa et al. (2025) who reported tachycardia as one of the most common clinical signs in cats with HCM, particularly in stage B2. The high frequency in stage B2 observed in this study is consistent with the characteristics of this stage, in which structural cardiac changes are evident, but congestive heart failure has not yet developed (Autore & Musumeci, 2020). Respiratory signs, including tachypnea, dyspnea, cyanosis, and abdominal breathing, were observed in all three disease stages (Table 5) indicating an increased left ventricular filling pressure and pulmonary congestion; even in stage B1, cats with HCM may already show respiratory abnormalities (Han & Jung, 2019). In stage C, abdominal breathing and severe dyspnea are often associated with pulmonary oedema or pleural effusion, which are typical signs of advanced left-sided congestive heart failure (Gersh et al. 2011). Although rare, the one case of seizure observed in Stage C is a clinically important sign that is often associated with severe complications such as arterial thromboembolism, reduced cerebral perfusion, or life-threatening arrhythmias as described by Hogan (2017) in cats with late-stage HCM. Lethargy, reduced activity, and exercise intolerance that were most frequently recorded in stage B1 are early manifestations of reduced peripheral tissue perfusion and impaired left ventricular filling, leading to fatigue and decreased exercise capacity (Fox et al. 2011). Fox et al. (2011) also noted that cats with HCM tend to reduce their activity early, even before obvious respiratory signs appear. The low proportion of heart murmurs is consistent with the study by Stern et al. (2016), which reported that approximately 58% of cats with HCM had no audible heart murmur on clinical examination, indicating that cardiac murmurs are not a sensitive screening sign for HCM in cats.
Gallop rhythm was the most frequently recorded cardiac sound in this study and was present in all three disease stages (Table 6). This finding is consistent with Fox et al. (2011), who stated that gallop rhythm is the most characteristic cardiac sound of feline HCM and has greater diagnostic value than heart murmurs. From a pathophysiological perspective, the gallop sound (S3 or S4) is generated when blood filling the left ventricle encounters increased resistance during diastole, which is a direct consequence of myocardial hypertrophy and reduced left ventricular compliance (Saponaro et al., 2023). In addition, Payne et al. (2015) reported that gallop rhythm is present in approximately 40–60% of cats with HCM, even in cases without heart murmurs or obvious clinical signs. The high prevalence of gallop rhythm in this study indicates that most cats already had significant diastolic dysfunction, consistent with the distribution of disease stages, particularly stages B2 and C (Efthimiadis et al., 2014). Efthimiadis et al., (2014) also reported that the pre–heart failure stage is the most favourable time to detect gallop rhythm, because hemodynamic changes have not yet been masked by pulmonary oedema or acute respiratory emergencies. This is consistent with the present findings, in which the highest prevalence of gallop rhythm was observed in stage B2 (Table 6), a stage characterised by marked myocardial hypertrophy without overt congestive heart failure. In contrast, heart murmurs accounted for only 20.00% of all cases (Table 6). This result is in contrast with the study by Paige et al. (2009), which reported that approximately 58% of cats with HCM had no audible heart murmur on clinical examination. Other studies have shown that heart murmurs are not a sensitive screening sign for feline HCM, especially in the preclinical stage (Payne et al., 2015; Fuentes et al., 2020). The detection of heart murmurs in some cases in stages B1 and C in this study may be explained by different mechanisms: In stage B1, murmurs may be related to stress-induced hyperdynamic cardiac activity during examination or mild systolic anterior motion (SAM) of the mitral valve; whereas in stage C, murmurs are more likely secondary to severe hemodynamic changes, functional mitral regurgitation, or turbulent flow associated with congestive heart failure (Liu et al., 1984).
Pathophysiologically, HCM is characterized by left ventricular myocardial hypertrophy accompanied by secondary left atrial enlargement and could explain the strong association between VHS and disease progression portrayed in Table 7. Although VHS does not directly reflect myocardial wall thickness, it allows assessment of overall cardiac silhouette size on thoracic radiographs. Therefore, VHS tends to increase more markedly in later stages, when left atrial enlargement plays a major role in altering cardiac silhouette morphology (Sousa et al., 2025). The observation in stage B1 where VHS ranged from 7.80 to 8.20 (Table 7), is close to the normal reference range in healthy cats, and consistent with Sousa et al. (2025), who reported that thoracic radiography has low sensitivity for detecting early-stage HCM, because myocardial hypertrophy is not yet sufficient to cause obvious changes in cardiac silhouette. In stage B2, when hemodynamic alterations and cardiac remodelling become more evident, VHS increased slightly, in parallel with echocardiographic changes such as increased LA/Ao ratio and interventricular septal thickness. However, VHS may still not exceed clear diagnostic thresholds, highlighting its limitation in screening pre–heart failure HCM. In stage C, VHS increased markedly, and corresponds to the congestive heart failure stage, when severe left atrial enlargement, pulmonary oedema, or pleural effusion markedly alter cardiac silhouette appearance. This finding is consistent with reports by Ironside et al. (2021), in which high VHS values are considered a typical radiographic sign of heart failure in cats with HCM. With regards to echocardiographic indices, there was a significantly marked increase in LA/Ao ratio (Figure 3), reflective of the pathophysiology of HCM: left ventricular hypertrophy → reduced compliance → increased filling pressure → progressive left atrial enlargement over time. The LA/Ao value in stage C exceeded the reference threshold in the ACVIM Consensus Statement (2020), indicating chronically elevated filling pressures and high risk of pulmonary oedema and heart failure. This is consistent with Fuentes et al. (2020), who identified LA/Ao as the strongest predictor of congestive heart failure in HCM.
According to Wess (2010), LVPWd is often not the predominant site of hypertrophy in early to moderate HCM. Fuentes et al. (2020) reported that IVSd > 0.9 cm is strongly associated with heart failure and arterial thromboembolism. A hallmark of HCM is asymmetric myocardial hypertrophy, most prominent in the interventricular septum. As depicted in Table 8, myocardial hypertrophy was mild in Stage B1, primarily because when the left atrium is not yet markedly enlarged, cardiac haemodynamics remain close to normal, and complications such as pulmonary oedema or pleural effusion have not yet developed. Therefore, lifestyle treatment strategies mainly focus on stress reduction, heart rate control, periodic monitoring, and dietary adjustment. The results showed a 100% treatment response rate at this stage, reflecting an excellent prognosis when the disease is detected early. However, in stage B2, myocardial hypertrophy is more pronounced and left atrial remodelling has begun, requiring pharmacological intervention to improve haemodynamics and slow disease progression. In this study, the treatment response rate in stage B2 remained high at 94.44% (Table 8), indicating that management of HCM in the pre–heart failure stage is highly effective, especially when early screening and appropriate protocols are applied. This finding is consistent with Maron (2002), who described stage B2 as the “golden stage” for HCM treatment, with the primary goal of delaying the onset of congestive heart failure. In contrast, lifestyle treatment outcomes were poor in stage C, with a response rate of only 16.67% (Table 8). At this stage, severe interventricular septal hypertrophy and marked left atrial enlargement are present, together with adverse complications such as arrhythmias, pulmonary oedema, pleural effusion, or thromboembolism. These factors markedly reduce treatment efficacy and increase the failure rate (83.33%). This result is consistent with Autore and Musumeci (2020), who reported that the one-year mortality rate in cats with HCM and congestive heart failure ranges from 50–70%, indicating a very guarded prognosis in advanced disease (Gersh et al. 2011).
5.0 Conclusion
This study concludes that treatment is most effective when the disease is detected in the early or pre–heart failure stages, supplements scientific data on feline cardiovascular disease, supports early screening and the development of appropriate follow-up strategies in clinical veterinary practice in Vietnam. Based on the study findings, a key recommendation for future application by clinicians dealing with this condition in their practices is that echocardiography is essential for definitive diagnosis of HCM because clinical signs and cardiac auscultation findings have limited diagnostic specificity.
Author Contributions: Conceptualisation: Xuan Thanh Nguyen, Loan Vu Thuy Hong Nguyen; Methodology: Xuan Thanh Nguyen; Validation: Kinh Van La, Loan Vu Thuy Hong Nguyen; Formal analysis, investigation: Xuan Thanh Nguyen, Kinh Van La, Loan Vu Thuy Hong Nguyen; Data Curation: Xuan Thanh Nguyen; Writing—Original Draft Preparation: Xuan Thanh Nguyen; Writing—Review and Editing: Kinh Van La, Loan Vu Thuy Hong Nguyen. All authors have read and agreed to the published version of the manuscript.
Funding: This research received no external funding.
Ethics Approval Statement: All research procedures in this case study were conducted in compliance with Article 72 of the Law on Livestock Production (Law 32/2018/QH14) regarding humane treatment and animal welfare and followed the National Regulation (TCVN 12448:2018) on animal welfare management.
Acknowledgments: The authors sincerely thank Procare Veterinary Hospital for providing the clinical cases presented in this report.
Conflicts of Interest: The authors declare no conflicts of interest.
Artificial Intelligence: AI was not used for this original research article.
References
Autore C, Musumeci MB. 2020. The natural history of hypertrophic cardiomyopathy. Journal of the European Society of Cardiology, 22(Suppl L), L11–L14. https://doi.org/10.1093/eurheartj/suaa125
Efthimiadis GK, Pagourelias ED, Gossios T, Zegkos T. 2014. Hypertrophic cardiomyopathy in 2013: Current speculations and future perspectives. World Journal of Cardiology, 6(2), 26–37. https://doi.org/10.4330/wjc.v6.i2.26
Fox PR, Rush JE, Reynolds CA, Defrancesco TC, Keene BW, Atkins CE, Gordon SG, Schober KE, Bonagura JD, Stepien RL, Kellihan HB, Macdonald KA, Lehmkuhl LB, Nguyenba TP, Sydney-Moise N, Lefbom BK, Hogan DF, Oyama MA. 2011. Multicenter evaluation of plasma N-terminal probrain natriuretic peptide (NT-pro BNP) as a biochemical screening test for asymptomatic (occult) cardiomyopathy in cats. Journal of Veterinary Internal Medicine, 25(5), 1010–1016. https://doi.org/10.1111/j.1939-1676.2011.00776.x
Gaia de Sousa F, Mendes ACR, Carvalho LP, Beier SL. 2025. Clinical-diagnostic and therapeutic advances in feline hypertrophic cardiomyopathy. Veterinary Sciences, 12(3), 289. https://doi.org/10.3390/vetsci12030289
Gersh BJ, Maron BJ, Bonow RO, Dearani JA, Fifer MA, Link MS, Naidu SS, Nishimura RA, Ommen SR, Rakowski H, Seidman CE, Towbin JA, Udelson JE, Yancy CW 2011. The American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines 2011. 2011 ACCF/AHA Guidelines for the Diagnosis and Treatment of Hypertrophic Cardiomyopathy: A Report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Developed in collaboration with the American Association for Thoracic Surgery, American Society of Echocardiography, American Society of Nuclear Cardiology, Heart Failure Society of America, Heart Rhythm Society, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. Journal of the American College of Cardiology, 58(25), e212–e260. https://doi.org/10.1016/j.jacc.2011.06.011
Granström S, Godiksen MT, Christiansen M, Pipper CB, Willesen JL, Koch J. 2011. Prevalence of hypertrophic cardiomyopathy in a cohort of British Shorthair cats in Denmark. Journal of Veterinary Internal Medicine, 25(4), 866–871. https://doi.org/10.1111/j.1939-1676.2011.0751.x
Han D, Jung DI. 2019. A comprehensive review of hypertrophic cardiomyopathy and arterial thromboembolism in cats. Journal of Biomedical and Translational Research, 20(4), 82–90. https://doi.org/10.12729/jbtr.2019.20.4.082
Hogan DF. 2017. Feline cardiogenic arterial thromboembolism: Prevention and therapy. Small Animal Practice, 47(5), 1065–1082. https://doi.org/10.1016/j.cvsm.2017.05.001
Ironside VA, Tricklebank PR, Boswood A. 2021. Risk indictors in cats with preclinical hypertrophic cardiomyopathy: A prospective cohort study. Journal of Feline Medicine and Surgery, 23(2), 149–159. https://doi.org/10.1177/1098612X20938651
Kittleson MD, Meurs KM, Harris SP. 2015. The genetic basis of hypertrophic cardiomyopathy in cats and humans. Journal of Veterinary Cardiology, 17 Suppl 1(Suppl 1), S53–S73. https://doi.org/10.1016/j.jvc.2015.03.001
Liew AC, Vassiliou VS, Cooper R, Raphael CE. 2017. Hypertrophic cardiomyopathy – Past, present and future. Journal of Clinical Medicine, 6(12), 118. https://doi.org/10.3390/jcm6120118
Liu SK, Peterson ME, Fox PR. 1984. Hypertropic cardiomyopathy and hyperthyroidism in the cat. Journal of the American Veterinary Medical Association, 185(1), 52–57.
Luis Fuentes V, Abbott J, Chetboul V, Côté E, Fox PR, Häggström J, Kittleson MD, Schober K, Stern JA. 2020. ACVIM consensus statement guidelines for the classification, diagnosis, and management of cardiomyopathies in cats. Journal of Veterinary Internal Medicine, 34(3), 1062–1077. https://doi.org/10.1111/jvim.15745
Maron BJ. 2002. Hypertrophic cardiomyopathy: A systematic review. JAMA, 287(10), 1308–1320. https://doi.org/10.1001/jama.287.10.1308
Minitab. 2026. Minitab Statistical Software. PA, USA. https://www.minitab.com/en-us/products/minitab/
Paige CF, Abbott JA, Elvinger F, Pyle RL. 2009. Prevalence of cardiomyopathy in apparently healthy cats. Journal of the American Veterinary Medical Association, 234(11), 1398–1403. https://doi.org/10.2460/javma.234.11.1398
Partington C, Hodgkiss-Geere H, Woods GRT, Dukes-McEwan J, Flanagan J, Biourge V, German AJ. 2024. The effect of obesity and subsequent weight reduction on cardiac morphology and function in cats. BMC Veterinary Research, 20(1), 154. https://doi.org/10.1186/s12917-024-04011-0
Payne JR, Borgeat K, Connolly DJ, Boswood A, Dennis S, Wagner T, Menaut P, Maerz I, Evans D, Simons VE, Brodbelt DC, Luis Fuentes V. 2013. Prognostic indicators in cats with hypertrophic cardiomyopathy. Journal of Veterinary Internal Medicine, 27(6), 1427–1436. https://doi.org/10.1111/jvim.12215
Payne, J. R., Brodbelt, D. C., & Luis Fuentes, V. 2015. Cardiomyopathy prevalence in 780 apparently healthy cats in rehoming centres (the CatScan study). Journal of veterinary cardiology : the official journal of the European Society of Veterinary Cardiology, 17 Suppl 1, S244–S257. https://doi.org/10.1016/j.jvc.2015.03.008
Petrushko A, Grushanska N. 2022. Prevalence of feline cardiomyopathy phenotypes and arterial thromboembolism. ScienceRise: Biological Science, 4 (33), 35–43. doi: http://doi.org/10.15587/2519-8025.2022.271011
Santiago SL, Freeman LM, Rush JE. 2020. Cardiac cachexia in cats with congestive heart failure: Prevalence and clinical, laboratory, and survival findings. Journal of Veterinary Internal Medicine, 34(1), 35–44. https://doi.org/10.1111/jvim.15672
Saponaro V, Mey C, Vonfeld I, Chamagne A, Alvarado MP, Cadoré JL, Chetboul V, Desquilbet, L. 2023. Systolic third sound associated with systolic anterior motion of the mitral valve in cats with obstructive hypertrophic cardiomyopathy. Journal of veterinary internal medicine, 37(5), 1679–1684. https://doi.org/10.1111/jvim.16806
Sukumolanan P, Petchdee S. 2022. Prevalence of cardiac myosin-binding protein C3 mutations in Maine Coon cats with hypertrophic cardiomyopathy. Veterinary World, 15(2), 502–508. https://doi.org/10.14202/vetworld.2022.502-508
Trehiou-Sechi E, Tissier R, Gouni V, Misbach C, Petit AM, Balouka D, Sampedrano CC, Castaignet M, Pouchelon JL, Chetboul V. 2012. Comparative echocardiographic and clinical features of hypertrophic cardiomyopathy in 5 breeds of cats: a retrospective analysis of 344 cases (2001-2011). Journal of Veterinary Internal Medicine, 26(3), 532–541. https://doi.org/10.1111/j.1939-1676.2012.00906.x
Wess G, Sarkar R, Hartmann K. 2010. Assessment of left ventricular systolic function by strain imaging echocardiography in various stages of feline hypertrophic cardiomyopathy. Journal of Veterinary Internal Medicine, 24(6), 1375–1382. https://doi.org/10.1111/j.1939-1676.2010.0586.x
Disclaimer/Publisher’s Note: The statements, opinions, institutional affiliations, data contained in all publications, and all responsibilities for accuracy are solely those of the individual author(s) and contributor(s) and not of MARCIAS AUSTRALIA and AJAVAS/or the Editor(s). MARCIAS AUSTRALIA and AJAVAS/or the Editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.

