Morphological traits, reproductive performance, and egg quality of Ri, Dong Tao, and reciprocal F1 crossbred hens
Bui Thi Dieu Mai1 Do Vo Anh Khoa1 Hoang Thi Nguyet2 Pham Doan Lan2 Nguyen Thi Dieu Thuy3 Dinh Thi Ngoc Thuy3*
- Vietnam National University of Forestry, Dongnai, Vietnam
- Institute of Animal Science, Thuy Phuong, Hanoi, Vietnam
- Institute of Biology, Vietnam Academy of Science and Technology, Hanoi, Vietnam
Article Information
- Date Received: 09/01/2026
- Date Revised: 12/04/2026
- Date Accepted: 26/04/2026
- Date Published Online: 27/04/2026
Copyright: © 2026 The Authors. Published by MARCIAS AUSTRALIA, 32 Champion Drive, Rosslea, Queensland 4812, Australia. This is an open access publication under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Citation: Bui TDM, Do VAK, Hoang TN, Pham DL, Nguyen TDT, Dinh TNT (2026). Morphological traits, reproductive performance, and egg quality of Dong Tao, Ri and reciprocal F1 crossbred hens. Aust J Agric Vet Anim Sci (AJAVAS), 1(3), 100010
https://doi.org/10.64902/ajavas.2025.100010
Abstract
Dong Tao and Ri chickens are indigenous Vietnamese breeds that differ markedly in morphology and productive performance. This study evaluated morphological traits, reproductive performance, egg quality, and hatchability of Ri, Dong Tao, and their reciprocal F1 crossbred hens in order to assess genetic effects and crossbreeding potential. A total of 53 laying hens were raised under identical management conditions. Body weight and shank traits were measured at 24 weeks of age, while egg traits were monitored over a 19-24-week laying period. Dong Tao hens exhibited significantly greater body weight and shank dimensions (P<0.05), but showed delayed sexual maturity and lower egg production compared to Ri hens. In contrast, Ri hens reached sexual maturity earlier and achieved a higher laying rate and hatchability. Reciprocal F1 hens displayed predominantly intermediate morphological and egg quality traits, indicating strong additive genetic effects, while showing improved reproductive performance relative to Dong Tao hens. Partial heterosis was observed for age at first egg and egg production, with limited reciprocal effects. Overall, reciprocal crossbreeding between Ri and Dong Tao chickens mitigated the trade-off between body size and reproductive efficiency while maintaining acceptable egg quality and hatchability, thus supporting the sustainable utilisation of indigenous chicken genetic resources.
Keywords:
Dong Tao, Ri, reciprocal crossbreeding, leg morphology, egg production, bodyweight
Highlights
- Crossbreeding contributed to a more balanced trade-off between body size and reproductive efficiency
- Predominantly additive genetic effects resulted in intermediate morphological and egg quality traits in reciprocal F1crossbreds
- Partial heterosis was evident for AFE and EP, with limited reciprocal effects, suggesting a minor role of maternal inheritance
1.0 Introduction
Native chicken breeds play a vital role in food security and culture due to their high adaptability, low nutritional requirements (Dahloum et al., 2016), and diverse genetic value (Berthouly et al., 2009; Weigend & Romanov, 2001); hence, the exploitation of native chicken genetic resources for sustainable livestock development has been implemented in many countries. Local chicken breeds are also highly valued for their ease of rearing, disease resistance, and preferred egg and meat products (Berthouly-Salazar et al., 2010). Vietnam currently has 28 native chicken breeds and is considered as one of the countries with the highest chicken genetic diversities in the world (FAO, 2008). Dong Tao (DT) chickens are an ancient native breed from Northern Vietnam, known for their large body size and legs, but limited egg production performance (Lan Phuong et al., 2015). Their large, thick, and dark red shanks and layered horn scales stand out. In contrast, Ri (RI) native chickens are characterised by a small body size and slimmer shanks with high adaptability, disease resistance, and good egg laying capacity, and are widely raised in various regions (Mui & Dang, 2016). To overcome the disadvantages of native chickens, crossbreeding is a common strategy in poultry farming, implemented through mating individuals with different genetics to produce F₁ offspring capable of expressing heterosis, and improved quality traits compared to purebred native chickens (Choo et al., 2014). This is not only an effective technical solution for improving productivity, but also an important strategy in poultry breeding (Benabdeljelil & Arfaoui, 2001). Forward and reverse crossbreeding between these two native chicken breeds (DT and RI) with different phenotypes to create F1 and F2 crossbred chickens for genetic correlation analysis was the primary aim of this research. To achieve this objective, the present study evaluated morphological characteristics, reproductive performance and egg quality traits of hens. The findings provide a scientific basis for breeding and selection programs, improving economic efficiency and sustainable development of indigenous genetic resources using local chicken breeds.
2.0 Materials and methods
Animals: A total of 53 adult laying hens, including Dong Tao (n = 10), Ri (n = 10), F1-R crossbreds produced from ♂Dong Tao × ♀R1 (n = 15), and F1-D crossbreds derived from ♂RI × ♀Dong Tao (n = 18) under the same management system where the hens were kept in individual cages and sires brought in for mating were used in this study.
Management: The experiment was conducted at the Lien Ninh Experimental Research Station, Hanoi, Viet Nam (20.90275794947994° N, 105.84832816780413° E). Each hen was kept in a separate cage with independent feeders and waterers throughout the experiment. All chickens were given ad libitum access to water and feed with a nutritional diet containing 2.750 metabolizable energy (Kcal/kg), 16% crude protein, 3.8% ether extract, 5% crude fibre, 4% calcium, and 0.5% phosphorus. Additionally, the BLS-Pro DT multi-strain probiotic (Bacillus subtilis 3×106 CFU, Lactobacillus plantarum 3×106 CFU, and Saccharomyces cerevisiae 3×106 CFU) was added to drinking water at a concentration of 4%. Artificial insemination was performed according to the procedure of Doanh et al., (2023)
Parameters: (i) body weight (BW, kg), big and small shank diameters (BSD and SSD, cm), shank circumference (SC, cm), shank length (SL, cm) of all hens were measured at 24 weeks of age in accordance with FAO (2008) guidelines, (ii) age at first egg (AFE, days), (iii) egg production over 19 or 24 weeks of laying since the first egg was laid for Ri or others, respectively (EP, eggs/hen), average laying rate (ALR, %) were recorded, (iv) Egg weight (EW, g), big and small diameters of egg (BDE and SDE, cm), egg shape index (ESI), hatched eggs/incubated or embryonated eggs ratio (IER or EER, %) were calculated as described by Doan et al. (2011). Accordingly, 100 eggs (on average, 5-10 eggs per week) from each breed group were randomly collected from the 5th week after the first egg laying until the end of the study at 19 or 24 weeks of laying for Ri chickens or other breeds, respectively.
Statistical analysis: Data were recorded using Excel software. Differences among genetic groups for productive and reproductive traits were analysed fitting the fixed effect of breed using One-way Analysis of Variance (ANOVA) in Minitab software version 16.0 (2016). Mean comparisons were performed using Tukey test with statistical significance declared at P <0.05.
3.0 Results
3.1. Body weight and shank morphological traits
The results in Table 1 and Figure 1 indicate that significant differences in BW and shank morphological traits were observed among the experimental chicken groups (P<0.05). Dong Tao hens exhibited the highest body weight (2.63 kg), which was significantly greater than that of Ri and both F1 crossbred groups. In contrast, Ri hens showed the lowest body weight (1.38 kg), whereas F1-D and F1-R hens displayed intermediate values, with no significant difference between the two groups. Similar patterns were observed for shank morphological traits. Dong Tao hens showed markedly larger shank dimensions, including BSD and SSD, SC, and SL, compared with all other groups (P < 0.05). Ri hens consistently exhibited the smallest shank measurements across all traits. In terms of shank phenotype (shank diameter, shank circumference, shank color and morphology of horn scales), both F1 crossbred groups exhibited intermediate traits related to the parental breeds. However, it tended to be similar to those of the Ri hens. Regarding F1-D and F1-R hens, there was no significant difference for BSD and SSD or SC (P>0.05). However, SL differed between the two crossbred groups, with F1-D hens showing longer shanks (9.00 cm) than F1-R hens (7.94 cm) and values comparable to those observed in Ri hens.
Table 1. Body weight and shank morphological traits
| Traits | Ri | Dong Tao | F1-D | F1-R |
|---|---|---|---|---|
| BW (kg) | 1,38c ± 0,95 | 2,63a ± 0,24 | 1,67b ± 0,29 | 1,75b ± 0,18 |
| BSD (cm) | 1,28c ± 0,14 | 3,23a ± 0,40 | 1,61b ± 0,05 | 1,61b ± 0,12 |
| SSD (cm) | 1,06c ± 0,26 | 2,58a ± 0,19 | 1,34b ± 0,08 | 1,33b ± 0,13 |
| SC (cm) | 4,64c ± 0,61 | 12,22a ± 0,97 | 5,65b ± 0,35 | 5,72b ± 0,79 |
| SL (cm) | 8,04c ± 0,67 | 13,35a ± 1,39 | 9,00b ± 0,65 | 7,94c ± 0,75 |
BW: Body weight; BSD: Big shank diameter; SSD: Small shank diameter; SC: Shank circumference (cm). Values are presented as mean ± standard deviation. Different superscript letters (a-c) within the same row indicate statistically significant differences among groups (P<0.05).
 |
 |
 |
 |
| (a) | (b) | (c) | (d) |
Figure 1. Shank morphology and shank size (a): Ri hens; (b): Dong Tao hens; (c): F1-R hens; (d): F1-D hens
3.2. Egg traits
EP performance differed among the chicken groups (Table 2). Dong Tao hens reached sexual maturity significantly later than Ri and both F1 crossbred groups (P < 0.05), as reflected by a higher age at first egg (163.0 days). In contrast, Ri hens and F1-D hens exhibited earlier onset of laying, with no significant difference observed among these groups, while F1-R hens showed an intermediate age at first egg. The ALR per hen varied markedly among groups, with Ri hens showing the highest laying rate (43.76%), followed by F1-R and F1-D hens. Dong Tao hens exhibited the lowest ALR (21.67%). Total EP per Ri hen (over the 19-week laying period) or Dong Tao hen (over the 24-week laying period) followed a similar trend, with F1-D hens producing the highest number of eggs per hen (60.2 eggs), comparable to Ri hens (58.7 eggs), whereas Dong Tao hens showed the lowest egg output (36.4 eggs).
Table 2. Egg production performance
| Traits | Ri | Dong Tao | F1-D | F1-R |
|---|---|---|---|---|
| AFE (days) | 139.0b ± 5.21 | 163.0a ± 4.65 | 135.8b ± 5.89 | 143.8b ± 3.83 |
| ALR/hen (%) | 43.76a ± 13.79 (*) | 21.67c ± 5.88 | 35.85b ± 12.09 | 32.50b ± 6.52 |
| EP/hen (eggs/hen) | 58.7a (*) | 36.4c | 60.2a | 54.6b |
AFE: Age at first egg; ALR: Average laying rate; EP: Egg production. Values are presented as mean ± standard deviation. Different superscript letters (a-c) within the same row indicate statistically significant differences among groups (P<0.05). (*) Monitoring for 19 weeks of egg laying.
The results in Table 3 indicate that (i) there was statistically significant differences in EW among the four groups of chickens (P<0.05) in which EW ranged from 43.73 to 45.99g, with Dong Tao hens producing significantly heavier eggs than F1-D hens, while eggs from Ri and F1-R hens showed intermediate weights and did not differ significantly from either group; (ii) Significant differences were also observed in egg dimensions. Eggs from Dong Tao, F1-D, and F1-R hens exhibited greater egg length compared with those from Ri hens (P<0.05). Egg width varied among groups, with F1-D hens producing the widest eggs, followed by Dong Tao and F1-R hens, whereas Ri hens exhibited the smallest egg width (P<0.05); (iii) ESI differed significantly among groups (P<0.05). Eggs from F1-R hens showed the highest shape index, while those from Ri hens exhibited the lowest values. Eggs from Dong Tao and F1-D hens displayed intermediate shape index values and did not differ significantly from either group; and (iv) Hatchability traits varied across chicken groups. The ratio of hatched eggs to incubated eggs ranged from 72.89% in Dong Tao hens to 89.20% in Ri hens, whereas the ratio of hatched eggs to embryonated eggs ranged from 87.03% to 94.70%. Statistical comparisons were
not performed for hatchability parameters due to the absence of replicate-level variation.
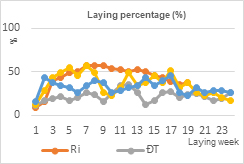 |
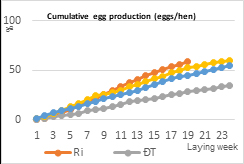 |
| (a) | (b) |
Figure 2. Laying performance (a) Laying percentage (%) (b) Cumulative egg production (eggs/hen)
Table 3. Egg quality and hatchability traits
| Traits | Ri | Dong Tao | F1-D | F1-R |
|---|---|---|---|---|
| EW (g) | 44.65ab ± 1.43 | 45.99a ± 3.30 | 43.73b ± 3.86 | 44.50ab ± 5.57 |
| BDE (cm) | 4.52b ± 0.27 | 4.96a ± 0.32 | 5.04a ± 0.18 | 5.09a ± 0.44 |
| SDE (cm) | 3.62c ± 0.41 | 3.84ab ± 0.13 | 3.99b ± 0.30 | 3.86a ± 0.20 |
| ESI | 1.26b ± 0.17 | 1.29ab ± 0.10 | 1.27ab ± 0.11 | 1.32a ± 0.13 |
| IER (%) | 89.2 | 72.89 | 78.45 | 73.61 |
| EER (%) | 94.7 | 87.03 | 90.61 | 88.83 |
EW: Egg weight; BDE: Big diameter of egg; SDE: Small diameter of egg; ESI: Egg shape index; IER: Hatched eggs/incubated eggs ratio; EER: Hatched eggs/embryonated eggs ratio. Values are presented as mean ± standard deviation. Different superscript letters (a-c) within the same row indicate statistically significant differences among groups (P < 0.05). Hatchability traits are expressed as percentages; no statistical comparison was performed for these parameters.
4.0 Discussion
The present study provides an integrated evaluation of morphological traits, egg production performance, egg quality, and hatchability in Ri, Dong Tao, and their reciprocal F1 crossbred hens. The results highlight strong breed-specific genetic effects, clear crossbreeding responses, and biologically meaningful relationships among body conformation, reproductive output, and egg characteristics. Adult Dong Tao and Ri hens (24 weeks old) have body weight indicators consistent with previous studies on their own breed characteristics, with body weights of approximately 1.33 kg for Ri hens (Tham et al., 2016) and 2.41 kg for Dong Tao hens (Mui & Dang, 2016) at the same age. Pronounced differences in BW and shank morphology between Dong Tao and Ri hens support the purpose of this study to produce crossbreeds for further genetic analysis. Dong Tao hens exhibited markedly greater BW and enlarged shank dimensions, traits that are characteristic of this breed and are largely governed by polygenic control of skeletal growth and ossification processes (Tatsuda & Fujinaka, 2001).
Generally, shank morphology and shank dimensions in both groups of F1 crossbreeds were expressed at an intermediate traits of parental breeds, the data showed that these traits were significantly lower than in Dong Tao hens and tended to be closer to the phenotype of Ri chickens. The absence of extreme phenotypes or transgressive segregation suggests limited dominance or epistatic effects for these traits, a pattern commonly reported for skeletal characteristics in chickens. Although most shank traits did not differ significantly between F1-D and F1-R hens, differences in shank length suggest a reciprocal cross effect. Such asymmetry may reflect maternal influences, including egg size and early embryonic development, or sex-linked genetic effects. Also, previous studies confirmed that maternal influences affect egg composition, particularly impacting testosterone levels in the egg yolk and the behavior of hatched chicks (Deeb & Lamont, 2002; Gong et al., 2023; Widowski et al., 2022). Similar reciprocal effects on skeletal traits as well as egg performance have been reported in indigenous chicken populations, emphasizing the importance of cross-direction in breeding program design (Ni et al., 2023; Tian et al., 2025).
EP results revealed a clear trade-off between body size and reproductive output. Dong Tao hens, characterized by greater BW and skeletal development, reached sexual maturity later and exhibited the lowest laying rate and total egg production. This inverse relationship between growth-related traits and reproductive performance has been widely documented and reflects antagonistic genetic correlations between somatic growth and ovarian function. Conversely, Ri hens reached sexual maturity earlier and exhibited superior laying performance, consistent with their genetic background as a light indigenous breed selected for reproductive efficiency. The F1 crossbred hens showed improved reproductive performance relative to Dong Tao hens, with earlier onset of lay and higher egg output, indicating partial heterosis for egg production traits, which are typically low to moderately heritable and responsive to crossbreeding (Norris & Ngambi, 2006). Egg quality traits were closely associated with hen body size and egg production intensity. Dong Tao hens produced larger eggs in terms of weight and dimensions, consistent with the positive correlation between body weight and egg size reported in previous studies. In contrast, Ri hens produced smaller eggs, a common feature of light indigenous breeds that allocate resources toward egg number rather than egg size. F1 crossbred hens produced eggs of intermediate size and shape, reflecting additive genetic inheritance and partial mitigation of the trade-off between egg number and egg size. The egg shape index was highest in F1-R hens, suggesting a more elongated egg form, which may be advantageous for shell strength and incubation efficiency (Narushin & Romanov, 2002).
Hatchability differed among groups, with Ri hens exhibiting the highest ratios of hatched eggs relative to incubated and embryonated eggs, whereas Dong Tao hens showed lower hatchability. Egg size and geometry are known to influence gas exchange, shell conductance, and thermal regulation during incubation, which may partially explain reduced hatchability in larger eggs (Mortola & Al Awam, 2010). F1 crossbred eggs showed intermediate hatchability values, suggesting additive genetic effects combined with maternal influences. Maternal regulation of egg size, yolk composition, and hormone deposition plays a critical role in embryonic survival and hatch success (Widowski et al., 2022). The relatively stable hatchability observed in F1 crossbreds indicates that crossbreeding did not negatively affect embryonic viability. When morphological traits, egg production, egg quality, and hatchability are considered collectively, F1 crossbred hens – particularly F1-R – demonstrated a more balanced overall performance compared with either parental breed. Dong Tao hens contribute desirable morphological traits and larger egg size, but exhibit reduced reproductive efficiency, whereas Ri hens excel in laying rate and hatchability but produce smaller eggs. Crossbreeding effectively combined complementary traits, partially alleviating biological trade-offs between growth and reproduction.
In summary, the F1 laying hens in this study clearly demonstrated heterosis by simultaneously improving two important indicators, which are body weight and leg size (inherited from DT chicken) and egg production capacity (the advantage of the Ri chicken). These results are entirely consistent with the general principle of heterosis effectiveness when crossing populations with different genetic backgrounds. This is also considered a strategy in breeding native chickens to improve productivity traits such as growth, reproduction, and hatchability (Chowdhury et al., 2014). These findings support the strategic use of reciprocal crossbreeding to improve reproductive performance while maintaining breed-specific characteristics in indigenous chicken populations. Future studies incorporating larger populations, multiple laying cycles, and molecular genetic approaches are recommended to further elucidate the genetic architecture of these traits and optimize sustainable breeding strategies.
Limitations of the study
The small sample size of this study was a major limitation; hence caution must be taken in interpreting the findings and generalising the outcomes. Individual feed intake of hens was not taken, hence feed efficiency by breed data were missed. Future studies with larger data size and individual hen intake are recommended.
5.0 Conclusion
Clear genetic differences were observed between Ri and Dong Tao chickens in BW and shank traits, reproductive performance, egg quality, and hatchability. Reciprocal F1 crossbred hens generally displayed intermediate morphological traits and egg quality, indicating predominantly additive genetic effects, while exhibiting improved reproductive performance compared with Dong Tao hens. Partial heterosis was evident for AFE and EP, with limited reciprocal effects, suggesting a minor role of maternal inheritance. Overall, crossbreeding contributed to a more balanced trade-off between body size and reproductive efficiency, highlighting its potential for improving productivity while conserving indigenous chicken genetic resources.
Author Contributions: Conceptualisation: Nguyen Thi Dieu Thuy, Do Vo Anh Khoa; Methodology: Do Vo Anh Khoa, Hoang Thi Nguyet; Experiments: Bui Thi Dieu Mai, Hoang Thi Nguyet, Dinh Thi Ngoc Thuy; Formal analysis: Bui Thi Dieu Mai; Writing—original draft preparation: Bui Thi Dieu Mai, Pham Doan Lan.; Writing—review and editing: Nguyen Thi Dieu Thuy, Do Vo Anh Khoa, Pham Doan Lan; Funding acquisition: Dinh Thi Ngoc Thuy. All authors have read and agreed to the published version of the manuscript.
Funding: This work was financially supported by Ministry of Science and Technology (MOST) under project grant NVQG.2021/DT.10.
Ethics Approval Statement: The study was conducted in accordance with the Research Ethics Approval by the Research Ethics Committee of Institute of Biology, Vietnam Academy of Science and Technology, Viet Nam (protocol code IB-AREC-2025-03, August 20, 2025).
Data Availability Statement: Data sharing is possible from the authors if a reasonable request is granted.
Conflicts of Interest: The authors declare no conflicts of interest.
References
Benabdeljelil K, Arfaoui T. 2001. Characterization of Beldi chicken and turkeys in rural poultry flocks of Morocco. Current state and future outlook. Animal Genetic Resources Information, 31, 87-95. https://doi.org/10.1017/S1014233900001516
Berthouly-Salazar C, Rognon X, Van T, Gély M, Chi CV, Tixier-Boichard M, Bed’Hom B, Bruneau N, Verrier E, Maillard JC, Michaux JR. 2010. Vietnamese chickens: A gate towards Asian genetic diversity. BMC Genetics, 11, 53. https://doi.org/10.1186/1471-2156-11-53
Berthouly C, Leroy G, Van TN, Thanh HH, Bed’Hom B, Nguyen BT, Vu CC, Monicat F, Tixier-Boichard M, Verrier E, Maillard JC, Rognon X. 2009. Genetic analysis of local Vietnamese chickens provides evidence of gene flow from wild to domestic populations. BMC Genetics, 10, 1. https://doi.org/10.1186/1471-2156-10-1
Choo YK, Kwon HJ, Oh ST, Um JS, Kim BG, Kang CW, Lee SK, An BK. 2014. Comparison of growth performance, carcass characteristics and meat quality of korean local chickens and silky fowl. Asian-Australasian Journal of Animal Sciences, 27(3), 398-405. https://doi.org/10.5713/ajas.2013.13638
Chowdhury V, Sultana H, Furuse M. 2014. International perspectives on impacts of reproductive technologies for world food production in Asia associated with poultry production. Advances in Experimental Medicine and Biology, 752, 229-237. https://doi.org/10.1007/978-1-4614-8887-3_12
Dahloum L, Moula N, Halbouche M, Grasteau S. 2016. Phenotypic characterization of the indigenous chickens (Gallus gallus) in the Northwest of Algeria. Archiv fur Tierzucht, 59. https://doi.org/10.5194/aab-59-79-2016
Deeb N, Lamont SJ. 2002. Genetic architecture of growth and body composition in unique chicken populations. Journal of Heredity, 93(2), 107-118. https://doi.org/10.1093/jhered/93.2.107
Doan BH, Mai NT, Son NT, Dat NH. 2011. Parameters and measurements used in poultry research. Agricultural Publishing House.
Doanh BH, Giang NTP, Yen ĐT, Giang NTC, PNT, Dang PK. 2023. Effects of collection frequency and duration of storage on semen quality of Hac Phong chickens. Vietnam Journal of Agricultural Sciences, 21(2), 161–168.
FAO. 2008. The state of the world’s animal genetic resources for food and agriculture (B. Rischkowsky, Ed. Pilling, Dafydd ed.). Food and Agriculture Organization of the United Nations. http://www.fao.org/3/a1250e/a1250e.pdf
Gong H, Wang T, Wu M, Chu Q, Lan H, Lang W, Zhu L, Song Y, Zhou Y, Wen Q, Yu J, Wang B, Zheng X. 2023. Maternal effects drive intestinal development beginning in the embryonic period on the basis of maternal immune and microbial transfer in chickens. Microbiome, 11(1), 41. https://doi.org/10.1186/s40168-023-01490-5
Lan Phuong TN, Dong Xuan KDT, Szalay I. 2015. Traditions and local use of native Vietnamese chicken breeds in sustainable rural farming. World’s Poultry Science Journal, 71(2), 385-396. https://doi.org/10.1017/S0043933915000380
Mortola JP, Al Awam K. 2010. Growth of the chicken embryo: implications of egg size. Comparative Biochemistry and Physiology A Molecular Integrated Physiology, 156(4), 373-379. https://doi.org/10.1016/j.cbpa.2010.03.011
Mui NB, Dang PK. 2016. Reproductive capacity of Ri and Crossbred chickens raised in An Duong District, Hai Phong City. Vietnam Journal of Agricultural Sciences, 14(3), 392-399.
Narushin V, Romanov M. 2002. Physical characteristics of chicken eggs in relation to their hatchability and chick weight in St. Joseph, Michigan. https://elibrary.asabe.org/abstract.asp?aid=9226&t=5
Ni A, Calus MPL, Bovenhuis H, Yuan J, Wang Y, Sun Y, Chen J. 2023. Genetic parameters, reciprocal cross differences, and age-related heterosis of egg-laying performance in chickens. Genetics Selection Evolution, 55(1), 87. https://doi.org/10.1186/s12711-023-00862-7
Norris D, Ngambi JW. 2006. Genetic parameter estimates for body weight in local Venda chickens. Tropical Animal Health and Production 38(7-8), 605-609. https://doi.org/10.1007/s11250-006-4420-6
Tatsuda K, Fujinaka K. 2001. Genetic mapping of the QTL affecting body weight in chickens using a F2 family. British Poultry Science, 42(3), 333-337. https://doi.org/10.1080/00071660120055296
Tham LT, Thai NX, Thang VV, Hiep ĐT, Soan ĐV, Ton VĐ, Binh ĐV. 2016. Growth, carcass yield and meat quality of Dong Tao chickens. Vietnam Journal of Agricultural Sciences, 14(11), 1716–1725.
Tian Y, Wang X, Yue Y, Arif M, Jiang Y, Liu Q, Du Y, Zhao X, Zhang F. 2025. Comparative study on growth performance and meat production traits of reciprocal crosses between Guizhou recessive white chickens and Qiandongnan Xiaoxiang chickens. Animals, 15. https://doi.org/10.3390/ani15223262
Weigend S, Romanov MN. 2001. Current strategies for the assessment and evaluation of genetic diversity in chicken resources. World’s Poultry Science Journal, 57(3), 275-288. https://doi.org/10.1079/WPS20010020
Widowski TM, Cooley L, Hendriksen S, Peixoto M. 2022. Maternal age and maternal environment affect egg composition, yolk testosterone, offspring growth and behaviour in laying hens. Scientific Reports, 12(1), 1828. https://doi.org/10.1038/s41598-022-05491-6.
Disclaimer/Publisher’s Note: The statements, opinions, institutional affiliations, data contained in all publications, and all responsibilities for accuracy are solely those of the individual author(s) and contributor(s) and not of MARCIAS AUSTRALIA and AJAVAS/or the Editor(s). MARCIAS AUSTRALIA and AJAVAS/or the Editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.

