Characteristics of canine kidney disease at Pet Health Centre Veterinary Hospitals
Hau Van Bui1 Thong Trung Ho1 Anh Thi Lan Trinh1 Loan Vu Thuy Hong Nguyen1*
- Faculty of Veterinary Medicine and Animal Science, HUTECH University, Binh Thanh District, Ho Chi Minh City 717000, Vietnam
Article Information
- Date Received: 17/01/2026
- Date Revised: 12/04/2026
- Date Accepted: 26/04/2026
- Date Published Online: 27/04/2026
Copyright: © 2026 The Authors. Published by MARCIAS AUSTRALIA, 32 Champion Drive, Rosslea, Queensland 4812, Australia. This is an open access publication under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Citation: Bui HV, Ho TT, Trinh ATL, Nguyen LVTH (2026). Characteristics of canine kidney disease at Pet Health Centre Veterinary Hospitals. Aust J Agric Vet Anim Sci (AJAVAS), 2(1), 100016 https://doi.org/10.64902/ajavas.2026.100016
Abstract
This study describes the clinical characteristics, paraclinical findings, ultrasonographic features, and treatment outcomes of canine renal diseases at Ho Chi Minh and Dong Nai Provincial network of Pet Health Centre Veterinary Hospitals from November 2024 to June 2025. Among 219 dogs presenting with suspected renal disease, 183 dogs were diagnosed, accounting for 83.56% of cases. Acute kidney disease was identified in 53.42% (117/219) of dogs, while chronic kidney disease accounted for 30.14% (66/219). Common clinical signs in acute and chronic cases included anorexia/lethargy (81.28%), vomiting/diarrhea (60.54%), oliguria or urinary retention (100%), and polyuria/polydipsia (100%). Haematological analysis revealed leukocytosis in 46.45%, anemia in 61.75%, and thrombocytopenia in 48.09% of affected dogs. Biochemical abnormalities showed elevated creatinine in 77.60% and increased blood urea nitrogen in 65.57% of cases. Urinalysis demonstrated proteinuria (82%), leukocyturia (64%), and hematuria (54%). Renal ultrasonography performed in 83 dogs revealed nephritis/pyelonephritis (39.76%), hydronephrosis (25.30%), and small kidneys (22.89%). The prevalence of renal disease was significantly higher in exotic breeds (91.06%), male dogs (93.08%), kennel-raised dogs (98.78%), and dogs fed commercial dry diets (91.49%). Treatment outcomes showed a recovery rate of 41.03% in acute kidney disease, while no complete recovery was observed in chronic kidney disease; mortality rates were 42.74% and 25.76%, respectively. These findings indicate that most dogs were diagnosed at advanced stages, highlighting the importance of early detection and routine renal monitoring in veterinary clinical practice.
Keywords
Canine kidney disease; acute kidney injury; chronic kidney disease; ultrasonography
Highlights
- Acute kidney disease was more prevalent than chronic kidney disease in dogs.
- Most dogs showed severe renal dysfunction at the time of diagnosis.
- Early detection is crucial to improve treatment outcomes.
1.0 Introduction
Renal diseases in dogs often progress silently, are difficult to detect at an early stage, but have a serious impact on overall health and lifespan. Goldston (1995) estimated that about 15% of adult dogs suffer from kidney-related diseases. In Vietnam, Nguyen (2006) reported that approximately 7.1% of dogs presented to veterinary clinics were diagnosed with kidney disease, indicating that this is a relatively common condition in clinical practice. Many domestic studies have described the typical clinical signs associated with canine renal disease. Tran et al. (2018) reported a high prevalence of urinary tract–related signs, including changes in urine color, dysuria, hematuria, and pyuria. In addition, clinical signs such as stranguria, anuria, urinary incontinence, and strong ammoniacal odor of urine were frequently observed, reflecting severe dysfunction of the urinary system. In cases of acute kidney disease, complete recovery can be achieved if the disease is detected early and treated with an appropriate therapeutic protocol. However, early diagnosis is often difficult because the clinical signs are nonspecific and easily confused with other diseases. Ettinger and Feldman (2017) noted that signs such as vomiting, lethargy, dysuria, and dehydration may occur in many different disorders, increasing the risk of delayed diagnosis; as a result, mortality rates may reach 30–50% if timely intervention is not provided (Cooper, 2013).
Acute kidney disease (AKD) has rapid onset over days, with sudden increases in creatinine and blood urinary nitrogen, vomiting, dehydration, oliguria or anuria, lethargy, and anorexia; early ultrasound may be normal, and early treatment can allow recovery (Bartges, 2012). Chronic kidney disease (CKD) progresses slowly over months to years, with vague early signs that worsen over time; ultrasound typically shows small, irregular kidneys with structural distortion, and the disease is irreversible, with treatment aimed at slowing progression. If not treated promptly and effectively, AKD may rapidly progress to chronic kidney disease, leading to irreversible renal damage and posing a direct threat to life. Bartges (2012) reported that in severe renal injury, dogs may die within only a few days to a few weeks without appropriate treatment.
Therefore, the primary objective of this study aimed at the early detection, accurate diagnosis, and timely treatment of AKD was to examine and describe the clinical characteristics, paraclinical findings, ultrasonographic features, and treatment outcomes of canine renal diseases at Ho Chi Minh and Dong Nai Provincial network of Pet Health Centre Veterinary Hospitals in Vietnam.
2.0 Materials and methods
2.1. Study site, animals, and presenting clinical signs
This study was conducted from November 2024 to June 2025 at the Pet Health Centre Veterinary Hospital System including: The Main Veterinary Hospital and the Veterinary Clinical Training Center in Ho Chi Minh City; Provincial Pet Health Centres in Dong Nai Province. Dogs presented to the clinics, regardless of breed, sex, or age, were included if they showed clinical signs suggestive of renal disease, such as lethargy, anorexia, vomiting, dyspnea, halitosis, oral ulceration, decreased or increased water intake, tachypnea, abdominal pain, polyuria, oliguria, or urinary retention.
2.2. Data Collection
2.2.1. Clinical examination
Dogs showing clinical signs indicative of renal disease were initially screened. Information collected from the dog owners included breed, sex, age, body weight, housing method, diet, and medical history. During examination, general condition, coat and skin, mucous membranes, respiratory rate, and behavior were observed. The kidney area was palpated to assess pain; skin turgor was checked to evaluate dehydration, and body temperature was measured to detect inflammatory signs. Based on these findings, dogs were classified as suspected renal cases for further investigations to complete the diagnosis These included blood tests, urinalysis, and ultrasonographic examinations were performed.
2.2.2. Paraclinical examination
Suspected dogs had about 3 mL of blood collected from the cephalicvein for biochemical and hematological tests, placed into heparin and EDTA tubes, labeled, and analyzed immediately or stored at 2–8°C for no more than 6 hours. Urine samples were collected as midstream urine during spontaneous urination; in small dogs, gentle bladder expression was applied. In cases of urinary retention, samples were obtained by urethral catheterization or cystocentesis. Urine was placed in sterile containers and analyzed using dipsticks and an automated analyzer within one minute. Ultrasonography was performed with dogs in dorsal recumbency on a V-table using a micro-convex probe (4.5–8 MHz) on a DW-360 machine. Kidneys and related organs (bladder, liver, spleen, intestines, uterus, prostate) were evaluated for renal margins, corticomedullary junction, cortex/medulla ratio, cortical and medullary thickness, echogenicity, and the presence of cysts, fibrosis, hemorrhage, pelvic dilation, or perirenal effusion as previously described (Perondi et al. 2020).
2.2.3. Differentiation between acute and chronic kidney disease
Differentiation was based on clinical signs, history, results of blood and urine tests, ultrasonographic findings, and treatment response (Bartges, 2012).
2.2.4. Treatment
Dogs were hospitalised or treated as outpatients during the first 5 days to stabilize and reduce clinical signs, then continued as outpatients with a standardised follow-up by phone every 1–2 weeks during the first month.
2.2.5 Treatment of acute kidney disease
Dehydration was corrected using isotonic fluids such as Lactated Ringer’s or 0.9% NaCl at 40–60 mL/kg/day IV or SC. If it was suspected that intoxication had occurred within 1 hour, emesis was induced via gastric tube with 3% hydrogen peroxide. For oliguria or dysuria, furosemide 0.5–1 mL/10 kg was given 4–3 times/day at 6–8 h intervals. In urinary retention, urethral catheterization was maintained for 5–7 days. Vomiting was treated with famotidine (0.5 mg/kg) or omeprazole (0.7–1 mg/kg) every 12–24 h by oral administration for 3–5 days. In cases of fever or infection, amoxicillin, ceftriaxone, or lincomycin at 1 mL/5 kg for 4–5 days were used. For haemoparasitosis, doxycycline 100 mg at 1 tablet/10 kg orally was given. Toxic agents were removed, and potentially nephrotoxic drugs previously administered to treat other underlying conditions were discontinued and replaced if applicable. Protocols were adapted from Bartges et al. (2012).
2.2.6. Treatment of chronic kidney disease
Dogs with small kidneys on ultrasound and high creatinine/BUN were switched to low-protein, low-phosphorus diets, avoiding salty food and commercial kibble, using bland home-cooked diets or prescription renal diets. In case of anemia diagnosed based on blood hematology, iron or erythropoietin at 2.2 mL/10 kg once daily was given. If urine pH was high, correction was done using sodium bicarbonate 8–12 mg/kg every 8–12 h or potassium citrate 40–60 mg/kg every 12 h. In end-stage CKD, peritoneal dialysis at 30–50 mL/kg was applied, with catheter removal after 48–72 h to prevent infection, following referenced guidelines.
2.3. Statistical analysis
Collected data were classified into defined groups and analysed using Minitab 17 software (Minitab 2026, PA, USA). Continuous variables were expressed as mean ± SE. For categorical variables, analysis of proportions was performed using the chi-square (χ²) test, and differences were considered statistically significant when P ≤ 0.05.
3.0 Results
3.1. Prevalence of kidney disease and its types in dogs
Results in Fig. 1 show that among 219 dogs with clinical signs suspicious for renal disease, 183 were confirmed to have kidney disease, accounting for 83.56%. This indicates that the clinical signs used for initial screening were reliable and that most dogs were presented for examination when renal dysfunction had already become relatively evident.
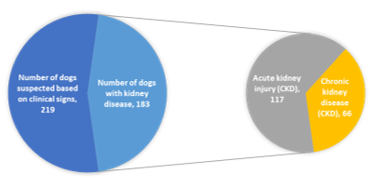
Fig. 1. Distribution of suspected and confirmed HCM cases among examined cats
Among dogs diagnosed with kidney disease, acute kidney disease was the most common, with 117/219 cases (53.42%), whereas chronic kidney disease accounted for 66/219 cases (30.14%).
3.2. Frequency of clinical signs
Among the 183 dogs diagnosed with kidney disease, clinical signs were recorded through physical examination and observation, and are presented in detail in Fig. 2.
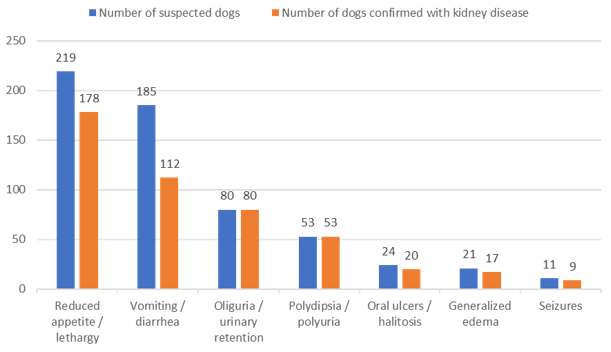
Fig. 2. Frequency of clinical signs in dogs with kidney disease (n = 183)
Fig. 2 shows that among 183 dogs with kidney disease, clinical signs appeared with different frequencies, reflecting the diversity and heterogeneity of the disease. Reduced appetite/lethargy was the most common sign (81.28%), indicating that this is a very frequent but nonspecific sign that can be confused with many other internal diseases. Vomiting/diarrhea accounted for 60.54% and occurred in both acute and chronic kidney disease. Oliguria and polydipsia/polyuria were recorded in 100% of cases, indicating that this is a highly valuable sign for diagnosis, especially in acute kidney disease. Severe signs such as oral ulcers/halitosis (83.33%), generalised oedema (80.95%), and seizures (81.82%) were usually observed in advanced stages.
3.3. Some haematological and biochemical parameters in dogs with kidney disease
Haematological examinations were performed in dogs with kidney disease, and the results are presented in Table 1.
Table 1. Selected haematological parameters in dogs with kidney disease
| Parameter (unit) Reference value* |
Observed range | Interpretation | Mean ± SE | Number of dogs (n = 183) | Percentage (%) |
|---|---|---|---|---|---|
| Leukocytes (×109/L) 6.0 – 17.0 |
0.1 – 5.5 | Low | 3.89 ± 0.72 | 27 | 14.75 |
| 6.0 – 17.7 | Normal | 15.46 ± 0.53 | 71 | 38.80 | |
| 17.1 – 100.1 | High | 66.57 ± 0.11 | 85 | 46.45 | |
| Erythrocytes (×1012/L) 5.5 – 8.5 |
1.5 – 5.4 | Low | 4.01 ± 0.36 | 113 | 61.75 |
| 5.5 – 8.5 | Normal | 5.81 ± 0.49 | 59 | 32.24 | |
| 8.6 – 24.1 | High | 19.06 ± 0.12 | 11 | 6.01 | |
| Haemoglobin (g/dL) 12.0 – 18.0 |
2.5 – 11.9 | Low | 8.08 ± 0.34 | 138 | 75.41 |
| 12.0 – 18.0 | Normal | 16.15 ± 0.65 | 45 | 24.59 | |
| Haematocrit (%) 33.0 – 56.0 |
9.3 – 29.3 | Low | 27.80 ± 0.12 | 77 | 42.08 |
| 33.0 – 56.0 | Normal | 44.63 ± 1.01 | 69 | 37.70 | |
| 56.1 – 61.7 | High | 59.04 ± 1.38 | 37 | 20.22 | |
| Platelets (×109/L) 200 – 500 |
189 – 199 | Low | 191.7 ± 3.23 | 88 | 48.09 |
| 200 – 500 | Normal | 384.9 ± 17.0 | 85 | 46.45 | |
| 501 – 565 | High | 555.0 ± 10.0 | 10 | 5.46 |
(*)Haematological reference values were taken from Ettinger and Feldman (2017).
Most cases showed an increase in leukocytes, with 46.45% of dogs presenting leukocytosis, 38.80% within the normal range, and 14.75% with leukopenia. About 61.75% of dogs had low red blood cell counts, only 32.24% were within the normal range, and 6.01% were increased, indicating that anaemia is very common, especially in chronic kidney disease. Haemoglobin was decreased in 75.41% of dogs, reflecting marked anaemia, which reduces oxygen-carrying capacity and contributes to clinical signs such as fatigue and lethargy. Haematocrit reduced, with 42.08% low, 37.70% normal, and 20.22% increased, further confirming prolonged anaemia, characteristic of chronic kidney disease. It was also evident that 48.09% of dogs had thrombocytopenia, 46.45% were within the normal range, and 5.46% showed thrombocytosis.
Table 2. Blood biochemical parameters in dogs with kidney disease
| Parameter (unit) Reference value* |
Observed range | Interpretation | Mean ± SE | Number of dogs (n = 183) | Percentage (%) |
|---|---|---|---|---|---|
| Leukocytes (×109/L) 6.0 – 17.0 |
0.1 – 5.5 | Low | 3.89 ± 0.72 | 27 | 14.75 |
| 6.0 – 17.7 | Normal | 15.46 ± 0.53 | 71 | 38.80 | |
| 17.1 – 100.1 | High | 66.57 ± 0.11 | 85 | 46.45 | |
| Erythrocytes (×1012/L) 5.5 – 8.5 |
1.5 – 5.4 | Low | 4.01 ± 0.36 | 113 | 61.75 |
| 5.5 – 8.5 | Normal | 5.81 ± 0.49 | 59 | 32.24 | |
| 8.6 – 24.1 | High | 19.06 ± 0.12 | 11 | 6.01 | |
| Haemoglobin (g/dL) 12.0 – 18.0 |
2.5 – 11.9 | Low | 8.08 ± 0.34 | 138 | 75.41 |
| 12.0 – 18.0 | Normal | 16.15 ± 0.65 | 45 | 24.59 | |
| Haematocrit (%) 33.0 – 56.0 |
9.3 – 29.3 | Low | 27.80 ± 0.12 | 77 | 42.08 |
| 33.0 – 56.0 | Normal | 44.63 ± 1.01 | 69 | 37.70 | |
| 56.1 – 61.7 | High | 59.04 ± 1.38 | 37 | 20.22 | |
| Platelets (×109/L) 200 – 500 |
189 – 199 | Low | 191.7 ± 3.23 | 88 | 48.09 |
| 200 – 500 | Normal | 384.9 ± 17.0 | 85 | 46.45 | |
| 501 – 565 | High | 555.0 ± 10.0 | 10 | 5.46 | |
| Creatinine (µmol/L) 44 – 132 |
44 – 132 | Normal | 122.54 ± 3.13 | 41 | 22.40 |
| 133 – 440 | High | 312.7 ± 8.15 | 20 | 10.93 | |
| 441 – 2000 | Severe | 1601.7 ± 2.10 | 122 | 66.67 | |
| BUN (mmol/L) 3.2 – 11.1 |
3.2 – 11.1 | Normal | 9.18 ± 0.11 | 63 | 34.43 |
| 11.2 – 35.7 | High | 32.58 ± 1.43 | 120 | 65.57 | |
| Ionized Calcium (mmol/L) 2.2 – 2.8 |
1.2 – 2.18 | Low | 1.22 ± 0.14 | 79 | 43.17 |
| 2.2 – 2.8 | Normal | 2.14 ± 0.06 | 56 | 30.60 | |
| 2.81 – 3.4 | High | 3.27 ± 0.03 | 48 | 26.23 | |
| Phosphorus (mmol/L) 0.8 – 2.2 |
0.8 – 2.2 | Normal | 1.69 ± 0.05 | 86 | 46.99 |
| 2.2 – 7 | High | 5.24 ± 0.38 | 97 | 53.01 |
(*)Blood biochemical reference values based on IRIS (2019) and Ettinger (2017).
Results in Table 2 show that blood biochemical parameters in dogs with kidney disease differed markedly from normal physiological reference values, reflecting impaired filtration function and metabolic disturbances caused by renal damage. Among these, creatinine and BUN showed the most pronounced changes, indicating that most cases were detected when renal function had already declined significantly. Elevated serum creatinine predominated, with 66.67% of dogs in the severe increase range (441–2000 µmol/L) and 10.93% in the moderate increase range (133–440 µmol/L), while only 22.40% remained within the normal range. BUN was elevated in 65.57% of dogs, whereas 34.43% were within the normal range, showing that accumulation of nitrogenous waste products in the blood was common.
It was observed that 43.17% of dogs had hypocalcaemia, 30.60% had normal calcium serum levels, and 26.23% had hypercalcemia. This variability reflects the complex nature of mineral disturbances in dogs with kidney disease. Serum phosphorus was elevated in 53.01% of dogs, while 46.99% were within the normal range, indicating reduced renal excretion of phosphorus.
3.4. Urinalysis parameters in dogs with kidney disease
Among the 183 dogs diagnosed with kidney disease, urine samples were obtained from 50 cases for biochemical analysis; the results are presented in Table 3. Urinary parameters showed marked variation, reflecting different degrees of impairment in renal filtration and urine-concentrating ability.
Table 3. Selected urinary biochemical parameters in dogs with kidney disease
| Parameter (unit) Reference value* |
Observed range | Interpretation | Mean ± SE | Number of dogs | Percentage (%) |
|---|---|---|---|---|---|
| Hematology Data (n = 183) [1] | |||||
| Leukocytes (×109/L) 6.0 – 17.0 |
0.1 – 5.5 | Low | 3.89 ± 0.72 | 27 | 14.75 |
| 6.0 – 17.7 | Normal | 15.46 ± 0.53 | 71 | 38.80 | |
| 17.1 – 100.1 | High | 66.57 ± 0.11 | 85 | 46.45 | |
| Erythrocytes (×1012/L) 5.5 – 8.5 |
1.5 – 5.4 | Low | 4.01 ± 0.36 | 113 | 61.75 |
| 5.5 – 8.5 | Normal | 5.81 ± 0.49 | 59 | 32.24 | |
| 8.6 – 24.1 | High | 19.06 ± 0.12 | 11 | 6.01 | |
| Haemoglobin (g/dL) 12.0 – 18.0 |
2.5 – 11.9 | Low | 8.08 ± 0.34 | 138 | 75.41 |
| 12.0 – 18.0 | Normal | 16.15 ± 0.65 | 45 | 24.59 | |
| Haematocrit (%) 33.0 – 56.0 |
9.3 – 29.3 | Low | 27.80 ± 0.12 | 77 | 42.08 |
| 33.0 – 56.0 | Normal | 44.63 ± 1.01 | 69 | 37.70 | |
| 56.1 – 61.7 | High | 59.04 ± 1.38 | 37 | 20.22 | |
| Platelets (×109/L) 200 – 500 |
189 – 199 | Low | 191.7 ± 3.23 | 88 | 48.09 |
| 200 – 500 | Normal | 384.9 ± 17.0 | 85 | 46.45 | |
| 501 – 565 | High | 555.0 ± 10.0 | 10 | 5.46 | |
| Biochemical Data (n = 183) [2] | |||||
| Creatinine (µmol/L) 44 – 132 |
44 – 132 | Normal | 122.54 ± 3.13 | 41 | 22.40 |
| 133 – 440 | High | 312.7 ± 8.15 | 20 | 10.93 | |
| 441 – 2000 | Severe | 1601.7 ± 2.10 | 122 | 66.67 | |
| BUN (mmol/L) 3.2 – 11.1 |
3.2 – 11.1 | Normal | 9.18 ± 0.11 | 63 | 34.43 |
| 11.2 – 35.7 | High | 32.58 ± 1.43 | 120 | 65.57 | |
| Ionized Calcium (mmol/L) 2.2 – 2.8 |
1.2 – 2.18 | Low | 1.22 ± 0.14 | 79 | 43.17 |
| 2.2 – 2.8 | Normal | 2.14 ± 0.06 | 56 | 30.60 | |
| 2.81 – 3.4 | High | 3.27 ± 0.03 | 48 | 26.23 | |
| Phosphorus (mmol/L) 0.8 – 2.2 |
0.8 – 2.2 | Normal | 1.69 ± 0.05 | 86 | 46.99 |
| 2.2 – 7 | High | 5.24 ± 0.38 | 97 | 53.01 | |
| Urinalysis Data (n = 50) [3] | |||||
| Specific gravity 1.015 – 1.045 |
1.000 – 1.014 | Low | 1.004 ± 0.08 | 16 | 32 |
| 1.015 – 1.045 | Normal | 1.023 ± 0.021 | 29 | 58 | |
| 1.045 – 1.052 | High | 1.050 ± 0.01 | 5 | 10 | |
| Urine pH 5.5 – 7.0 |
4.5 – 5.0 | Low | 4.9 ± 0.1 | 8 | 16 |
| 5.5 – 7.0 | Normal | 6.8 ± 0.1 | 37 | 74 | |
| 7.5 – 9 | High | 8.4 ± 0.2 | 5 | 10 | |
| Protein Negative |
0 | Negative | 0.0 ± 0.0 | 9 | 18 |
| 30 – 1000 | Positive | 185.6 ± 62.54 | 41 | 82 | |
| Leukocytes (urine) Negative |
0 | Negative | 0.0 ± 0.0 | 18 | 36 |
| 25 – 500 | Positive | 219.1 ± 23.07 | 32 | 64 | |
| Red blood cells (urine) Negative |
0 | Negative | 0.0 ± 0.0 | 23 | 46 |
| 10 – 250 | Positive | 101.9 ± 22.3 | 27 | 54 | |
(*)Urinary biochemical reference values are based on Ettinger and Feldman (2017) and IRIS (2019).
Regarding urine specific gravity, most samples were in the intermediate range (1.015–1.045), accounting for 58%, indicating that many cases may have been in an early stage or at the beginning of renal functional decline. Low specific gravity (1.000–1.014) was found in 32% of samples, reflecting loss of urine-concentrating ability, commonly seen in chronic kidney disease or prolonged tubular damage. In contrast, 10% of samples had high specific gravity (1.045–1.052), which may be associated with dehydration, often observed in acute kidney disease. For urine pH, 74% of samples were within the intermediate range (5.5–7.0), indicating relatively stable acid–base balance. However, 16% of dogs had low pH (<5.5), suggesting acidification, commonly seen in acute kidney disease or metabolic disorders. Conversely, 10% of samples had high pH (>7.5), which may be related to advanced chronic kidney disease or secondary urinary tract infection (Bartges, 2012).
Proteinuria was detected in 41/50 dogs (82%) with concentrations ranging from 30–1000, reflecting damage to the glomerular filtration barrier, commonly seen in advanced chronic kidney disease or severe acute kidney disease. Only 18% had no proteinuria, indicating that most cases already had marked filtration dysfunction. Leukocyturia was recorded in 32/50 dogs (64%), indicating inflammation or infection of the urinary tract, nephritis, or pyelonephritis. In contrast, 36% showed no leukocytes in urine, reflecting variability in inflammatory status. Haematuria was present in 27/50 dogs (54%), suggesting renal or urinary tract damage such as inflammation, urolithiasis, or vascular injury; 46% showed no haematuria, indicating differences in severity and location of lesions among cases.
3.5. Prevalence of renal lesions detected by ultrasonography
Among dogs confirmed to have kidney disease, 83 were examined by ultrasonography to identify renal lesions, and the results are presented in Fig. 3.
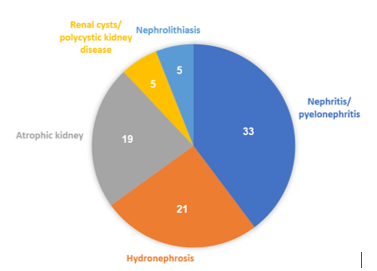
Fig. 3. Renal lesions detected by ultrasonography
As depicted in Fig. 3, nephritis/pyelonephritis was the most frequently recorded lesion, accounting for 39.76% (33/83 cases). Small kidneys were found in 22.89% (19/83 cases), while renal cysts/polycystic kidneys and renal calculi each accounted for 6.02% (5/83 cases).
3.6. Prevalence of kidney disease according to breed, sex, and age
Table 4. Prevalence of kidney disease by breed, sex, and age
| Category | Dogs with suspected kidney disease (n=219) | Dogs diagnosed with kidney disease (n=183) | Percentage (%) | P |
|---|---|---|---|---|
| Breed | ||||
| Native breed | 96 | 71 | 73.96 | 0.001 |
| Foreign breed | 123 | 112 | 91.06 | |
| Sex | ||||
| Male | 130 | 121 | 93.08 | 0.000 |
| Female | 89 | 62 | 69.66 | |
| Age (years) | ||||
| 0 – 2 | 60 | 52 | 86.67 | 0.038 |
| 3 – 5 | 72 | 63 | 87.50 | |
| 6 – 8 | 73 | 60 | 82.19 | |
| >9 | 14 | 8 | 57.14 | |
Dogs presented for examination showed wide variation in breed, sex, and age, reflecting the characteristics of the canine population in real clinical practice. Results in Table 4 show a clear difference in the prevalence of kidney disease between local and exotic breeds. Among 123 exotic dogs suspected of renal disease, 112 were confirmed (91.06%), significantly higher than in local dogs (71/96, 73.96%); this difference was statistically significant (p = 0.001).
By sex, male dogs had a higher prevalence of kidney disease than females. Among 130 suspected males, 121 were confirmed (93.08%), whereas in females the rate was 69.66% (62/89); this difference was highly significant (p = 0.000). By age, prevalence differed among age groups and was statistically significant (p = 0.038). The highest rate was in dogs aged 3–5 years (87.50%), followed by 0–2 years (86.67%) and 6–8 years (82.19%); dogs over 9 years had a lower rate (57.14%). This may reflect that young and middle-aged dogs are more often brought for examination when abnormal signs appear, whereas older dogs are examined less frequently or only when disease is advanced.
3.7. Prevalence of kidney disease according to housing method and diet
The prevalence of kidney disease according to housing method and diet among dogs with clinical signs suggestive of renal disease is presented in Table 5.
Table 5. Prevalence of kidney disease according to housing method and diet
| Category | Dogs with suspected kidney disease (n=219) | Dogs diagnosed with kidney disease (n=183) | Percentage (%) | P |
|---|---|---|---|---|
| Rearing method | ||||
| Kennel-kept | 164 | 162 | 98.78 | 0.000 |
| Free-roaming | 55 | 21 | 38.18 | |
| Diet | ||||
| Commercial dry food | 141 | 129 | 91.49 | 0.000 |
| Home-prepared food | 78 | 54 | 69.23 | |
Results in Table 5 show that kennel-kept dogs had a very high prevalence of kidney disease (98.78%), significantly higher than free-roaming dogs (38.18%); this difference was statistically significant (p = 0.000). Regarding diet, dogs fed commercial dry food had a higher prevalence of kidney disease than dogs fed home-prepared food (91.49% vs. 69.23%), and this difference was statistically significant (p = 0.000).
3.8. Treatment outcomes of kidney disease in dogs After diagnosis, dogs with renal disease were treated, and treatment outcomes are presented in Table 6.
Table 6. Treatment outcomes of acute and chronic kidney disease in dogs
| Treatment outcome | Acute kidney disease (n = 117) | Chronic kidney disease (n = 66) | ||||
|---|---|---|---|---|---|---|
| Number of dogs | Percentage (%) | P | Number of dogs | Percentage (%) | P | |
| Recovered | 48 | 41.03 | – | – | ||
| Improved | 19 | 16.24 | 0.000 | 49 | 74.24 | 0.000 |
| Died | 50 | 42.74 | 17 | 25.76 | ||
(1) Treatment outcomes were recorded after 1–2 weeks of therapy.
(2) Treatment outcomes were recorded after 3–4 weeks of therapy.
Results in Table 6 show that the recovery rate in dogs with acute kidney disease reached 41.03%. In the acute kidney disease group, 19 cases (16.24%) were classified as “improved.” In the chronic kidney disease group, 49 out of 66 cases showed improvement, accounting for 74.24%. The mortality rate in the acute kidney disease group was 42.74%. The chronic kidney disease group had a lower mortality rate (25.76%).
4.0 Discussion
The high proportion of acute kidney disease in Figure 1 may be explained by its rapid onset and obvious clinical signs such as oliguria, urinary retention, vomiting, lethargy, and dehydration, which make owners more likely to recognize abnormalities and seek veterinary care early. Bartges (2012) and Cooper (2013) also reported that acute kidney disease is easier to diagnose due to its characteristic clinical signs and rapid progression, often requiring emergency treatment. In contrast, the lower proportion of chronic kidney disease may be due to its insidious progression and nonspecific early signs, which are easily overlooked, leading to many cases being detected only at advanced stages, which is consistent with the observations of Ettinger and Feldman (2017).
The most common sign of reduced appetite/lethargy (81.28%) depicted in Figure 2 indicates it this is a very frequent but nonspecific sign that can be confused with many other internal diseases, consistent with the observations of Ettinger and Feldman (2017). The observation that vomiting/diarrhea accounted for 60.54% and occurred in both acute and chronic kidney disease (Figure 2) was mainly due to accumulation of metabolic waste products such as urea and creatinine that irritate the gastrointestinal tract (Polzin, 2011). However, these signs are also easily confused with gastrointestinal disease or intoxication and therefore must be evaluated together with paraclinical indices. The fact that oliguria was recorded in 100% of cases, indicates that it is a highly valuable sign for diagnosis, especially in acute kidney disease. Cooper (2013) and Ettinger and Feldman (2017) emphasised that oliguria or anuria is a key sign reflecting severe reduction in renal function. In contrast, polydipsia/polyuria also appeared in 100% of cases, mainly in chronic kidney disease, due to loss of the kidney’s ability to deal with concentrates (Kim et al., 2020). Furthermore, the severe signs of oral ulcers and halitosis observed are related to uremia (Steinbach et al., 2010), oedema results from impaired water–electrolyte regulation (Bartges, 2012), and seizures represent a serious sign associated with electrolyte imbalance and uremic neurotoxicity (Sundararajan & Vishnu Rahav (2025).
The observation in Figure 3 that nephritis was a common renal disorder in dogs when evaluated by ultrasonography, is consistent with Bartges (2012) who reported that nephritis and pyelonephritis are frequently associated with urinary tract infections and acute inflammation. The fact that hydronephrosis accounted for 25.30% of cases is reflective of a relatively common condition of urinary tract obstruction, often related to renal calculi, bladder stones, or ureteritis that impedes urine flow (Nyland, 2002). Therefore, hydronephrosis is an important lesion to monitor by ultrasonography because of its major significance in guiding treatment. The observation of small kidneys in 22.89% of the cases (Figure 3), is reflective of chronic structural changes in advanced chronic kidney disease, usually associated with fibrosis and prolonged parenchymal atrophy (Elliott, 2006). The renal cysts/polycystic kidneys and renal calculi cases may be related to genetic factors or prolonged chronic nephritis, while renal calculi, although less common, can cause obstruction and worsen renal damage through inflammation and urine retention.
With regards to the haematological parameters observed in dogs with kidney disease in Table 1, leukocytosis reflects inflammatory or infectious responses, commonly seen in nephritis, pyelonephritis, or acute kidney disease, consistent with the observations of Stockham and Scott (2008). The low red blood cell counts indicate that anaemia was very common, especially in chronic kidney disease. Polzin (2011) reported that non-regenerative anaemia is a frequent complication due to decreased renal production of erythropoietin. The decreased haemoglobin levels in 75.41% of the dogs was reflective of marked anaemia, which reduces oxygen-carrying capacity and contributes to clinical signs such as fatigue and lethargy. Chalhoub et al. (2011) also noted that decreased haemoglobin worsens general weakness in dogs with chronic kidney disease. Haematocrit was reduced, further confirming prolonged anaemia, characteristic of chronic kidney disease. It was also evident that the had thrombocytopenia, which indicates coagulation disorders and increased bleeding risk, consistent with the report of Bartges (2012) on haematological complications in chronic kidney disease.
The blood biochemical parameters in dogs with kidney disease displayed in Table 2 showed marked differences from normal physiological reference values, reflecting impaired filtration function and metabolic disturbances caused by renal damage. Elevated serum creatinine indicates marked reduction of glomerular filtration since Polzin (2011) and IRIS (2019) reported that creatinine is a key indicator for assessing the severity and staging of kidney disease because it is minimally influenced by extra-renal factors. Elevated BUN was indicative of the accumulation of nitrogenous waste products in the blood. Ettinger and Feldman (2017) noted that BUN reflects renal excretory capacity and is influenced by diet and hydration status, so it should be interpreted together with creatinine. The concurrent increase of both indices in this study further supports the diagnosis of impaired renal function. The observed hypercalcemia was consistent with Polzin (2011) who reported that hypocalcaemia is common in chronic kidney disease due to mineral imbalance that may be associated with dehydration or metabolic disorders in acute kidney disease. Serum phosphorus was also elevated indicating reduced renal excretion of phosphorus. According to IRIS (2019), hyperphosphatemia is an important factor promoting progression of chronic kidney disease and is associated with poor prognosis, consistent with the present findings. It would have been interesting to investigate if there was any link between IRIS classification and changes in parameters/ specific clinical findings, unfortunately, this could not be done retrospectively.
The breed, age and sex variations in kidney disease prevalence observed in Table 4 may be related to genetic factors and physical characteristics of exotic breeds, especially small breeds, as well as long-term indoor keeping and prolonged use of commercial diets, consistent with the reports of Polzin (2011) and Bartges (2012). Previously, Dadousis et al., (2024) showed that chronic kidney disease is more common in older dogs, so the lower rate in dogs over 9 years in this study may be due to small sample size or population characteristics, consistent with other Vietnamese studies by Khanh (2020) and Phu (2023).
The significant housing differences portrayed in Table 5 where kennel-kept dogs had a higher prevalence of kidney disease (98.78%) than free-roaming dogs (38.18%) could be attributed to kennel-kept dogs often associated with limited exercise, prolonged urine retention, and diets mainly based on commercial dry food rich in protein and minerals, which increases the burden on renal function. This agrees with Thao (2008) and Polzin (2011), who considered reduced activity and sedentary lifestyle as important risk factors for kidney disease in dogs. In contrast, free-roaming dogs are more physically active, drink water and urinate more regularly, hence the prevalence of kidney disease is lower, although they are still exposed to environmental risks and their diet is more difficult to control. In terms of dietary differences, commercial diets are often high in protein and phosphorus; long-term use without appropriate adjustment may increase renal workload, consistent with Bartges (2012). Home-prepared diets, although potentially nutritionally imbalanced, generally contain lower mineral levels, so their impact on the kidneys is usually less severe.
Treatment outcomes of acute and chronic kidney disease in dogs in Table 6 show that the recovery rate in dogs with acute kidney disease reached 41.03%, reflecting relatively good treatment effectiveness when the disease was detected and treated early. In contrast, no case of complete recovery was recorded in the chronic kidney disease group, which is consistent with the progressive and irreversible nature of this condition. In the acute kidney disease group, 19 cases (16.24%) were classified as “improved.” These dogs were initially diagnosed with acute kidney disease; however, after treatment, although clinical signs improved, serum creatinine decreased only slightly and did not return to normal values, so they were reclassified as chronic kidney disease. This indicates that some acute kidney disease cases may progress to chronic kidney disease if renal parenchymal damage does not completely recover, consistent with the findings of Vaden & Elliott (2016) and Srivastava et al. (2011). In the chronic kidney disease group, treatment mainly focused on controlling clinical signs, adjusting diet, and correcting electrolyte imbalance in order to slow disease progression and improve quality of life, rather than achieving complete recovery, in agreement with Polzin (2011) and O’Neill et al. (2013). The relatively high mortality rate in the acute kidney disease group of 42.74% was mainly due to delayed diagnosis leading to severe dehydration, electrolyte imbalance, and multiple organ failure. This is similar to the observation of Dadousis et al. (2024). The lower mortality rate (25.76%) in the chronic kidney disease group could be indicative of the follow-up period being not long enough to determine median survival time. Treatment goals therefore focused mainly on prolonging survival and maintaining quality of life, consistent with Rimer et al. (2022).
5.0 Conclusion
Among 219 dogs showing clinical signs suspicious for kidney disease, 183 were confirmed to be affected, accounting for 83.56%. Of these, acute kidney disease was more prevalent (53.42%) than chronic kidney disease (30.14%). The most common clinical signs were oliguria/anuria and polydipsia/polyuria, accompanied by systemic signs such as anorexia, lethargy, vomiting, and in advanced stages, oral ulcers, halitosis, and generalized oedema. Major risk factors identified included kennel housing, long-term feeding of commercial dry diets, urinary tract infection, nephritis/pyelonephritis, exposure to toxins, and metabolic disorders. Paraclinical results showed markedly elevated serum creatinine, especially at severe levels (>440 µmol/L), together with increased BUN and phosphorus, indicating significant impairment of renal filtration in most cases. In addition, haematological abnormalities such as anaemia (decreased RBC count, haemoglobin, and haematocrit) and thrombocytopenia were common, demonstrating the systemic impact of renal disease. Ultrasonographic findings frequently included nephritis/pyelonephritis, hydronephrosis, and small kidneys, consistent with both acute and chronic renal lesions. The prevalence of kidney disease was significantly higher in exotic breeds, male dogs, and kennel-kept dogs, highlighting the roles of breed, physiology, and management conditions as risk factors. Regarding treatment outcome, acute kidney disease showed a relatively good potential for recovery when detected and treated early, with a recovery rate of 41.03%; however, mortality remained high (42.74%) in cases diagnosed late or at severe stages. In contrast, no complete recovery was recorded in chronic kidney disease; treatment mainly reduced clinical signs and slowed disease progression, with 74.24% of cases showing improvement rather than cure. The key take home message for clinicians dealing with canine kidney disease is to factor in the roles of breed, sex, housing and management conditions in its prevalence. The practical
application and implications of the study findings highlight the importance of early detection and routine renal monitoring in veterinary clinical practices.
Author Contributions: Conceptualisation: Hau Van Bui, Thong Trung Ho, Loan Vu Thuy Hong Nguyen; Methodology: Hau Van Bui, Anh Thi Lan Trinh, Loan Vu Thuy Hong Nguyen; Validation: Thong Trung Ho, Loan Vu Thuy Hong Nguyen; Formal analysis, investigation: Hau Van Bui, Thong Trung Ho, Loan Vu Thuy Hong Nguyen; Data Curation: Hau Van Bui; Writing—Original Draft Preparation: Hau Van Bui; Writing—Review and Editing: Thong Trung Ho, Loan Vu Thuy Hong Nguyen, Anh Thi Lan Trinh. All authors have read and agreed to the published version of the manuscript.
Funding: This research received no external funding.
Acknowledgments: The authors sincerely thank the PET HEALTH CENTRE Veterinary Hospital System for providing the clinical cases presented in this report.
Conflicts of Interest: The authors declare no conflicts of interest.
Artificial Intelligence: AI was not used for this original research article.
References
Bartges JW. 2012. Chronic kidney disease in dogs and cats. Small Animal Practice, 42(4), 669. https://doi.org/10.1016/j.cvsm.2012.04.008
Chalhoub S, Langston C, Eatroff A. 2011. Anaemia of renal disease: What it is, what to do and what’s new. Journal of Feline Medicine and Surgery, 13(9), 629–640. https://doi.org/10.1016/j.jfms.2011.07.016
Cooper C. 2013. Fluid, electrolyte, and acid-base disorders in small animal practice. The Canadian Veterinary Journal, 54(4), 386. https://pmc.ncbi.nlm.nih.gov/articles/PMC3595945/
Dadousis C, Whetton AD, Mwacalimba K, Merlo A, Wright A, Geifman N. 2024. Renal disease in cats and dogs – lessons learned from text-mined trends in humans. Animals, 14(23), 3349. https://doi.org/10.3390/ani14233349
Dan TT, Khang ND. 2018. Renal physiology and acid–base balance. In: Animal Physiology pp146–182. Ho Chi Minh City: Agriculture Publishing House. Cited Lote CJ. 2012. Principles of renal physiology. 5th Edition. Springer, London. ISBN 978-1-4614-3784-0 ISBN 978-1-4614-3785-7 (eBook) DOI https://doi.org/10.1007/978-1-4614-3785-7
Elliott DA. 2006. Nutritional management of chronic renal disease in dogs and cats. Small Animal Practice, 36(6), 1377–viii. https://doi.org/10.1016/j.cvsm.2006.08.011
Ettinger SJ, Feldman EC. 2017. Textbook of Veterinary Internal Medicine: Diseases of the Dog and Cat (8th ed.). Elsevier, St. Louis, Missouri, USA. 2220 pages. https://shop.elsevier.com/books/textbook-of-veterinary-internal-medicine-expert-consult/ettinger/978-0-323-31211-0
Goldston RT. 1995. Introduction and overview of geriatrics. Geriatrics and Gerontology of the Dog and Cat. W.B. Saunders.
International Renal Interest Society. IRIS staging of CKD. https://www.iris-kidney.com/guidelines/staging.html
Khanh PT. 2020. Study on clinical and paraclinical characteristics of kidney diseases in dogs in Ho Chi Minh City. Master’s Thesis, Nong Lam University, Ho Chi Minh City, Vietnam. Cited Ettinger SJ, Feldman EC. 2017. Textbook of Veterinary Internal Medicine: Diseases of the Dog and Cat (8th ed.). Elsevier, St. Louis, Missouri, USA. 2220 pages. https://shop.elsevier.com/books/textbook-of-veterinary-internal-medicine-expert-consult/ettinger/978-0-323-31211-0
Kim J, Lee CM, Kim HJ. 2020. Biomarkers for chronic kidney disease in dogs: a comparison study. The Journal of Veterinary Medical Science, 82(8), 1130–1137. https://doi.org/10.1292/jvms.20-0125
Minitab. 2026. Minitab Statistical Software. PA, USA. https://www.minitab.com/en-us/products/minitab/
Nguyen TPK. 2006. Application of X-ray techniques in the diagnosis of urinary system diseases in dogs at the Ho Chi Minh City Veterinary Sub-Department. Master’s Thesis, Nong Lam University, Ho Chi Minh City, Vietnam. Cited Ettinger SJ, Feldman EC. 2017. Textbook of Veterinary Internal Medicine: Diseases of the Dog and Cat (8th ed.). Elsevier, St. Louis, Missouri, USA. 2220 pages. https://shop.elsevier.com/books/textbook-of-veterinary-internal-medicine-expert-consult/ettinger/978-0-323-31211-0
Nyland T. 2002. Small animal diagnostic ultrasound (2nd Edition). Elsevier, USA. 480 pages. https://www.elsevierhealth.com.au/small-animal-diagnostic-ultrasound-9780721677880.html?srsltid=AfmBOoqzvO8dutQ6I6br0auQhpFlqOKttOlxyG8azQDbDZarT7nHfMZ8
O’Neill DG, Elliott J, Church DB, McGreevy PD, Thomson PC, Brodbelt DC. 2013. Chronic kidney disease in dogs in UK veterinary practices: prevalence, risk factors, and survival. Journal of Veterinary Internal Medicine, 27(4), 814–821. https://doi.org/10.1111/jvim.12090
Perondi F, Lippi I, Marchetti V, Bruno B, Borrelli A, Citi S. 2020. How ultrasound can be useful for staging chronic kidney disease in dogs: Ultrasound findings in 855 cases. Veterinary Sciences, 7(4), 147. https://doi.org/10.3390/vetsci7040147
Phu QT. 2023. Investigation of renal diseases using clinical and paraclinical techniques in dogs in Ho Chi Minh City. Undergraduate thesis, Ho Chi Minh City University of Technology, Vietnam. Cited Ettinger SJ, Feldman EC. 2017. Textbook of Veterinary Internal Medicine: Diseases of the Dog and Cat (8th ed.). Elsevier, St. Louis, Missouri, USA. 2220 pages. https://shop.elsevier.com/books/textbook-of-veterinary-internal-medicine-expert-consult/ettinger/978-0-323-31211-0
Polzin DJ. 2011. Chronic kidney disease in small animals. Small Animal Practice, 41(1), 15–30. https://doi.org/10.1016/j.cvsm.2010.09.004
Rimer D, Chen H, Bar-Nathan M, Segev G. 2022. Acute kidney injury in dogs: Etiology, clinical and clinicopathologic findings, prognostic markers, and outcome. Journal of Veterinary Internal Medicine, 36(2), 609–618. https://doi.org/10.1111/jvim.16375
Srivastava M, Gaikwad RV, Samad A, Sharma B, Srivastava A. 2011. Relationship of serum creatinine and glomerular filtration rate by 99mTc-DTPA scintigraphy in dogs with renal failure. Asian Journal of Animal Sciences, 5, 381–386. https://doi.org/10.3923/ajas.2011.381.386
Steinbach S, Binkert B, Schweighauser A, Reynolds B, Séguéla J, Lefebvre H, Francey T. 2010. Quantitative assessment of urea generation and elimination in healthy dogs and in dogs with chronic kidney disease. Journal of Veterinary Internal Medicine, 24(6), 1283–1289. https://doi.org/10.1111/j.1939-1676.2010.0608.x
Stockham SL, Scott MA. 2008. Fundamentals of veterinary clinical pathology (2nd ed.). Wiley-Blackwell, Ames, Iowa, USA. 928 pages. https://www.wiley.com/en-ae/Fundamentals+of+Veterinary+Clinical+Pathology%2C+2nd+Edition-p-9781118686072
Sundararajan RC, Vishnu-Rahav RB. 2025. Chronic kidney disease in a dog and its clinical management. https://doi.org/10.13140/RG.2.2.24698.35522
Thao TT. 2008. Effects of housing conditions on the health of dogs in Hanoi. Master’s Thesis, Hanoi University of Agriculture, Vietnam. Cited Ettinger SJ, Feldman EC. 2017. Textbook of Veterinary Internal Medicine: Diseases of the Dog and Cat (8th ed.). Elsevier, St. Louis, Missouri, USA. 2220 pages. https://shop.elsevier.com/books/textbook-of-veterinary-internal-medicine-expert-consult/ettinger/978-0-323-31211-0
Vaden SL, Elliott J. 2016. Management of proteinuria in dogs and cats with chronic kidney disease. Small Animal Practice, 46(6), 1115–1130. https://doi.org/10.1016/j.cvsm.2016.06.009
Disclaimer/Publisher’s Note: The statements, opinions, institutional affiliations, data contained in all publications, and all responsibilities for accuracy are solely those of the individual author(s) and contributor(s) and not of MARCIAS AUSTRALIA and AJAVAS/or the Editor(s). MARCIAS AUSTRALIA and AJAVAS/or the Editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.

