Evaluation of myxomatous mitral valve disease in dogs
Thuy Hoang Nguyen 1 Kinh Van La 1 Yen Hai Vu 1 Quynh Ngoc Nhu Nguyen 1 Loan Vu Thuy Hong Nguyen 1
- Faculty of Veterinary Medicine and Animal Science, HUTECH University, Binh Thanh District, Ho Chi Minh City 717000, Vietnam
Article Information
- Date Received: 21/01/2026
- Date Revised: 10/04/2026
- Date Accepted: 11/04/2026
- Date Published Online: 27/04/2026
Copyright: © 2026 The Authors. Published by MARCIAS AUSTRALIA, 32 Champion Drive, Rosslea, Queensland 4812, Australia. This is an open access publication under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Citation: Nguyen TH, La KV, Vu YH, Nguyen QNN, Nguyen LVTH (2026). Evaluation of myxomatous mitral valve disease in dogs. Aust J Agric Vet Anim Sci (AJAVAS), 2(1), 100013
https://doi.org/10.64902/ajavas.2026.100013
Abstract
This study evaluated the prevalence, clinical characteristics, diagnostic indicators, and treatment outcomes of myxomatous mitral valve disease (MMVD) in dogs at Procare Veterinary Hospital. From October 2024 to October 2025, a total of 3,840 dogs presenting with respiratory signs or undergoing health screening were examined. Dogs suspected of cardiac disease were further evaluated using thoracic radiography, echocardiography, and NT-proBNP quantification. Results showed that 232/3,840 dogs (6.04%) were suspected of MMVD, of which 84 dogs (36.21%) were definitively diagnosed by echocardiography, accounting for 2.19% of all examined dogs. The most common clinical signs were dyspnea/pulmonary edema (38.94%) and cough/respiratory inflammation (33.17%), with increasing frequency from stage B1 to C. Cardiac murmur intensity increased with disease progression; most dogs in stages B2 and C had murmurs ≥ grade III/VI. VHS, LA/Ao, LVIDd, and LVIDs increased significantly across stages (p < 0.001), while fractional shortening (FS) showed no significant change (p = 0.575). Mean LA/Ao reached 1.76 in stage B2 and 2.18 in stage C. NT-proBNP concentrations increased from 500 pg/mL (B1) to 953.8 ± 294.0 pg/mL (B2) and 2,131.8 ± 354.4 pg/mL (C). Treatment outcomes were excellent in stage B1 (100%) and B2 (98.18%), but markedly lower in stage C (43.48%). These findings indicate that MMVD is a common acquired cardiac disease in dogs. Combined clinical evaluation, diagnostic imaging, and NT-proBNP measurement are essential for staging, monitoring disease progression, and improving treatment outcomes through early detection.
Keywords
Myxomatous mitral valve disease; dogs; echocardiography; NT-proBNP.
Highlights
- Myxomatous mitral valve disease affected 2.19% of examined dogs; respiratory signs increased with disease stage.
- Cardiac indices and NT-proBNP rose significantly from stage B1 to C.
- Early-stage treatment was highly successful, but outcomes declined markedly in stage C.
1.0 Introduction
Myxomatous mitral valve disease (MMVD) is one of the most common cardiac diseases in dogs, particularly prevalent in small breeds and geriatric dogs. The disease is characterised by chronic degeneration of the mitral valve leaflets, leading to incomplete valve closure, mitral regurgitation, left atrial enlargement, and chronic volume overload, which may eventually progress to congestive heart failure if not diagnosed and treated in time. In Vietnam, especially in Ho Chi Minh City, studies on the epidemiology, clinical features, and paraclinical characteristics of MMVD remain limited. There are still few published data on the integrated use of echocardiography, thoracic radiography, and NT-proBNP testing for the diagnosis and monitoring of MMVD in domestic veterinary practice.
Recent advances in diagnostic imaging and biomarkers, together with the staging system proposed by the American College of Veterinary Internal Medicine (ACVIM), have improved diagnostic accuracy and enabled individualised management of MMVD (Van Staveren et al., 2023). In this context, the present study collected and analysed data on disease prevalence, clinical characteristics, and treatment outcomes at a specialised veterinary facility, thereby contributing scientific evidence for the diagnosis, monitoring, and management of MMVD under practical conditions in Vietnam.
2.0 Materials and methods
2.1. Study site, animals, housing, and experimental design
The study was conducted from October 2024 to October 2025 at Procare Veterinary Hospital, Phu Nhuan District, Ho Chi Minh City. Dogs included in the study were those presenting with clinical signs such as cough, dyspnea, exercise intolerance, fatigue, or syncope, as well as dogs without clinical signs that were brought for routine annual health screening or pre-anaesthetic examination.
2.2. Data collection
2.2.1. Clinical examination and classification of dogs suspected of MMVD – Cardiovascular clinical Examination
After collecting a complete history from the owner, dogs underwent a general clinical examination with particular focus on the cardiovascular system. Cardiac auscultation was performed using a stethoscope to detect systolic murmurs, a characteristic sign of myxomatous mitral valve disease, most clearly heard at the 4th–6th intercostal spaces (Turgut et al., 2019). Murmur intensity was graded from I to VI according to severity (Pedersen et al., 1999). Heart rate was recorded, with normal physiological values ranging from 60 to 140 beats per minute; in dogs with MMVD, heart rate may increase due to compensatory mechanisms. Peripheral pulse was assessed to reflect circulatory status; weak or irregular pulses may be associated with cardiac dysfunction. Resting respiratory rate was measured, with normal values of 18–34 breaths per minute; a rate above 30 breaths per minute often suggests congestive heart failure or pulmonary oedema (Goutal et al., 2010).
2.2.2 Classification of dogs suspected of MMVD
Stage B1: Dogs with MMVD but without evident left atrial enlargement; echocardiography and radiography do not show significant regurgitation. Monitoring is mainly based on periodic auscultation, resting respiratory rate, and scheduled echocardiographic follow-up to detect early disease progression.
Stage B2: Dogs without clinical signs but with left atrial and/or left ventricular enlargement detected by echocardiography or radiography. These dogs are monitored using LA/Ao, LVIDd, LVIDs, VHS ≥ 10.5, and NT-proBNP testing to assess the degree of cardiac enlargement and risk of progression to heart failure.
Stage C (with clinical signs): Dogs show obvious signs such as loud murmurs, dyspnea, fatigue, pulmonary edema, or congestive heart failure. This stage is subdivided into C1 (outpatient treatment) and C2 (requires hospitalization). Evaluation is based on echocardiography with LA/Ao > 1.6 and increased LVID (Keene et al., 2019), thoracic radiography with VHS > 10.5 and signs of pulmonary edema or pleural effusion (Buchanan & Bücheler, 1995), and NT-proBNP usually > 2000 pmol/L indicating severe congestive heart failure (Oyama et al., 2009).
2.2.3. Procedures for assessing paraclinical parameters in MMVD
Paraclinical evaluation in this MMVD study followed a standardized protocol including the three main techniques of thoracic radiography, echocardiography, and biochemical testing of NT-proBNP.
Thoracic radiography: Before radiography, dogs were briefly examined to assess respiratory status and stabilised if critical. Dogs were positioned in right lateral and ventrodorsal recumbency with gentle restraint to minimize stress. Images were obtained using a digital X-ray system and analyzed for cardiac size and shape, left atrial and ventricular enlargement, and signs of pulmonary edema or pleural effusion. The vertebral heart scale (VHS) was calculated according to Buchanan and Bücheler, with normal values ranging from 8.7 to 10.5; higher values indicate cardiomegaly (Buchanan & Bücheler, 1995).
Echocardiographic evaluation: The thoracic area of dogs was clipped, gently disinfected, and positioned in lateral recumbency with light restraint before examination. Transthoracic echocardiography was performed using 2D, M-mode, and color Doppler techniques. Two-dimensional imaging was used to detect thickening or prolapse of the mitral valve in standard views (Menciotti & Borgarelli, 2017). M-mode was used to measure LVIDD and LVIDS, which were then normalized to body weight to calculate LVIDDN and LVIDSN according to Cornell et al. (2004), and fractional shortening (FS) was calculated. Color Doppler was used to detect and assess mitral regurgitation. The LA/Ao ratio was used to evaluate left atrial enlargement, with values > 1.6 indicating progressive MMVD (Keene et al., 2019). Additional parameters included ejection fraction (EF; normal 50–70%) and the E/A ratio to assess systolic and diastolic function (Oyama et al., 2009).
NT-proBNP Testing: Blood samples were collected from peripheral veins into anticoagulant tubes, gently mixed, and analyzed using a fluorescence analyzer according to the manufacturer’s protocol. Results were recorded for follow-up. NT-proBNP values < 900 pmol/L were considered normal; values > 1500–1800 pmol/L indicated high risk of MMVD-related heart failure, and levels > 2000 pmol/L were commonly associated with overt congestive heart failure (Oyama et al., 2009). This biomarker was used in combination with echocardiography and radiography to assess disease severity and monitor treatment response.
2.3. Statistical analysis
Data were collected on an individual basis and analysed using Minitab 17. Quantitative variables were presented as n, mean ± SD, minimum, and maximum. Comparisons of means were performed using one-way analysis of variance (ANOVA, F-test) followed by Tukey’s post-hoc test. Percentages were compared using the Chi-square (χ²) test. Differences were considered statistically significant when p ≤ 0.05.
3.0 Results and discussion
3.1. Survey of dogs suspected of and diagnosed with MMVD
From October 2024 to October 2025, a total of 3,840 dogs were presented for health screening or examination due to respiratory signs at Procare Veterinary Hospital. Among these, dogs showing clinical signs suggestive of cardiac disease were selected for further evaluation using diagnostic imaging and supportive laboratory tests; the results are presented in Figure 1.
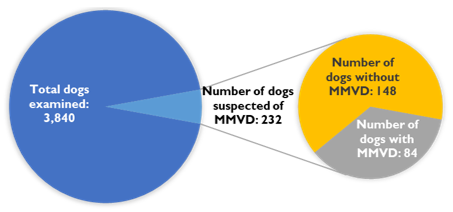
Fig. 1. Number of dogs suspected of MMVD and dogs diagnosed with MMVD at Procare Veterinary Hospital
Figure 1 shows that 232/3,840 dogs (6.04%) were suspected of having cardiac disease based on clinical signs such as heart murmur, coughing, and reduced exercise tolerance. Echocardiography confirmed MMVD in 84/232 dogs (36.21%), corresponding to 2.19% of all examined dogs. This proportion is consistent with previous studies in small and medium-sized dogs (Borgarelli and Buchanan, 2012).
Although 6.04% of dogs were suspected of cardiac disease, only about one third of these were definitively diagnosed with MMVD by echocardiography, highlighting the crucial role of diagnostic imaging in confirming the disease. Most dogs presented with respiratory signs, but only 2.19% were confirmed to have MMVD, indicating that respiratory signs are nonspecific and have limited screening value if not combined with echocardiography. This finding is consistent with Lam et al. (2022), who reported that 74.6% of dogs with MMVD in stages B1–B2 had concurrent lower respiratory tract disease, complicating differential diagnosis.
3.2. Frequency of clinical signs in dogs with MMVD
Assessment of the frequency of clinical signs in dogs with MMVD is important for staging, supporting differential diagnosis with non-cardiac diseases, and evaluating disease progression to guide appropriate treatment strategies.
Table 1. Frequency of Clinical Signs in Dogs with MMVD (n = 84)
| Clinical sign | Stage B1 | Stage B2 | Stage C | Total | P-value | ||||
|---|---|---|---|---|---|---|---|---|---|
| Number of dogs | Percentage (%) | Number of dogs | Percentage (%) | Number of dogs | Percentage (%) | Number of dogs | Percentage (%) | ||
| Tachypnea/ Dyspnea/ Pulmonary oedema | 0 | 0.00 | 55 | 36.67 | 23 | 44.23 | 78 | 37.50 | |
| Cough / Respiratory Inflammation | 0 | 0.00 | 53 | 35.33 | 13 | 25.00 | 66 | 31.73 | |
| Asymptomatic / Decreased appetite | 6 | 100 | 22 | 14.67 | 1 | 1.92 | 29 | 13.94 | 0.000 |
| Reduced exercise tolerance | 0 | 0.00 | 19 | 12.67 | 2 | 3.85 | 21 | 10.10 | |
| Syncope / Transient loss of consciousness | 0 | 0.00 | 1 | 0.66 | 13 | 25.00 | 14 | 6.73 | |
As shown in Table 1, the two most common groups of clinical signs were dyspnea/pulmonary oedema (37.50%) and cough/respiratory inflammation (31.73%). This pattern is consistent with the pathophysiology of MMVD, in which mitral regurgitation and left atrial enlargement increase pulmonary venous pressure, leading to pulmonary congestion, chronic cough, and possible progression to pulmonary oedema.
By disease stage, dogs in stage B1 showed almost no clinical signs other than a heart murmur, in agreement with the ACVIM consensus (2019) that this stage is typically asymptomatic. In stage B2, the frequency of clinical signs increased markedly, with dyspnea/pulmonary oedema observed in 36.67% of dogs, cough in 35.33%, and poor appetite or gastrointestinal signs in 14.67%. These findings are consistent with Häggström et al. (2009) and Moonarmart et al. (2020), who reported that stage B2 may show early pulmonary congestion or reduced exercise tolerance despite the absence of overt congestive heart failure.
In stage C, typical signs of MMVD were clearly evident. Dyspnea/pulmonary oedema was the most frequent sign (44.23%), reflecting left-sided heart failure and increased pulmonary pressure (Keene et al., 2019). Syncope or transient loss of consciousness occurred in 25.00% of cases, associated with reduced cardiac output and cerebral perfusion, similar to the findings of Borgarelli et al. (2012). Reduced exercise tolerance reflected the systemic impact of chronic heart failure. The frequency and severity of clinical signs increased with disease progression; however, because clinical signs are nonspecific and influenced by many factors, they should be interpreted together with radiography and echocardiography to accurately diagnose MMVD and determine disease stage.
3.3. Classification of cardiac murmur intensity by disease stage according to ACVIM
The analysis showed that cardiac murmur intensity in dogs with MMVD increased markedly as the disease progressed from stage B1 to B2 and C, reflecting a close relationship between murmur severity and disease severity.
Table 2. Cardiac murmur intensity by disease stage according to ACVIM
| Heart murmur grade | Stage B1 | Stage B2 | Stage C | Total | P-value | ||||
|---|---|---|---|---|---|---|---|---|---|
| Number of dogs | Percentage (%) | Number of dogs | Percentage (%) | Number of dogs | Percentage (%) | Number of dogs | Percentage (%) | ||
| 0/VI | 2 | 33.33 | 0 | 0.00 | 0 | 0.00 | 2 | 2.38 | 0.000 |
| I/VI | 3 | 50.00 | 2 | 3.64 | 0 | 0.00 | 5 | 5.95 | |
| II/VI | 1 | 16.67 | 12 | 21.82 | 0 | 0.00 | 13 | 15.48 | |
| III/VI | 0 | 0.00 | 31 | 56.35 | 6 | 26.09 | 37 | 44.05 | |
| IV/VI | 0 | 0.00 | 8 | 14.55 | 12 | 52.17 | 20 | 23.81 | |
| V-VI/VI | 0 | 0.00 | 2 | 3.64 | 5 | 21.74 | 7 | 8.33 | |
| Total | 6 | 100 | 55 | 100 | 23 | 100 | 84 | 100 | |
In stage B1, murmurs were mainly mild, consistent with Borgarelli et al. (2012), who reported that valvular lesions are already present but regurgitation is still mild and does not create a large pressure gradient. In stage B2, grade III/VI murmurs were most common (56.35%), as left atrial and ventricular enlargement increases blood flow across the valve, making the murmur more audible. This finding is consistent with the EPIC study by Boswood et al. (2016), which showed that most stage B2 dogs have murmurs of grade III–IV/VI due to strong regurgitant jets and increased regurgitant orifice area.
In stage C, murmurs shifted to more severe grades, with grade IV/VI accounting for 52.17% and grades V–VI/VI for 21.74%. However, 26.09% of dogs still had grade III/VI murmurs, likely due to factors such as pulmonary edema, lung crackles, or reduced cardiac output that can diminish murmur audibility. The high proportion of murmurs ≥ IV/VI in this stage is consistent with Fox et al. (2012), who reported that 80–90% of stage C dogs have murmurs of grade IV/VI or higher.
Häggström et al. (2009) reported that murmur intensity not only reflects the severity of mitral regurgitation but also has prognostic value for disease progression and the risk of congestive heart failure. Borgarelli et al. (2010) also showed that murmurs of grade III/VI or higher have high sensitivity for predicting moderate to severe mitral regurgitation on color Doppler echocardiography. In this study, most stage B2 and C dogs had murmurs ≥ III/VI, indicating a close relationship between murmur intensity and the degree of valvular deformation, a hallmark of MMVD.
Keene et al. (2019) noted that changes in murmur intensity are often more pronounced in small breeds because of smaller valve orifice area, whereas in large breeds, murmur intensity may not accurately reflect regurgitation severity due to larger heart size and thicker chest walls. This explains why grade III/VI murmurs were still observed in some stage C dogs, especially when pulmonary oedema or reduced cardiac output was present.
Boswood et al. (2016) further reported that murmurs ≥ III/VI combined with LA/Ao > 1.6 and increased LVIDDN indicate progression to stage B2 and a high risk of transition to stage C within 1–2 years. Therefore, regular clinical examination and murmur assessment remain simple but valuable screening tools.
3.4. Radiographic and echocardiographic indices in dogs at different ACVIM stages
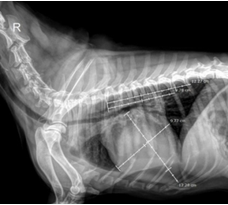
Fig. 2. Measurement of VHS in Dogs with MMVD
The results in Figure 2 show that imaging parameters changed markedly with disease progression. The vertebral heart scale (VHS) increased significantly from stage B1 to B2 and C (p < 0.001), reflecting cardiomegaly due to chronic volume overload. Mean VHS increased from 10.00 ± 0.19 (B1) to 10.85 ± 0.18 (B2) and 11.66 ± 0.32 (C), consistent with the findings of Levicar et al. (2022), who reported that VHS > 11 is an early indicator of congestive heart failure. The QUEST study and Häggström et al. (2008) also indicated that VHS > 10.5 is associated with poor prognosis.
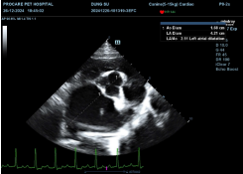
Fig. 3. Measurement of the LA/Ao ratio in dogs with MMVD
The LA/Ao ratio increased significantly across stages (p < 0.001), from 1.38 ± 0.10 (B1) to 1.76 ± 0.08 (B2) and 2.18 ± 0.17 (C). According to the EPIC trial (Boswood et al., 2016), LA/Ao ≥ 1.6 defines stage B2. The high LA/Ao values in stage C reflect severe left atrial enlargement due to chronic mitral regurgitation and are closely associated with the risk of congestive heart failure and survival time (Boswood et al., 2016).
Table 3. Radiographic and echocardiographic parameters in dogs with MMVD at different ACVIM stages
| Stage | Statistical parameters | Indices | ||||
|---|---|---|---|---|---|---|
| VHS | LA/Ao | FS | LVIDd | LVIDs | ||
| B1 | n | 6 | 6 | 6 | 6 | 6 |
| Mean | 10.00c | 1.38c | 59.82c | 1.47c | 0.67c | |
| SD | 0.19 | 0.10 | 9.35 | 0.03 | 0.12 | |
| Min | 9.80 | 1.26 | 40.51 | 1.42 | 0.54 | |
| Max | 10.30 | 1.51 | 60.72 | 1.50 | 0.85 | |
| B2 | n | 55 | 55 | 55 | 55 | 55 |
| Mean | 10.85b | 1.76b | 52.03a | 1.75b | 0.81b | |
| SD | 0.18 | 0.08 | 9.47 | 0.03 | 0.10 | |
| Min | 10.60 | 1.62 | 37.26 | 1.70 | 0.65 | |
| Max | 11.20 | 1.91 | 80.35 | 1.84 | 0.99 | |
| C | n | 23 | 23 | 23 | 23 | 23 |
| Mean | 11.66a | 2.18a | 49.70a | 2.03a | 0.99a | |
| SD | 0.32 | 0.17 | 9.90 | 0.06 | 0.09 | |
| Min | 11.00 | 1.96 | 29.64 | 1.70 | 0.79 | |
| Max | 12.00 | 2.50 | 81.49 | 1.84 | 1.10 | |
| P – value | 0.000 | 0.000 | 0.575 | 0.000 | 0.000 | |
In contrast, fractional shortening (FS) decreased only slightly from 52.92% (B1) to 49.70% (C) and showed no significant difference (p = 0.575). This agrees the ACVIM consensus that MMVD mainly causes volume overload rather than primary systolic dysfunction; therefore, FS has limited value in assessing disease severity.
Both LVIDd and LVIDs increased significantly (p < 0.001) from B1 to C, indicating progressive eccentric hypertrophy and left ventricular remodelling. These findings are consistent with Franco et al. (2011), who showed that these indices are sensitive markers of disease progression and treatment response. Thus, imaging parameters, especially VHS, LA/Ao, and LVIDd/LVIDs, are highly valuable for staging and prognostication of MMVD. Thresholds such as VHS > 10.5–11, LA/Ao > 1.6–2.0, and LVIDd > 2.0 help distinguish compensated from decompensated stages. Although FS remains within normal limits in early stages, structural changes of the cardiac chambers occur early, indicating that assessment of cardiac morphology should be prioritized over systolic function alone in monitoring MMVD.
3.5. NT-proBNP quantification across disease stages
Table 4 shows that NT-proBNP concentrations increased markedly with disease progression, reaching 953.8 ± 294.0 pg/mL in stage B2 and 2,131.8 ± 354.4 pg/mL in stage C, with a statistically significant difference (p < 0.001). This increase reflects chronic volume overload and sustained pressure on the myocardium, especially the left ventricle, caused by long-standing mitral regurgitation in MMVD. These conditions activate the natriuretic peptide system, leading to increased release of BNP and NT-proBNP; therefore, NT-proBNP levels rise in parallel with the degree of cardiac remodelling and myocardial stress.
Table 4. NT-proBNP concentrations in different disease stages
| Stage | Statistical parameters | Indices (NT-proBNP) |
|---|---|---|
| B1 | n | 6 |
| Mean | 500c | |
| SD | 0.00 | |
| Min | 500 | |
| Max | 500 | |
| B2 | n | 55 |
| Mean | 953.8b | |
| SD | 294.0 | |
| Min | 500 | |
| Max | 1356.2 | |
| C | n | 23 |
| Mean | 2131.8a | |
| SD | 354.4 | |
| Min | 1625.5 | |
| Max | 2935.2 | |
| P – value | 0.000 | |
At stage B2, NT-proBNP increased to nearly twice the level observed in stage B1 and showed wide inter-individual variation, indicating that the degree of cardiac remodelling is not uniform within the same stage. Several studies have shown that NT-proBNP rises significantly in B2 dogs with LA/Ao ≥ 1.6 and increased LVIDd, even in the absence of clinical signs of heart failure (Borgarelli et al., 2012). Therefore, this biomarker is useful for identifying B2 dogs at high risk of progressing to congestive heart failure.
At stage C, NT-proBNP commonly exceeds 2,000 pg/mL, reflecting overt congestive heart failure and severe myocardial stress. Studies have demonstrated that NT-proBNP has high sensitivity and specificity in distinguishing MMVD dogs with and without clinical heart failure. High NT-proBNP levels are often associated with increased left atrial pressure, pulmonary congestion, and reduced cardiac output, and are linked to a poorer survival prognosis. Moreover, correlations between NT-proBNP and imaging parameters such as LA/Ao, LVIDd, and VHS have been reported, indicating that NT-proBNP reflects not only the current heart failure status but also the extent of chronic cardiac remodelling.
3.6. Treatment outcomes of MMVD according to disease stage
The treatment outcomes of MMVD according to disease stage are summarized and presented in Table 5.
Table 5. Treatment outcomes of MMVD by disease stage
| Stage | B1 | B2 | C | Total |
|---|---|---|---|---|
| Number of cases with effective treatment | 6 | 54 | 10 | 70 |
| Treatment success rate (%) | 100 | 98.18 | 43.48 | 83.33 |
| Number of cases with ineffective treatment | 0 | 1 | 13 | 14 |
| Treatment failure rate (%) | 0 | 1.82 | 56.52 | 16.67 |
At stage B1, the treatment response rate was 100%, with no non-responsive cases recorded. This is consistent with the nature of stage B1, in which there is no marked cardiac remodelling, mitral regurgitation is mild, and clinical signs are minimal or absent. At this stage, periodic echocardiographic monitoring every 6 months is important for early detection of disease progression.
At stage B2, the treatment success rate was 98.18%, with only 1 case (1.82%) showing no response. Although cardiac chamber remodelling is already present, compensatory mechanisms remain effective, allowing good response to therapy and delaying progression to heart failure.
In contrast, at stage C, treatment success dropped markedly to 43.48%, while 56.52% of cases were non-responsive. This indicates that once the disease enters the decompensated stage, the effectiveness of standard treatment protocols decreases substantially. Possible reasons include reduced cardiac compensatory capacity, pulmonary hypertension, recurrent pulmonary oedema, arrhythmias, and the need for more complex care. Overall, treatment efficacy declines with disease progression, from B1 (100%) to B2 (98.18%) and lowest at stage C (43.48%), highlighting the importance of early detection and stage-based management to optimize treatment outcomes, prolong survival, and improve quality of life in dogs with MMVD.
4.0 Conclusion
4.0 Conclusion
The study showed that 6.04% (232/3,840) of dogs were suspected of having cardiac disease. Among these, 36.21% (84/232) were confirmed to have myxomatous mitral valve disease (MMVD) by echocardiography, corresponding to 2.19% (84/3,840) of all dogs presented with respiratory signs. Clinically, respiratory signs (dyspnea/pulmonary edema, cough/respiratory inflammation) were the most common manifestations and increased markedly with disease progression, especially at stage C. The intensity of cardiac murmurs increased from stages B1 to B2 to C, with most B2 and C dogs having murmurs ≥ grade III/VI. Regarding diagnostic imaging, VHS increased progressively with disease stage; LA/Ao increased significantly and met ACVIM criteria for stage B2 classification; LVIDd and LVIDs increased, reflecting left ventricular remodelling due to chronic volume overload; while FS showed no statistically significant change, supporting that FS is not a sensitive indicator of MMVD severity. NT-proBNP concentrations rose markedly from B1 to C, demonstrating its value in disease staging and reflecting myocardial stress; it is also useful in differentiating cardiac versus respiratory causes and in monitoring disease progression. In terms of treatment, stages B1 and B2 showed very high response rates, whereas stage C showed a marked decline with a high non-response rate. Regular screening combining clinical examination, radiography, echocardiography (LA/Ao, LVIDd/LVIDDN), and NT-proBNP measurement is an appropriate approach to improve diagnostic accuracy, risk stratification, and optimization of treatment outcomes in dogs with MMVD
Author Contributions: Conceptualisation: Thuy Hoang Nguyen, Quynh Ngoc Nhu Nguyen, Loan Vu Thuy Hong Nguyen; Methodology: Thuy Hoang Nguyen, Kinh Van La, Yen Hai Vu; Validation: Kinh Van La, Loan Vu Thuy Hong Nguyen; Formal analysis: Thuy Hoang Nguyen, Quynh Ngoc Nhu Nguyen, Yen Hai Vu; Investigation: Thuy Hoang Nguyen, Quynh Ngoc Nhu Nguyen; Data Curation: Quynh Ngoc Nhu Nguyen, Loan Vu Thuy Hong Nguyen; Writing—Original Draft Preparation: Thuy Hoang Nguyen, Quynh Ngoc Nhu Nguyen, Yen Hai Vu; Writing—Review and Editing: Kinh Van La, Loan Vu Thuy Hong Nguyen. All authors have read and agreed to the published version of the manuscript.
Funding: This research received no external funding.
Data Availability Statement: All the relevant data that support the findings of this study are available from the corresponding author upon reasonable request.
Acknowledgments: The authors sincerely thank Procare Veterinary Hospital for providing the clinical cases presented in this report.
Conflicts of Interest: The authors declare no conflicts of interest.
Artificial Intelligence: AI was not used for this original research article.
References
Boswood A, Häggström J, Gordon SG, Wess G, Stepien RL, Oyama MA, Keene BW, Bonagura J, MacDonald KA, Patteson M, Smith S, Fox PR, Sanderson K, Woolley R, Szatmári V, Menaut P, Church WM, O’Sullivan ML, Jaudon JP, Kresken JG, … Watson P. 2016. Effect of pimobendan in dogs with preclinical myxomatous mitral valve disease and cardiomegaly: The EPIC study – A randomized clinical trial. Journal of Veterinary Internal Medicine, 30(6), 1765–1779. https://doi.org/10.1111/jvim.14586
Borgarelli M, Crosara S, Lamb K, Savarino P, La Rosa G, Tarducci A, Haggstrom J. 2012. Survival characteristics and prognostic variables of dogs with preclinical chronic degenerative mitral valve disease attributable to myxomatous degeneration. Journal of Veterinary Internal Medicine, 26(1), 69–75. https://doi.org/10.1111/j.1939-1676.2011.00860.x
Borgarelli M, Buchanan JW. 2012. Historical review, epidemiology and natural history of degenerative mitral valve disease. Journal of Veterinary Cardiology, 14(1), 93–101. https://doi.org/10.1016/j.jvc.2012.01.011
Buchanan JW, Bücheler J. 1995. Vertebral scale system to measure canine heart size in radiographs. Journal of the American Veterinary Medical Association, 206(2), 194–199.
Cornell CC, Kittleson MD, Della Torre P, Häggström J, Lombard CW, Pedersen HD, Vollmar A, Wey A. 2004. Allometric scaling of M-mode cardiac measurements in normal adult dogs. Journal of Veterinary Internal Medicine, 18(3), 311–321. https://doi.org/10.1892/0891-6640(2004)18<311:asomcm>2.0.co;2
Fox PR. 2012. Pathology of myxomatous mitral valve disease in the dog. Journal of Veterinary Cardiology, 14(1), 103–126. https://doi.org/10.1016/j.jvc.2012.02.001
Goutal CM, Keir I, Kenney S, Rush JE, Freeman LM. 2010. Evaluation of acute congestive heart failure in dogs and cats: 145 cases (2007-2008). Journal of Veterinary Emergency and Critical Care, 20(3), 330–337. https://doi.org/10.1111/j.1476-4431.2010.00524.x
Keene BW, Atkins CE, Bonagura JD, Fox PR, Häggström J, Fuentes VL, Oyama MA, Rush JE, Stepien R, Uechi M. 2019. ACVIM Consensus Guidelines for the diagnosis and treatment of myxomatous mitral valve disease in dogs. Journal of Veterinary Internal Medicine, 33(3), 1127–1140. https://doi.org/10.1111/jvim.15488
Lam MC, Lin CH, Lo PY, Wu HD. 2022. Influence of concurrent lower respiratory tract disease on point-of-care lung ultrasound in small-breed dogs with myxomatous mitral valve disease. Journal of Veterinary Internal Medicine, 36(3), 1075–1081. https://doi.org/10.1111/jvim.16428
Levicar C, Nolte I, Granados-Soler JL, Freise F, Raue JF, Bach JP. 2022. Methods of radiographic measurements of heart and left atrial size in dogs with and without myxomatous mitral valve disease: Intra- and interobserver agreement and practicability of different methods. Animals, 12(19), 2531. https://doi.org/10.3390/ani12192531
Menciotti G, Borgarelli M. 2017. Review of diagnostic and therapeutic approach to canine myxomatous mitral valve disease. Veterinary Sciences, 4(4), 47. https://doi.org/10.3390/vetsci4040047
Oyama MA, Rush JE, Rozanski EA, Fox PR, Reynolds CA, Gordon SG, Bulmer BJ. Lefbom BK, Brown BA, Lehmkuhl LB, Prosek R, Lesser MB, Kraus MS, Bossbaly MJ, Rapoport GS, Boileau JS. 2009. Assessment of serum N-terminal pro-B-type natriuretic peptide concentration for differentiation of congestive heart failure from primary respiratory tract disease as the cause of respiratory signs in dogs. Journal of the American Veterinary Medical Association, 235(11), 1319–1325. https://doi.org/10.2460/javma.235.11.1319
Pedersen HD, Lorentzen KA, Kristensen BO. 1999. Echocardiographic mitral valve prolapse in Cavalier King Charles Spaniels: Epidemiology and prognostic significance for regurgitation. The Veterinary Record, 144(12), 315–320. https://doi.org/10.1136/vr.144.12.315
Pedersen HD, Häggström J, Falk T, Mow T, Olsen LH, Iversen L, Jensen AL. 1999. Auscultation in mild mitral regurgitation in dogs: Observer variation, effects of physical maneuvers, and agreement with color doppler echocardiography and phonocardiography. Journal of Veterinary Internal medicine, 13(1), 56–64.
Turgut K, Naseri A, Ince ME, Süleymanoğlu H, Ertan M, Sağmanlıgil V, Sen I. 2019. Clinical and cardiologic assessment of Anatolian shepherd dogs with asymptomatic degenerative mitral valve disease. International Journal of Advanced and Applied Sciences, 6(7), 29–35. https://doi.org/10.21833/ijaas.2019.07.004
van Staveren MDB, Muis E, Szatmári V. 2023. Self-reported utilization of international guidelines for staging dogs with myxomatous mitral valve degeneration: A survey among veterinary practitioners. Veterinary Sciences, 10(12), 687. https://doi.org/10.3390/vetsci10120687
Disclaimer/Publisher’s Note: The statements, opinions, institutional affiliations, data contained in all publications, and all responsibilities for accuracy are solely those of the individual author(s) and contributor(s) and not of MARCIAS AUSTRALIA and AJAVAS/or the Editor(s). MARCIAS AUSTRALIA and AJAVAS/or the Editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.

