Occupational exposure to Japanese encephalitis in India
Bukar Mustapha1* Garba Ibrahim2 Muhammad A Ado3 Musa A Usman4 Abubakar S Lawan5 Ismail S Musa5 Zara S Nur5
- Department of Community Medicine, IIAHSR, Integral University, Lucknow 226026, India
- Department of Veterinary Public Health and Preventive Medicine, Faculty of Veterinary Medicine, Ahmadu Bello University, Zaria, Nigeria
- Department of Veterinary Public Health and Epidemiology, Sanskaram University, Jhajjar-124108, Haryana, India
- Department of Public Health, Faculty of Health Science, Ahmadu Bello University, Zaria, Nigeria
- Department of Veterinary Public Health and Preventive Medicine, Faculty of Veterinary Medicine, University of Maiduguri, Nigeria
Article Information
- Date Received: 04/01/2026
- Date Revised: 31/03/2026
- Date Accepted: 02/04/2026
- Date Published Online: 27/04/2026
Copyright: © 2026 The Authors. Published by MARCIAS AUSTRALIA, 32 Champion Drive, Rosslea, Queensland 4812, Australia. This is an open access publication under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Citation: Mustapha B, Ibrahim G, Ado MA, Usman MA, Lawan AS, Musa IS, Nur ZS (2026). Occupational exposure to Japanese encephalitis in India. Aust J Agric Vet Anim Sci (AJAVAS), 2(1), 100012 https://doi.org/10.64902/ajavas.2026.100012
Abstract
Japanese Encephalitis (JE) remains endemic in Indian agricultural belts despite vaccination efforts. Historically, a paediatric ailment, new findings indicate a demographic transition towards adult infections. This systematic review compiles evidence on occupational, environmental, and socio-behavioral variables influencing JE transmission in India to guide targeted control initiatives. In accordance with PRISMA 2020 recommendations, electronic databases (PubMed, Scopus, Google Scholar) were searched for published articles between January 2000 and October 2025. Of 160 records, 23 observational, entomological, and veterinary research fulfilled the inclusion criteria. Data were consolidated concerning geographic location, risk factors, and “One Health” indicators. Significant occupational risks were identified for populations engaged in paddy cultivation, tea plantation work, and pig rearing, particularly in Assam, Uttar Pradesh, and Odisha. The review identified key factors reported across studies including: (1) Ecological: Close proximity (<100m) to fragmented rain-fed farmland; (2) Entomological: Peak Culex abundance corresponding with monsoon cycles; and (3) Veterinary: Reported seropositivity in animal hosts in studies conducted in endemic settings. Additionally, a critical gap was identified between community knowledge and preventive practices. JE in India is increasingly manifesting as an occupational hazard for the adult agrarian workforce. Current pediatric-focused strategies are insufficient. An integrated approach incorporating adult vaccination, veterinary sentinel surveillance, and occupational health measures may help reduce transmission risk.
Keywords
Japanese encephalitis, occupational one health epidemiology, vector-borne disease
Highlights
- Japanese Encephalitis (JE) remains endemic in Indian agricultural belts despite vaccination efforts.
- JE in India is increasingly manifesting as an occupational hazard for the adult agrarian workforce.
- Veterinary sentinel surveillance, and occupational safety interventions may help reduce transmission.
1.0 Introduction
Japanese encephalitis (JE) is a viral zoonosis transmitted by mosquitoes, specifically caused by the Japanese encephalitis virus (JEV), a flavivirus that persists in a transmission cycle involving Culex species mosquitoes and amplifying hosts such as domestic pigs and wild birds (Ahmad et al., 2015). JE remains a leading cause of viral encephalitis in Asia. Older estimates reported 67,900 cases a year (Campbell et al., 2011), but new modelling estimates suggest 100,000 cases per year, despite vaccine efforts (Quan et al., 2020; WHO, 2024). India endures an unequal burden, exhibiting verified endemicity in 201 districts across 21 states (Das et al., 2022). The five states of Assam, Bihar, Tamil Nadu, Uttar Pradesh, and West Bengal account for nearly 80% of India’s national burden of JE (Das et al., 2022). Assam is the most impacted state, representing approximately 51% of the nationally reported cases of JE during previous epidemic years (Ahmad et al., 2015). Since the introduction of the live attenuated SA 14-14-2 vaccine for children aged 1-15 years in 2006, there has been an epidemiological shift towards adult cases, with adults now representing the majority of hospital-confirmed JE cases in endemic areas (Medhi et al., 2017).
The shift towards adult disease has increasingly highlighted the occupational and environmental factors associated with JE transmission (Sarkari et al., 2012; WHO/Global burden study, 2022). Agricultural workers (Sahu et al., 2018; Singh et al., 2016), pig farmers (Baruah et al. (2018), tea estate labourers (Kurmi & Kurmi, 2024) in endemic regions represent cohorts with potentially elevated occupational exposure due to their frequent interactions with environments abundant in vectors, increased animal hosts, and infected individuals (Walsh et al., 2022; Kakkar et al., 2017). Understanding occupational risk factors is essential for developing targeted preventive interventions based on the current epidemiology of JE in India. Despite growing recognition of workplace factors in JE transmission, a comprehensive synthesis of data concerning occupational exposure in India is lacking. The existing literature comprises isolated epidemiological studies (Gulati et al. 2011; Singh et al., 2016), outbreak reports (Sahu et al., 2018; Sarkari et al., 2012), and cross-sectional surveys (Sahoo et al., 2021; Kurmi & Kurmi, 2024; Ahmad et al., 2015) from specific geographic regions, devoid of systematic comparison or methodological assessment. A scoping review synthesizing diverse evidence would identify high-risk occupational groups, describe patterns of transmission relevant to occupational setting, evaluate the effectiveness of existing preventive measures, and highlight evidence gaps necessitating further research. This synthesis is crucial for directing occupational health policies and formulating public health interventions targeting susceptible occupational groups in JE-endemic areas. Therefore, the primary objective of this systematic review was to consolidate published research regarding occupational exposure to JE in India. The specific objectives were:
(1) To identify and characterize high-risk occupational groups;
(2) To examine the drivers of occupational exposure and determinants of transmission;
(3) To evaluate socioeconomic and behavioral factors in populations exposed occupationally;
(4) To assess deficiencies in occupational protection and prevention; and
(5) To identify evidence gaps and research priorities for the prevention of occupational JE in India.
2.0 Materials and methods
2.1. Study design
This study was conducted as a systematic review to synthesize existing evidence on occupational exposure to JE in India. The review followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) extension guidelines.
2.2 Search strategy
A search of electronic databases including PubMed, Scopus, and Google Scholar for articles published between January 2000 and October 2025 was conducted. The search approach utilised a combination of MeSH terms and free-text keywords with Boolean operators (AND/OR)
- “Japanese Encephalitis,” “JEV”, “Acute Encephalitis Syndrome” and “AES (terms related to disease).
- “Occupational exposure,” “Risk factors”, “Agriculture”, “Paddy cultivation”, “Pig farming”, “Vector abundance”, “Socio-economic factors”, “Tea Garden workers”, and “One Health” (terms related to exposure).
2.3 Selection criteria
The studies were selected on the basis of predefined inclusion and exclusion criteria.
The Inclusion criteria were:
- Original research articles, including cross-sectional surveys, case-control studies, cohort studies, entomological/vector surveillance, and animal serosurveillance.
- Studies involving humans (occupationally exposed populations), reservoir hosts (pigs, equines, ardeid birds), or vectors (Culex spp.).
- Studies examining environmental or occupational risk factor (e.g., Close proximity to rice paddies, pig farming, agricultural activities.
- Studies conducted in India.
The exclusion criteria were:
- Studies focusing on non-JE pathogens (e.g., tick-borne diseases, West Nile Virus), unless co-infection with JE was assessed.
- Studies conducted outside India.
- Studies limited to clinical, molecular, or vaccine efficacy research without epidemiological, environmental, or occupational relevance.
2.4 Study selection process
A total of 156 records were initially identified across the three databases. After removal of 10 duplicate records, 146 studies remained for title and abstract screening. Study selection and screenings were conducted independently by two reviewers. Discrepancies were resolved through discussion and consensus. Of the 146 records screened, 106 were excluded based on irrelevance (e.g., non-JE focus, non-India setting, or absence of occupational/environmental exposure data). A total of 40 full-text articles were assessed for eligibility, of which 4 could not be retrieved. Of the remaining 36 articles, 17 were excluded due to lack of occupational data linkage. Ultimately, 23 studies met all inclusion criteria and were included in the final review. The study selection process is illustrated in Figure 1.
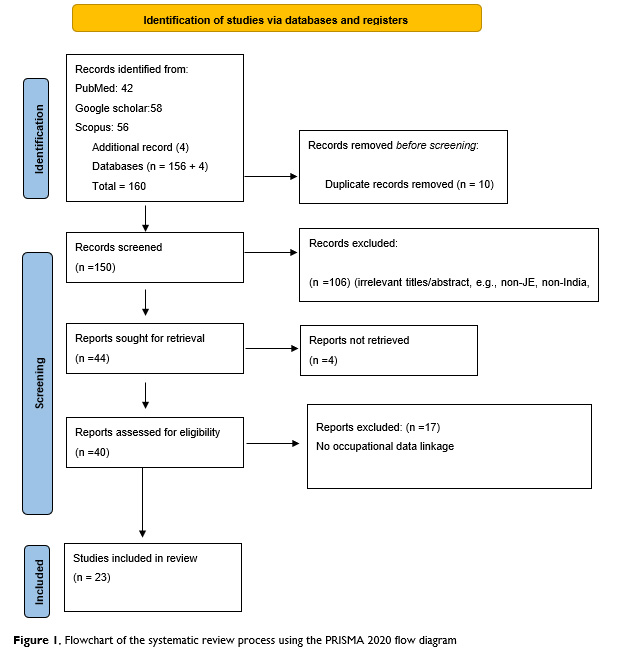
2.5 Data extraction and management
Data were extracted using a standardized data extraction framework. Extracted variables included study location, study design, sample size, occupational groups, environmental and ecological risk factors, and key findings related to JE transmission.
2.6 Data synthesis
A thematic synthesis approach was used to analyse the included studies. Findings were grouped according to predefined review objectives, including:
- High-risk occupational groups
- Drivers of occupational exposure and transmission
- Socioeconomic and behavioural determinants
- Gaps in occupational protection and prevention
- Evidence gaps and research priorities
This approach enabled systematic organization of findings across diverse study designs and settings.
3.0 Results
A total of 23 studies met the inclusion criteria for the final review. The studies were predominantly conducted in high-endemic regions of India, with a significant concentration in Assam and Uttar Pradesh, followed by Odisha, Jharkhand, Bihar, and Tamil Nadu. Findings are presented according to the five predefined review objectives
Table 1. Characteristics of included studies examining occupational, environmental, and epidemiological determinants of JE in India.
| Author (Year) | Location | Study Design | Sample Size | Key Findings |
|---|---|---|---|---|
| Das et al., (2022) | Assam (Northeast India) | Case-control study | 280 (49 Cases) | Proximity to paddy field <100m and pigs increase risk factors; impregnated bed nets were protective. [1] |
| Mote et al. (2024) | Multi-state (Northeast, Northern, Western India) | Case-control study | 1205 pigs | 17.7% prevalence; associated with paddy cultivation and swine density. [1] |
| Kumar et al., (2020) | Goa (west India) | Cross-sectional study | 666 pigs | 7.1% seroprevalence; evidence of ongoing JEV circulation in pigs. [1] |
| Sarkar et al., (2012) | West Bengal (Eastern India) | Hospital-based observational study | 648 AES cases | 74.3% cases near water bodies; higher risk in low SES and monsoon season. [1] |
| Hussain et al., (2022) | Assam (Northeast India) | Cross-sectional study | 423 pigs | High prevalence; waterlogging and monsoon season increased infection risk. [1] |
| Verma et al., (2025) | Uttar Pradesh (North India) | Cross-sectional | 8,635 AES Cases | Age shift from children to adults; higher positivity in males suggests outdoor occupational exposure. [1] |
| Borah et al. (2013) | Assam (Northeast India) | Retrospective Ecological | 178 JE Cases | Rainfall and temperature predict outbreaks with a 1-2-month lag; clusters coincide with peak monsoon agricultural activity. [2] |
| Ahmad et al., (2015) | Assam (Northern India) | Cross-sectional study (KAP) | 218 HCWs | Poor knowledge of zoonotic transmission among hospital staff; 79% relied on TV for info rather than professional training. [2] |
| Sarkari et al., (2012) | Uttar Pradesh (Northern India) | Prospective Clinical | 1,282 Adults | Large adult cohort study during epidemics; high mortality (42%) highlights burden on the adult agricultural workforce. [2] |
| Chaturvedi et al., (2017) | Uttar Pradesh (North India) | Qualitative study (FGDs) | 17 Interviews | Community attributes JE to “filth” or “dirty water” rather than pigs/mosquitoes; low uptake of preventive measures. [2] |
| Medhi et al., (2017) | Assam (Northern India) | Cross-sectional study | 1,707 AES Cases | 94% of cases from rural areas; significant clustering in rainy season (June-Aug) linked to paddy cultivation cycles. [2] |
| Borah et al., (2025) | North East | Scoping review study | Not applicable | Lack of veterinary surveillance; convergence of climate change, deforestation, and livestock identified as drivers. [2] |
| Baruah et al., (2018) | Assam (Northeast India) | Entomological / Zoonotic study | 997 Mosquitoes, 335 Pigs | Strong seasonal correlation found between mosquito abundance, pig seropositivity (22.99%), and human cases. [2] |
| Kurmi & Kurmi., (2024) | Assam (Northeast India) | Occupational cohort study | 280 Workers | Tea garden workers identified as a distinct high-risk group due to biological hazards (insect bites) and lack of protection. [2] |
| Kakkar et al., (2017) | Uttar Pradesh (North India) | Mixed Methods study | 242 Children, 51 Pigs | Lack of vector control and vaccination; gender, religion, and indoor vector density were significant risk factors. [3] |
| Sahu et al., (2018) | Odisha (East India) | Entomological outbreak investigation | 1,136 Mosquitoes | Confirmed JE outbreak in tribal district (Malkangiri); Culex vishnui pool found positive; paddy fields <500m from homes. [3] |
| Kamath et al., (2023) | Jharkhand (East India) | Retrospective Observational study | 14 JE Cases | Post-monsoon peak; rural background and outdoor exposure made patients more susceptible. [4] |
| Singh et al., (2016) | Bihar (East India) | Case-control study | AES Cases | Investigated AES clusters (Muzaffarpur): identified litchi orchards and malnutrition as co-factors distinct from JEV. [4] |
| Ramesh et al., (2015) | Tamil Nadu (South India) | Longitudinal entomological study | 46,343 Mosquitoes | High abundance of Cx. tritaeniorhynchus and Cx. gelidus in cattle sheds; vectors active during dusk (occupational risk). [4] |
| Balakrishnan et al., (2017) | Kerala (South India) | Serological survey | 1,125 Humans | WNV (21.5%) was more prevalent than JEV (15.9%); indicated silent circulation in coastal agricultural districts. [4] |
| Walsh et al., (2022) | India (National) | Ecological modelling study | Not applicable | Risk hotspots converge where “fragmented rain-fed agriculture” meets wetlands, pigs, and wading birds (Ardeidae). [4] |
| Gulati et al., (2011) | Multi-state (India) | Veterinary surveillance study | 3,286 Equines | 10% seropositivity in horses (91.7% in Manipur); indicates JEV circulation in non-pig hosts, broadening risk exposure. [4] |
| Sahoo et al., (2021) | Odisha | Qualitative study (FGD/IDI) | 29 Interviews | Identified gaps in health system preparedness and community participation in tribal districts during outbreaks. [4] |
Note. AES = Acute Encephalitis Syndrome; JE (Japanese Encephalitis); JEV (Japanese Encephalitis Virus); KAP (Knowledge, Attitudes, and Practices); HCWs (Healthcare Workers); FGD (Focus Group Discussion); IDI (In-Depth Interview); WNV (West Nile Virus); Cx. (Culex mosquito species); NR (Not Reported).
3.1 High-risk occupational group
Agricultural workers involved in rice and paddy cultivation were identified as a high-risk occupational group for JE exposure. A hospital-based case-control study in Assam reported proximity to paddy fields within 100 meters of households was significantly associated with JE in multivariable analysis (AOR 8.470, 95% CI 2.025-35.438, p=0.003) (Das et al,. 2022). Similar findings were reported in Dibrugarh, where proximity to rice fields near households was identified as a significant risk factor for JE (P < 0.05) (Borah et al., 2013). Further evidence from West Bengal showed that 74.3% of JE-positive cases resided near rice fields, lakes, or ponds, with most cases occurring during the monsoon and post-monsoon seasons (69.7%), corresponding with peak agricultural activity (Sarkar et al., 2012). Pig-rearing households and pig husbandry workers represent a high-risk occupational group exposed to JE. A field study in Assam reported pig seropositivity of 22.99% alongside 45.65% human JE positivity, with significant correlations between mosquito abundance and pig infection, both reported alongside human cases in endemic settings. Higher human risk was observed in high-burden districts (OR 6.46; 95% CI 3.74-11.15; p < 0.0001) (Baruah et al., 2018). Further evidence from Assam demonstrated high JEV prevalence in swine populations, with 51.6% positivity at farm level and 47.1% at slaughter premises. Key risk factors included water logging near pig shelters (AOR 16.9; 95% CI 6.1-47.3; p < 0.001) and monsoon season (AOR 5.6; 95% CI 2.1-14.9; p < 0.001) (Hussain et al., 2022).
On a broader level, national sero-epidemiological evidence reported an overall JEV seroprevalence of 17.7% in swine populations, with higher prevalence in Assam (30.2%). Increased odds of infection were associated with dense swine populations (OR 2.59; 95% CI 1.77-3.81) and paddy-related environmental exposure (OR 5.77; 95% CI 3.80-8.76), with peak transmission observed during the post-monsoon season (40.3%) (Mote et al., 2024). Additionally, pig husbandry involves close human-animal contact, increasing exposure risk, particularly in settings with limited awareness of transmission pathways (Kakkar et al., 2017). Tea garden workers in Upper Assam represent a high-risk occupational group due to frequent outdoor exposure to mosquito bites. A cross-sectional study in Dibrugarh reported that 80.83% of male and 94.38% of female workers were engaged in outdoor field work. Although a small proportion reported mosquito-borne illnesses (3.33% males; 3.12% females), widespread exposure to biological hazards such as insect bites (35.83% males; 35.62% females) and leech bites (80.83% males; 94.38% females) indicating exposure to mosquito bites in occupational settings (Kurmi & Kurmi, 2024). In addition, large-scale surveillance data from Uttar Pradesh demonstrated a shift in disease burden from children to adults, with higher positivity observed among males, indicating an increased occupational exposure among adult populations engaged in outdoor activities (Verma et al., 2025).
3.2 Drivers of occupational exposure and economic determinants
Across the included studies, occupational exposure to JE was driven by the interaction of ecological, entomological, occupational, and zoonotic factors within agricultural environments. Environmental and ecological conditions were consistently reported across multiple studies as determinants of transmission. The persistence of JE Virus (JEV) is supported by an ecological cycle involving Culex mosquito vectors, domestic pigs, and wild ardeid birds, with humans acting as incidental hosts. Vector breeding was strongly associated with stagnant freshwater environments, including paddy fields, irrigation channels, and marshlands (Borah et al., 2013). Seasonal variation was reported in multiple studies across, with vector density peaking during the monsoon period (June–September), coinciding with intensified agricultural activity and increased waterlogging (Baruah et al., 2018; Borah et al., 2013). The association between irrigated rice agriculture and JE transmission was reported in studies examining occupational and environmental exposure (Keiser et al., 2005). Entomological evidence further demonstrated that Culex tritaeniorhynchus, Cx. vishnui, and Cx. gelidus were the predominant vector species in endemic regions. In rice-growing areas, Cx. tritaeniorhynchus (46.26%) and Cx. gelidus (43.12%) accounted for the majority of collected mosquitoes, with peak abundance during the monsoon and reduced activity in winter months (Ramesh et al., 2015). Vector behaviour was reported as a factor associated with exposure, as exophilic resting in vegetation and animal shelters, combined with opportunistic indoor feeding, increased human–vector contact in both outdoor and peri-domestic settings (Kakkar et al., 2017; Borah et al., 2013). Similarly, A retrospective study from Jharkhand reported a post-monsoon peak in JE cases, with higher occurrence among individuals from rural backgrounds and those engaged in outdoor activities (Kamath et al., 2023). Occupational exposure patterns were closely aligned with ecological transmission windows. Agricultural activities such as ploughing, transplanting, and harvesting occurred predominantly between April and October (Walsh et al., 2022; Borah et al., 2013). Similarly, pig husbandry activities involving feeding, cleaning, and animal care were typically conducted during early morning and evening hours, coinciding with peak mosquito biting times (Baruah et al., 2018; Kakkar et al., 2017). Tea plantation work also involved continuous outdoor exposure in vector-prone environments, particularly during early morning hours (Kurmi & Kurmi, 2024). In several studies, occupational risk was inferred through environmental and livelihood proxies rather than direct exposure measurements.
The interaction of environmental, entomological, and occupational factors reported across the included studies is summarised in Figure 2.

Figure 2
Schematic summary of factors reported across included studies related to occupational exposure to JE in India. Based on thematic synthesis of findings from included studies (n = 23). Illustrates commonly reported factors and does not represent causal relationships.
Economic determinants further reinforced occupational exposure. High-risk populations were predominantly engaged in subsistence agriculture and animal husbandry, with limited alternative livelihood opportunities (Baruah et al., 2018; Ahmad et al., 2015; Walsh et al., 2022; Borah et al., 2013). Fragmented rain-fed agricultural systems were identified as high-risk landscapes, typically associated with economically disadvantaged communities and a disproportionate burden of JE cases (Walsh et al., 2022). In northern India, pig ownership was concentrated among socially marginalized groups, where it served as a primary source of income despite associated health risks (Kakkar et al., 2017). Similarly, tea plantation workers were reported to have significantly lower household incomes compared to state averages, limiting access to preventive measures such as repellents and treated bed nets (Singh et al., 2016). These economic constraints resulted in sustained occupational exposure, even in the presence of risk awareness.
Zoonotic amplification dynamics were consistently reported as important factors for transmission. Domestic pigs were consistently identified as the primary amplifying hosts of JEV, maintaining high levels of viraemia and facilitating virus transmission to mosquito vectors (Baruah et al., 2018; Kakkar et al., 2017; Borah et al., 2013). Higher pig density and close proximity of pig shelters to human dwellings were strongly associated with increased risk of human infection. At the national level, pig density was significantly associated with JE outbreak risk (RR 1.30; 95% CI 1.22-1.39) (Walsh et al., 2022). In addition, poultry density was also associated with increased transmission risk (RR 1.09; 95% CI 1.03-1.15), suggesting a broader role of animal hosts in shaping the transmission landscape (Walsh et al., 2022). The distribution of findings related to JE Virus across human, animal, and environmental settings is summarised in Figure 3.
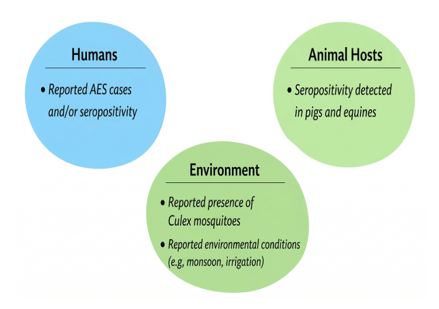
Figure 3
Summary of reported findings on JE Virus in humans, animal hosts, and environmental settings.Based on descriptive synthesis of included studies (n = 23). Provided for illustrative purposes and does not represent causal or quantitative relationships
3.3 Socioeconomic and behavioural factors
Across the included studies, multiple socioeconomic and behavioural factors were identified as important determinants of occupational exposure to JE. Low levels of education and literacy were consistently associated with increased risk. A case-control study reported that poor parental education was significantly associated with Acute Encephalitis Syndrome (AES) (crude OR 2.03; 95% CI 1.33-3.10; p=0.001), and remained independently associated after adjustment (AOR 1.73; 95% CI 1.10-2.74; p=0.018) (Singh et al., 2016). Qualitative studies further demonstrated limited understanding of JE transmission among affected communities, particularly among farmers and pig-rearing households (Chaturvedi et al., 2017; Kakkar et al., 2017). Misconceptions were common, with the disease frequently attributed to “filthy” environmental conditions rather than mosquito vectors or animal hosts. In some settings, pigs were not identified as a risk factor unless specifically probed, and were often not perceived as directly involved in disease transmission (Chaturvedi et al., 2017).Health-seeking behaviour was also identified as a key determinant influencing exposure and outcomes. Delayed care-seeking was frequently reported, with patients presenting to healthcare facilities after 7-10 days of illness (Singh et al., 2016). Initial care was commonly sought from informal or non-formal healthcare providers, with limited involvement of formal health systems (Chaturvedi et al., 2017). In occupational settings, continued work during illness was also observed among tea plantation workers, limited access to workplace healthcare and first-aid services resulted in workers continuing field activities despite illness or injury, prolonging exposure to vector-prone environments (Kurmi & Kurmi, 2024).
Perceptions of disease risk varied across population groups. In some communities, JE was recognised as a serious disease but was not prioritised relative to other common health conditions such as diarrhoea, malaria, or malnutrition (Chaturvedi et al., 2017). In contrast, healthcare workers demonstrated higher awareness of occupational risk, with 83% correctly identifying JE risk and 74.3% expressing favourable attitudes towards preventive measures (Ahmad et al., 2015). However, knowledge gaps remained, including limited awareness of seasonal transmission patterns (89%) and misconceptions regarding appropriate treatment, with 70.7% incorrectly identifying antibiotics as a primary treatment option (Ahmad et al., 2015). Gender differences in occupational exposure were also reported. In tea plantation settings, women constituted a larger proportion of the workforce engaged in outdoor activities (94.38% of females vs 80.83% of males), resulting in increased exposure to vector-prone environments (Kurmi & Kurmi, 2024). At the same time, some studies reported higher JE risk among males, particularly in children, which was attributed to increased outdoor exposure during peak mosquito activity periods (OR 4.83; 95% CI 1.68-13.88; p=0.003) (Kakkar et al., 2017). Gender-related differences in occupational roles and activity patterns contributed to variation in exposure risk. Social marginalization and economic disadvantage further concentrated risk among specific population groups. Studies reported significantly higher AES risk among scheduled castes (OR 2.18; 95% CI 1.10-4.30; p=0.028) and other backward castes (OR 1.91; 95% CI 1.00-3.66; p=0.049) (Singh et al., 2016). In some regions, pig-rearing an occupation associated with increased JE risk was predominantly practiced by socially marginalized communities with limited alternative livelihood options (Kakkar et al., 2017; Chaturvedi et al., 2017). These populations were often located at the periphery of villages and had reduced access to healthcare and preventive infrastructure.
3.4 Gaps in occupational protection and prevention
The included studies highlighted several gaps in occupational protection and prevention measures among high-risk populations. Limited use of personal protective measures, including insecticide-treated bed nets and repellents, was reported across multiple settings, particularly in rural and agricultural communities (Kakkar et al., 2017; Chaturvedi et al., 2017). Inadequate implementation of vector control strategies in endemic regions further contributed to sustained transmission (Sahu et al., 2018; Kakkar et al., 2017). Vaccination programmes were predominantly focused on paediatric populations, with limited emphasis on adults engaged in high-risk occupations such as agriculture and pig farming (Medhi et al., 2017; Kakkar et al., 2017). In addition, workplace-level health and safety interventions were largely absent, particularly in informal sectors such as agriculture and tea plantations, where workers reported limited access to protective measures and occupational health services (Kurmi & Kurmi, 2024). Health system limitations were also identified. Several studies reported delayed diagnosis and treatment, low community awareness, and inadequate preparedness of healthcare systems during outbreaks (Sahoo et al., 2021; Chaturvedi et al., 2017). Gaps in surveillance systems and limited integration of veterinary and human health data were also noted, reducing the effectiveness of early warning and response mechanisms (Walsh et al., 2022; Borah et al., 2025).
3.5 Knowledge gaps and research needs
The review identified knowledge gaps in the existing literature on occupational exposure to JE. First, there is a dearth of studies specifically quantifying occupational exposure, because previous studies relied on indirect environmental or livelihood proxies rather than direct measurement of occupational risk (Kakkar et al., 2017; Walsh et al., 2022). Secondly, the limited integration of human, animal, and environmental data indicates gaps in the application of a One Health approach (Durrance-Bagale et al., 2021; Walsh et al., 2022). Thirdly, geographical disparities in research were observed, in which the majority of previous studies were concentrated in high-endemic regions such as Assam and Uttar Pradesh, while other regions remained under-represented (Das et al., 2022; Sahu et al., 2018). Fourthly, there is limited evidence evaluating the effectiveness of occupational health interventions, vector control strategies, and adult vaccination programmes in reducing JE transmission because most studies primarily focussed on epidemiological patterns rather than intervention outcomes (Kakkar et al., 2017; Sahoo et al., 2021; Walsh et al., 2022).
4.0 Discussion
The findings indicate that JE is not only a vector-borne disease but also an occupationally mediated health risk in endemic agricultural settings. The review identified agricultural workers, pig-rearing households, and tea plantation workers, highlighting the role of occupational exposure to JE transmission. These populations are routinely exposed to environments that support mosquito breeding, including paddy fields, stagnant water bodies, and livestock shelters (Borah et al., 2013; Baruah et al., 2018). The overlap of occupational activities with peak vector density periods, particularly during the monsoon season, appears to play a central role in shaping exposure patterns (Walsh et al., 2022). This is consistent with previous evidence highlighting the importance of environmental and occupational interfaces in JE transmission (Kakkar et al., 2017). This shift towards an increasing adult disease burden is further supported by large-scale surveillance data from Uttar Pradesh (Verma et al., 2025), supporting the role of occupational exposure in shaping current epidemiological patterns
4.2 Occupational and ecological drivers
A key finding of this review is the role of ecological and zoonotic factors in sustaining transmission. The interaction between mosquito vectors, amplifying hosts such as pigs, and human populations reflects a complex transmission system closely linked to agricultural practices (Baruah et al., 2018; Walsh et al., 2022). The presence of pigs in close proximity to human dwellings, combined with favourable environmental conditions for vector breeding was reported in settings where human cases occurred (Kakkar et al., 2017). These findings support the relevance of a One Health approach in understanding and managing JE transmission (Durrance-Bagale et al., 2021). The findings of this review are consistent with the evidence reported across JE-endemic regions in Asia. Occupational exposure linked to agricultural practices and proximity to animal hosts plays a key role in transmission dynamics (Walsh et al., 2022; Kakkar et al., 2017). Similar patterns of risk associated with irrigated agricultural environments, vector abundance, and human animal interaction have been described in multiple endemic settings. However, the present review highlights a more pronounced occupational dimension in the Indian context, particularly with respect to the increasing burden among adult agricultural workers (Medhi et al., 2017; Walsh et al., 2022). This shift suggested that, while ecological factors remains important, the epidemiological status of JE in India is increasingly shaped by occupational and socioeconomic factors, including labour patterns, livelihood dependence, and limited access to protective measures.
4.3 Socioeconomic and behavioural determinants
Socioeconomic and behavioural determinants played an important role in shaping occupational exposure to JE. Limited awareness of disease transmission, misconceptions regarding causation, and delayed health-seeking behaviour were consistently reported across studies (Chaturvedi et al., 2017; Singh et al., 2016). These knowledge gaps may contribute to inadequate adoption of preventive measures and prolonged exposure to vector-prone environments. In addition, economic constraints and dependence on agriculture or animal husbandry appear to limit the feasibility of implementing protective strategies, thereby supporting exposure risk (Walsh et al., 2022). Social factors, including gender roles and marginalised populations, further influence exposure patterns by shaping occupational roles and access to resources (Kakkar et al., 2017). For instance, differential engagement in outdoor labour between males and females and limited access to health information or preventive tools may increase vulnerability among specific population groups. These findings highlight the interaction between socioeconomic disadvantage and occupational risk which suggests that structural inequalities play a significant role in sustaining JE transmission especially in endemic settings.
4.4 Implications for public health and policy
The review also identified important gaps in occupational protection and prevention. Current JE control strategies in India have largely focused on paediatric vaccination programmes, with comparatively less attention given to adults engaged in high-risk occupations (Medhi et al., 2017). In addition, limited use of personal protective measures and gaps in vector control may contribute to continued transmission in endemic areas (Sahu et al., 2018). Strengthening occupational health interventions and integrating them into existing public health programmes may help reduce exposure among vulnerable populations. From a policy perspective, the findings suggest the need for a more comprehensive approach to JE prevention that incorporates occupational risk. This may include expanding vaccination strategies to include high-risk adult populations, improving vector control in agricultural settings, and promoting community-based awareness programmes that address misconceptions about disease transmission (Ahmad et al., 2015). Enhancing surveillance systems through better integration of human, animal, and environmental data may also support early detection and response (Walsh et al., 2022).
4.5 Strengths and limitations
This review has several strengths. It provides a focused synthesis of occupational exposure to JE in India and it integrates evidence from epidemiological, entomological, and veterinary studies within a One Health framework. However, several limitations should be considered when interpreting the findings. The number of eligible studies was limited, and there was variability in study design, populations, and outcome measures, which may restrict direct comparability across studies. Many studies relied on indirect measures of occupational exposure, such as environmental or livelihood proxies, rather than direct measurement of occupational risk affecting the precision of findings (Kakkar et al., 2017). In addition, the concentration of studies in specific geographic regions, particularly high-endemic areas, may limit the generalizability of the findings to other regions of India. Furthermore, potential publication bias cannot be excluded, as studies reporting significant associations are more likely to be published. As this was a scoping review, a formal quality assessment of included studies was not conducted, which may influence the strength of the evidence synthesized. Despite these limitations, the review provides a comprehensive synthesis of available evidence and highlights important gaps for future research and policy development.
4.6 Future research
Future research should aim to quantify occupational exposure more directly and evaluate the effectiveness of targeted interventions, including workplace-based protective measures and adult vaccination strategies. Further studies incorporating integrated One Health surveillance approaches may also improve understanding of transmission dynamics and inform more effective control strategies.
5.0 Conclusion
JE in India has transitioned from a childhood illness to an occupational risk impacting adults, particularly agricultural labourers. This review of 23 studies identified four key findings:
(1) Veterinary findings, including reported seropositivity in pigs and equines, were described in studies conducted in endemic settings.
(2) Communities misunderstood transmission, attributing JE to ‘filth’ rather than vectors, undermining preventive behaviour.
(3) Risk was reported in settings where rain-fed agriculture meets wetlands and animal hosts co-occur.
(4) Adults engaged in paddy cultivation, tea plantation work and pig farming were frequently represented in studies conducted in high-burden settings. As such, current child-focused policies may not fully address adult occupational risks. Effective control may require consideration of adult vaccination in high risk occupational groups and strengthen vector management in agricultural settings. Community awareness programmes should address misconceptions about disease transmission and farm workers need to follow occupational health requirements. Future research should evaluate workplace interventions and measure occupational risk to guide evidence-based policy.
Author Contributions: Conceptualisation, Bukar Mustapha, Garba Ibrahim and Muhammad A Ado; methodology, Musa A Usman, Bukar Mustapha. Zara S Nur; resources, Zara S Nur, Musa A Usman, Abubakar S Lawan; writing original draft preparation, Bukar Mustapha, Garba Ibrahim, Muhammad A Ado, Musa A Usman, Zara S Nur; writing review and editing, Bukar Mustapha, Garba Ibrahim, Muhammad A Ado, Musa Usman Abubakar, Abubakar S Lawan, Ismail S Musa, Zara S Nur; All authors have read and agreed to the published version of the manuscript.
Funding: This research received no external funding.
Ethics Approval Statement: Not applicable.
Informed Consent Statement: Not applicable.
Acknowledgments: The authors acknowledge the Department of Community Medicine, IIAHSR, Integral University Lucknow India;
Department of Veterinary Public Health and Preventive Medicine, Faculty of Veterinary Medicine, Ahmadu Bello University Zaria Nigeria;
Department of Veterinary Public Health and Epidemiology, Sanskaram University, Jhajjar, Haryana, India and Department of Public Health, Faculty of Health Science, Ahmadu Bello University Zaria Nigeria.
Conflicts of Interest: The authors declare no conflicts of interest.
Artificial Intelligence: AI was not used for this study.
References
Ahmad A, Khan MU, Gogoi LJ, Kalita M, Sikdar AP, Pandey S, Dhingra S. 2015. Japanese encephalitis in Assam, India: Need to increase healthcare workers’ understanding to improve health care. PLOS ONE, 10(8), e0135767. https://doi.org/10.1371/journal.pone.0135767
Balakrishnan A, Thekkekare RJ, Sapkal G, Tandale BV. 2017. Seroprevalence of Japanese encephalitis virus & West Nile virus in Alappuzha district, Kerala. Indian Journal of Medical Research, 146(Supplement), 70–76. https://doi.org/10.4103/ijmr.IJMR_1638_15
Baruah A, Hazarika RA, Barman NN, Islam S, Gulati BR. 2018. Mosquito abundance and pig seropositivity as a correlate of Japanese encephalitis in human population in Assam, India. Journal of Vector Borne Diseases, 55(4), 291–296. https://doi.org/10.4103/0972-9062.256564
Borah J, Dutta P, Khan SA, Mahanta J. 2013. Association of weather and anthropogenic factors for transmission of Japanese encephalitis in an endemic area of India. EcoHealth, 10(2), 129-136. https://doi.org/10.1007/s10393-013-0849-z
Borah P, Das P, Bordoloi R, Begum R, Hazarika G, Saikia DP, Chutia BC, Baruah C, Dutta R. 2025. Prevalence of zoonotic diseases in the Northeastern region, one health perspective. Animals and Zoonoses, 1, 178–187. https://doi.org/10.1016/j.azn.2025.01.001
Campbell GL, Hills SL, Fischer M, Jacobson JA, Hoke CH, Hombach JM, Marfin AA, Solomon T, Tsai TF, Tsu VD, Ginsburg AS. 2011. Estimated global incidence of Japanese encephalitis: a systematic review. Bulletin of the World Health Organization, 89(10), 766-774E. https://doi.org/10.2471/BLT.10.085233
Chaturvedi S, Sharma N, Kakkar M. 2017. Perceptions, practices and health seeking behaviour constrain JE/AES interventions in high endemic district of North India. BMC Public Health, 17, 625. https://doi.org/10.1186/s12889-017-4654-4
Das BR, Kakoti G, Chetri M, Biswanath P. 2022. Eco-epidemiological risk factors for Japanese encephalitis in the endemic region of North East India: A hospital-based case-control study. Encephalitis, 2(4), 115-124. https://doi.org/10.47936/encephalitis.2022.00066
Durrance-Bagale A, Rudge JW, Singh NB, Belmain SR, Howard N. 2021. Drivers of zoonotic disease risk in the Indian subcontinent: A scoping review. One Health, 13, 100310. https://doi.org/10.1016/j.onehlt.2021.100310
Gulati BR, Singha H, Singh BK, Virmani N, Khurana SK, Singh RK. 2011. Serosurveillance for Japanese encephalitis virus infection among equines in India. Journal of Veterinary Science, 12(4), 341–345. https://doi.org/10.4142/jvs.2011.12.4.341
Hussain M, Dhanze H, Mehta D, Kumar MS, Gandham RK, Gupta M, … Singh BB. 2022. Prevalence and risk factors associated with Japanese encephalitis virus infection in swine population of Assam, India. Asian Pacific Journal of Tropical Medicine, 15(11), 503–510. https://doi.org/10.4103/1995-7645.359788
Kakkar M, Chaturvedi S, Saxena VK, Dhole TN, Kumar A, Rogawski ET, Abbas S, Venkataramanan VV, Chatterjee P. 2017. Identifying sources, pathways and risk drivers in ecosystems of Japanese Encephalitis in an epidemic-prone north Indian district. PLOS ONE, 12(5), e0175745. https://doi.org/10.1371/journal.pone.0175745
Kamath SD, Jha B, Ahmed T, Sarkar N. 2023. A profile study of Japanese encephalitis in an industrial hospital in eastern India. Cureus, 15(5), e38455. https://doi.org/10.7759/cureus.38455
Kumar HBC, Dhanze H, Bhilegaonkar KN, Chakurkar EB, Kumar A, Yathish HM. 2020. Serological evidence of Japanese encephalitis virus infection in pigs in a low human incidence state, Goa, India. Preventive Veterinary Medicine, 175, 104882. https://doi.org/10.1016/j.prevetmed.2020.104882
Kurmi S, Kurmi D. 2024. Occupational health hazard among the tea garden workers- a study in a district of upper Assam. IOSR Journal of Nursing and Health Science, 13(5), 01-05. https://doi.org/10.9790/1959-1305010105
Medhi M, Saikia L, Patgiri SJ, Lahkar V, Hussain ME, Kakati S. 2017. Incidence of Japanese Encephalitis amongst acute encephalitis syndrome cases in upper Assam districts from 2012 to 2014: A report from a tertiary care hospital. Indian Journal of Medical Research, 146(2), 267–271. https://doi.org/10.4103/ijmr.IJMR_1303_15
Mote AB, Singh BB, Mehta D, Patel SM, Dhanalakshmi M, Suman Kumar M, … Dhanze H. 2024. Sero-epidemiology and risk factor investigation for exposure to Japanese encephalitis virus in swine populations reared in climatically diverse regions of India. Tropical Animal Health and Production, 56(9), 397. https://doi.org/10.1007/s11250-024-04243-0
Quan TM, Thao TTN, Duy NM, Nhat TM, Clapham H. 2020. Estimates of the global burden of Japanese encephalitis and the impact of vaccination from 2000-2015. eLife, 9, e51027. https://doi.org/10.7554/eLife.51027
Ramesh D, Muniaraj M, Samuel PP, Thenmozhi V, Venkatesh A, Nagaraj J, Tyagi BK. 2015. Seasonal abundance and role of predominant Japanese encephalitis vectors Culex tritaeniorhynchus and Cx. gelidus theobald in Cuddalore District, Tamil Nadu. Indian Journal of Medical Research, 142(Suppl), 23–29. https://doi.org/10.4103/0971-5916.176607
Sahoo KC, Negi S, Dash GC, Sahoo RK, Kshatri JS, Panda S, Pattanaik M, Badaik G, Pati S, Bhattacharya D. 2021. Health system preparedness and community participation in Japanese encephalitis/acute encephalitis syndrome (JE/AES) prevention in a tribal district of Odisha, India. Indian Journal of Medical Research, 153(3), 388–393. https://doi.org/10.4103/ijmr.IJMR_645_21
Sahu SS, Dash S, Sonia T, Muthukumaravel S, Sankari T, Gunasekaran K, Jambulingam P. 2018. Entomological investigation of Japanese encephalitis outbreak in Malkangiri district of Odisha state, India. Memórias do Instituto Oswaldo Cruz, 113(6), e170499. https://doi.org/10.1590/0074-02760170499
Sarkar A, Taraphdar D, Mukhopadhyay BB, Kumar M, Mukhopadhyay SK, Chatterjee S. 2012. Influence of socio-economic status and environmental factors on serologically diagnosed Japanese encephalitis cases in the state of West Bengal, India during 2005–2010. Health, 4(1), 6-12. http://dx.doi.org/10.4236/health.2012.41002
Sarkari NBS, Thacker AK, Barthwal SP, Mishra VK, Prapann S, Srivastava D, Sarkari M. 2012. Japanese encephalitis (JE). Part I: Clinical profile of 1,282 adult acute cases of four epidemics. Journal of Neurology, 259(1), 47-57. https://doi.org/10.1007/s00415-011-6118-6
Singh GK, Singh CM, Ranjan A, Agarwal N, Pandey S, Kumar P, Setty G. 2016. Determinants of Acute Encephalitis Syndrome (AES) in Muzaffarpur District of Bihar, India: A case-control study. Clinical Epidemiology and Global Health, 4(4), 181-187. https://doi.org/10.1016/j.cegh.2016.05.002
Verma A, Radera S, Jain A, Prakash O, Verma AK. 2025. The trend of Japanese encephalitis in Uttar Pradesh, India. Annals of African Medicine, 24(2), 263-267. https://doi.org/10.4103/aam.aam_185_24
Walsh MG, Pattanaik A, Vyas N, Saxena D, Webb C, Sawleshwarkar S, Mukhopadhyay C. 2022. High-risk landscapes of Japanese encephalitis virus outbreaks in India converge on wetlands, rain-fed agriculture, wild Ardeidae, and domestic pigs and chickens. International Journal of Epidemiology, 51(5), 1408-1418. https://doi.org/10.1093/ije/dyac050
World Health Organization. 2024. Japanese encephalitis. WHO Fact Sheets (August 5). https://www.who.int/news-room/fact-sheets/detail/japanese-encephalitis
Disclaimer/Publisher’s Note: The statements, opinions, institutional affiliations, data contained in all publications, and all responsibilities for accuracy are solely those of the individual author(s) and contributor(s) and not of MARCIAS AUSTRALIA and AJAVAS/or the Editor(s). MARCIAS AUSTRALIA and AJAVAS/or the Editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.

